Acidic Fibroblast Growth Factor in Spinal Cord Injury
Article information
Abstract
Spinal cord injury (SCI), with an incidence rate of 246 per million person-years among adults in Taiwan, remains a devastating disease in the modern day. Elderly men with lower socioeconomic status have an even higher risk for SCI. Despite advances made in medicine and technology to date, there are few effective treatments for SCI due to limitations in the regenerative capacity of the adult central nervous system. Experiments and clinical trials have explored neuro-regeneration in human SCI, encompassing cell- and molecule-based therapies. Furthermore, strategies have aimed at restoring connections, including autologous peripheral nerve grafts and biomaterial scaffolds that theoretically promote axonal growth. Most molecule-based therapies target the modulation of inhibitory molecules to promote axonal growth, degrade glial scarring obstacles, and stimulate intrinsic regenerative capacity. Among them, acidic fibroblast growth factor (aFGF) has been investigated for nerve repair; it is mitogenic and pluripotent in nature and could enhance axonal growth and mitigate glial scarring. For more than 2 decades, the authors have conducted multiple trials, including human and animal experiments, using aFGF to repair nerve injuries, including central and peripheral nerves. In these trials, aFGF has shown promise for neural regeneration, and in the future, more trials and applications should investigate aFGF as a neurotrophic factor. Focusing on aFGF, the current review aimed to summarize the historical evolution of the utilization of aFGF in SCI and nerve injuries, to present applications and trials, to summarize briefly its possible mechanisms, and to provide future perspectives.
INTRODUCTION
Spinal cord injury (SCI) is still a devastating disease in the current era. Due to the lack of effective treatment and limited regenerative capacity in the adult human spinal cord, life-long disability and accompanying complications are not uncommon consequences of severe SCI. The incidences of SCI are variable among regions and societies, and have been reported worldwide through the years. In Taiwan, a country where legislation made helmets mandatory for motorcyclists, the incidence rate of adult SCI was estimated at 246 per million-person-years [1]. In accordance with that reported around the world, road traffic accidents and falls from heights remain the leading causes of SCI [2-4]. Prevention of SCI is certainly the best strategy, since many of the causes sometimes might be avoidable. On the other hand, for those patients with chronic SCI, the most definite treatment currently available is still limited to neurorehabilitation and passive management of complications and comorbidities. There are technologies of exo-skeleton, electrophysiological stimulation, and brain-computer-interface-based prosthesis undergoing development and preclinical experiments [5-8]. However, from the patients’ and families’ standpoint, regenerative medicine is still the place where they send their hopes.
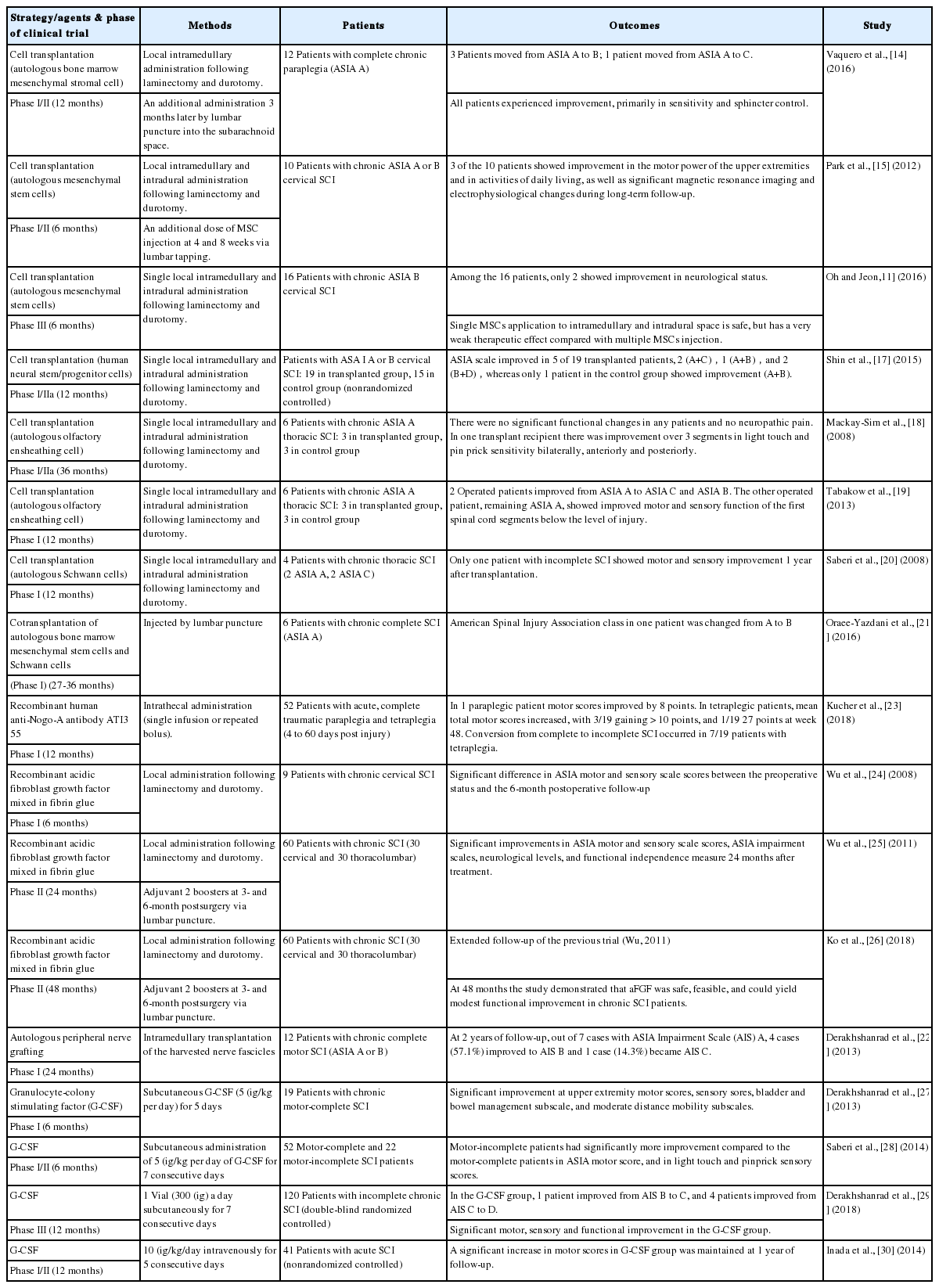
The pathophysiology of SCI is a sequential combination of primary trauma and secondary injury (Fig. 1). There are already many basic studies regarding regenerative therapy for chronic SCI, as well as review articles to catalog the existing clinical trials (Table 1) [9-30]. In general, these therapies fall into 2 categories: cell-based and molecule-based strategies. In the former, the investigators deliver specific types of cells into the damaged spinal cord. These transplanted cells mainly involve human embryonic stem cells, adult neural stem cells from cell lines of fetal brains or fetal spinal cords, autologous or fetal olfactory ensheathing cells, umbilical cord blood mononuclear cells, autologous bone marrow adult mesenchymal stem cells, and autologous Schwann cells from peripheral nerves. Cellular transplantation to chronic lesions is delivered to fill and bridge the cyst or cavity inside the lesioned spinal cord, to replace dead cells with new neurons or myelinating cells, and to create a favorable environment for axon regeneration against the hostile surroundings in the central nervous system (CNS). Broadly speaking, the utilization of autologous peripheral nerve graft (PNG) is also associated with this category. Autologous grafts act as immunogenically inert scaffolds, providing appropriate neurotrophic factors and viable Schwann cells for axonal regeneration [31]. Thanks to its multiple functions, PNG has been widely used in experiments and clinical trials. To sum up, there have been promising results of the cell-based repair strategies in animal experiments. Some related clinical trials are currently underway.
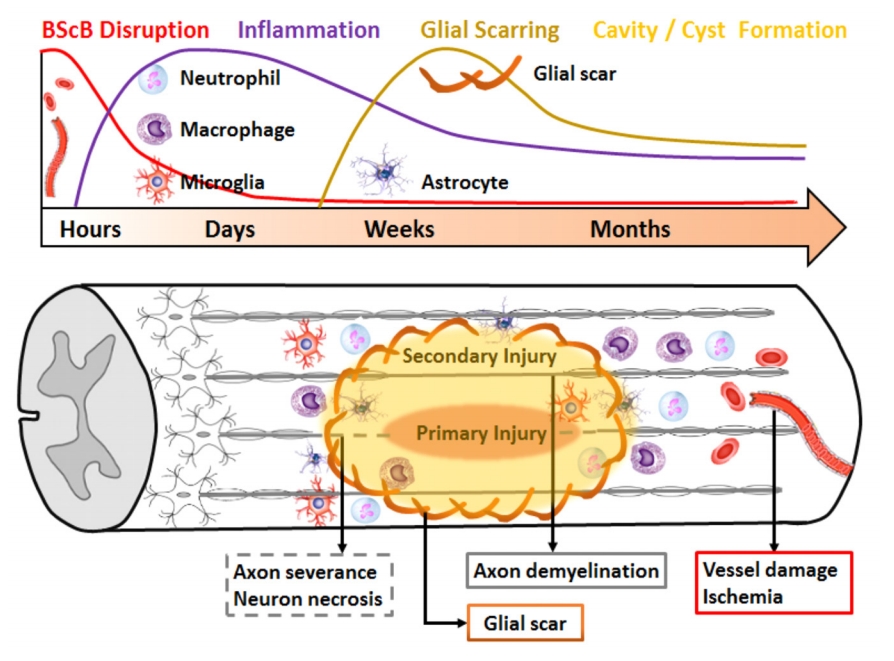
Brief pathophysiology of SCI. The sequential damage of SCI is a combination of the primary trauma and secondary injury. The primary mechanical injury directly injures the axons and breaks down the blood-spinal cord barrier (BScB) within the initial hours. In the following days, secondary injury flare-up occurs with the infiltration of the immune cells (macrophages, neutrophils, and microglia) into the injured site. Also, within days to weeks, the astrocytes are activated and form a glial scar to envelop the injured area and to limit the range of the inflammatory response. In the following weeks to months, the glial scar reconstructs a firm shell surrounding the injured area, and eventually forms a cyst or cavity consisting of necrotic cells inside.
The mechanisms of molecular therapies for nerve regeneration after chronic SCI in part involve the modulation of the inhibitory molecule in the CNS. Given the fact that axonal growth after injury can be inhibited by myelin and chondroitin sulfate proteoglycans (CSPG), which are in part controlled by RhoGTPase, pharmacological molecules aiming at suppression of Rho-GTPase activity have been tested in vitro. The CSPG-dependent inhibition of neurite extension could be overcome, and enhancement of axonal regeneration was discovered [32,33]. In addition to CSPG, the Nogo-A is another notorious growth inhibitory molecule. A variety of anti-Nogo therapies were suggested, including human antibodies against Nogo-A, molecules targeting Nogo receptors, and genetic deletion of Nogo-A or Nogo receptors [34-36]. With regard to the established obstacle formed by glial scarring in the injured spinal cord, degradation of inhibitory CSPG by delivery of the bacterial enzyme chondroitinase ABC might give rise to promising results in the treatment of subchronic and chronic SCI [37,38].
In contrast to the growth inhibitory molecules, a lack of growthpermissive molecules also contributes to the diminished intrinsic ability of regeneration in the CNS. There is plenty of growthpermissive neurotrophic factors allowing axons to lengthen during the phase of development. However, the expression of these factors slumps in the adult CNS. Studies focused on the administration of the neurotrophin family, such as nerve growth factor, brain-derived neurotrophic factor, and neurotrophin-3, have suggested favorable outcomes in experiments [39]. Apart from the neurotrophins, the family of fibroblast growth factors (FGF) is also essentially crucial in normal development. Both the neurotrophins and the FGFs, as well as their respective receptors, have been shown to be upregulated after experimental CNS injury [40]. It is evident that acidic FGF (aFGF) and basic FGF (bFGF) are potent trophic factors for many populations of CNS neurons and could potentially play a significant role in nervous system development [41]. Likewise, the FGFs are expected to become another clue to repair the injured spinal cord. For over 2 decades the authors’ laboratory, hosted by Professor Cheng, has conducted a series of investigations focusing on the application of aFGF in the injured nerve tissue to promote neural regeneration. This review article aims to inspect the historical evolution of the utilization of aFGF, and to present the application in current experiments and trials and its possible mechanisms, and to provide future perspectives.
HISTORY OF FGF
The FGFs are a family of endogenous polypeptides initially found in the pituitary and brain. They were first described in 1973 by Armelin [42] in bovine pituitary extracts as a peptide distinct from the classical pituitary hormones. The purification and research soon thrived in the 1970s and 1980s. Unlike general pituitary hormones, FGFs were found to be able to stimulate the growth and proliferation of mouse fibroblast. As a result, it was named FGF [43,44]. Using the laboratory method, such as isoelectric focusing, these pituitary extracts could be further fractionated into acidic and basic FGF (i.e., FGF1 and FGF2) [45].
Later on, a pair of heparin-binding growth factors (HBGF-1 and HBGF-2) and a pair of endothelium cell growth factors (ECGF1 and ECGF 2) were discovered. The investigators eventually realized that the separate polypeptides were actually the same group of molecules. That is to say, aFGF is the same with FGF1, HBGF-1, and ECGF-1, while bFGF is also referred to as FGF2, HBGF-2, and ECGF-2. There are 23 members (22 in the vertebrates) in the FGF family. Both the gene structure and amino acid sequence are highly conserved between vertebrate species [46]. Given the fact that the FGFs were historically isolated from different approaches, the investigators eventually revealed their pluripotent feature. The FGFs have diverse roles in regulating cell proliferation, migration and differentiation during embryonic development, and some of the FGF family in adult tissue are important for neuronal signal transduction in the central and peripheral nervous system [46]. Nowadays, it is well documented that the FGFs are a large family of heparin-binding proteins. They interact with membrane-associated proteoglycans and bind to 4 FGF receptor subtypes [47].
The FGFs are potent angiogenic factors. They may indirectly control neovascularization in concert with other growth factors [48]. As well as stimulating blood vessel growth, FGFs are important in wound healing. It is suggested that FGF2 has potential for cell proliferation, wound re-epithelialization, and collagen deposition [49]. The FGF7 and FGF10, also known as Keratinocyte Growth Factors (KGF and KGF2, respectively), promote the repair of injured skin and mucosal tissues by stimulating the proliferation, migration and differentiation of epithelial cells [50]. In the CNS, aFGF and bFGF are involved in the regulation of synaptic plasticity and processes, and have been widely tested for their potential therapeutic effects [47,51]. In a rat model, the application of aFGF accelerates the crush-injured sciatic nerve to regenerate both motor and sensory axons. It is suggested that aFGF may be clinically useful in the treatment of peripheral neuropathy in humans [52]. Recently, researchers found that aFGF improves functional recovery through inducing PRDX1 (peroxiredoxin 1) to regulate autophagy and anti-ROS (reactive oxygen species) after SCI in rat model. Hence, it is expected that aFGF probably play some roles in the repair of nerve injury, both in peripheral and in CNSs [53].
ACIDIC FGF USED FOR NEURAL REGENERATION
The fact that FGFs express mitogenic and pluripotent activities, and are closely related to the development and maintenance of neural structures, provides a clue to therapeutic application to repair the injured nerve [54]. Cordeiro et al. [55] demonstrated an increased number of myelinated axons and a greater number of primary sensory and motor neurons in animals treated with aFGF. They suggested aFGF to be a putative neurotrophic factor on peripheral nerve regeneration in vivo. With the evidence of rapid angiogenesis and neurogenesis induced by aFGF through a 15-mm surgical gap, Walter et al. [56] demonstrated the functional motor recovery of transected peripheral nerves with the application of aFGF. It has been gradually accepted that aFGF is beneficial for peripheral neural regeneration. Nowadays, not only in animal studies but also in clinical scenarios, the usage of aFGF has been proven to benefit the regeneration of the injured peripheral nerves. Tsai et al. [57] first reported a clinical trial of the administration of aFGF to patients with common peroneal nerve lesions. The results revealed a significantly increased average muscle strength score in the group of surgical repair with fibrin glue added with aFGF, compared to the other 2 groups of surgical repair only and of no any surgical intervention at 6-month follow-up. The positive role of aFGF in nerve regeneration is in little doubt; however, the detailed mechanism is still unclear. Both clinical and laboratory studies are still in progress.
PERIPHERAL NERVE GRAFTING WITH ACIDIC FGF IN SCI
It is well known that there is an environmental difference between the central and the peripheral nervous systems. Therefore, it was generally accepted that the neurological deficit following SCI was irreversible. The concept was unyielding until Cheng et al. [58] reported their creative study in 1996. Using the complete transected thoracic spinal cord in adult rats, they devised a novel strategy of PNGs bridging the spinal cord stumps, supplemented with fibrin glue mixed with aFGF. They found the corticospinal tract regenerated through the grafted area and several bulbospinal pathways as well. They demonstrated partial restoration of the hind limb function in adult paraplegic rats. The data have suggested a possible repair strategy for SCI. This model was proven to be reproducible in independent labs [59-61]. Lee et al. [59] demonstrated the ability of this repair strategy (PNGs with aFGF treatment) to facilitate the regeneration of spinal ascending and descending tracts and also the recovery of motor behavior following SCI. They also concluded that this strategy facilitates the regrowth of the spinal axons and improves hindlimb function in the T-8 spinal cord-transected rat model [61]. Using a similar model and treatment, Tsai et al. [60] showed that regeneration of the corticospinal tract into the lumbar gray matter is a mechanism of functional locomotor recovery after complete cord transection and repair. To test the efficacy of this strategy, in vivo and in vitro studies have been performed and have established firmer basic evidence [62-73]. These studies verify the high potential of this strategy to repair the SCI and inspired the investigators to translate the treatment to subclinical and clinical trial.
Cheng et al. [74] applied this therapy to a chronic paraplegic adult patient with a thoracic spinal cord transection due to a stab injury 4 years previously. With this repair, the patient obtained significant motor recovery, and his functional status improved from being wheelchair-bound to being able to ambulate independently with a walker two-and-a-half years after the treatment. Wu et al. [75] presented a series of 18 patients with preganglionic brachial plexus injuries. Significant clinical improvement in muscle strength was noted at 12 and 24 months after being treated with the repair strategy.
ACIDIC FGF ALONE IN CLINICAL SCI
The adoption of the complete spinal cord transection in rodent models in the early research by Cheng et al. [58] was in order to avoid ambiguity and to model the most severe scenarios. Following investigators consequently continued to use transection or hemisection as a SCI model. However, in the majority of clinical circumstances, in accordance with the epidemiology and etiology, the damaged spinal cords usually retain their continuity in their gross appearance. Given that no macroscopically structural defect exists, there is no gap to be bridged by the PNGs. As a result, the modification of the repair strategy in the treatment of the clinical patients of SCI became imperative. The amended therapy for clinical patients omits the PNG and merely utilizes aFGF in fibrin glue. Wu et al. [24] reported a preliminary phase I clinical study of 9 patients with cervical SCI. The patients were treated with aFGF and fibrin glue during the neurolysis surgery. Six months after the treatment, modest nerve regeneration occurred in all 9 patients, and no adverse effect was observed. Subsequently, in order to further verify the safety and feasibility of this modified repair strategy for nonacute SCI, a phase II clinical trial was conducted [25]. It was an open-label, prospective, uncontrolled human clinical trial recruiting 60 patients with SCI (30 cervical and 30 thoracolumbar). The published data demonstrated significant improvements in American Spinal Injury Association (ASIA) motor and sensory scale scores, ASIA impairment scales, neurological levels, and functional independence measure at 24 months after treatment. There also were no related adverse events in this trial. Despite the completion of the trial in 24 months, the majority of the participants were constantly traced in the clinics. In the clinic visits, no further intervention was administered. The data from the followup at 48 months after the primary treatment were collected and analyzed to verify the longstanding efficacy and safety. Ko et al. [26] have recently published these data and concluded that the initial therapeutic effect was able to last for a long period, and there were no related adverse events or unexpected results reported throughout the 4-year follow-up. With respect to a novel therapy, safety is always the first consideration. These clinical trials probably eliminate the query of the oncogenicity from the mitogenic potential of aFGF. In addition, they also provide a feasible way of practical application in compliance with the real scenarios.
Considering translating the therapy of neural regeneration from bench to bedside, these clinical data are certainly of keen value. Yet, more questions arose as more attention focused on the strategy. One leading issue is the lack of a control group. The question is not easy to resolve because of ethical problems. To arrive at a compromise, the patients enrolled were all in a chronic phase, in which the neurological status was by all odds the most stable and the patients owned the least probability to improve spontaneously. Thus, patients with chronic phase SCI are good targets to determinate whether a therapy is effective or not [9].
PROPOSED MECHANISMS OF ACIDIC FGF FOR REGENERATION OF SCI
AFGF is thought to exert protective and regenerative effects on neurons following SCI, but the mechanism of these effects remains unclear. Few studies exploring the possible pathway of signal transduction have been reported. In a rodent model of contusive SCI, Tsai et al. [67] uncovered the performance of aFGF in the process of nerve repair after injury. By the proteomics approach, it is evident that aFGF down-regulates the expression of the proteins involved in the process of secondary injury, such as astrocyte activation, inflammation and scar formation, which lead to the blocking of injured spinal cord regeneration. They proposed that aFGF might initiate a series of biological processes to prevent or attenuate secondary injury.
In another study of a cerebral ischemic rat model the investigators applied aFGF mixed in fibrin glue (as a slow-release carrier) topically over the peri-ischemic cortex, and demonstrated neurite extension from cortical neurons which was significantly enhanced by aFGF, mediated through activation of Akt and Erk, and improved functional restoration in ischemic stroke rats. The results suggest that aFGF mixed in fibrin glue could prolong the protective/regenerative efficacy of aFGF to the damaged brain tissue and thus improve the functional restorative effect of aFGF [76]. An in vitro and in vivo investigation in rodent models of Parkinson disease revealed that the administration of aFGF activated downstream signals PI3K/Akt and ERK1/2. The authors suggested that aFGF attenuates neurotoxicity by down regulating the level of apoptosis via activation of the PI3K/Akt and ERK1/2 signal pathways [77]. A study of the administration of aFGF and bFGF to protect blood-brain barrier (BBB) integrity after intracerebral hemorrhage (ICH) in mice was carried out. The investigators suggested that FGF treatment reduced Ras homolog gene family member A (RhoA) activity via FGF receptor-induced activation of the PI3K-Akt-Rac1 signaling pathway, thus preserving BBB integrity, and therefore attenuating secondary brain injury after experimental ICH in mice [78]. Similarly, it was reported that aFGF administration preserved BBB integrity by activating the PI3K-Akt-Rac1 pathway and inhibiting RhoA following traumatic brain injury [79]. There is also an experiment demonstrating that inhibition of phosphatidylinositol 3-kinase (PI3K) blocks translocation of aFGF to the cytosol and nucleus [80]. These data suggest that PI3K is essential to initiate the signal transduction of aFGF.
Although the documentation of the signal pathway of aFGF specific to the repair of SCI is short, one can still find some interesting correlation between aFGF and other CNS lesions. In the limited evidence, the activation of the PI3K-Akt pathway seems to be a common feature. The finding is not surprising given the fact that PI3K/Akt signaling integrates extracellular signaling information to promote cellular proliferation in adult neural progenitors [81].
CONCLUSIONS AND FUTURE PERSPECTIVES
There has been a long list of review articles summarizing the repair strategies of cellular transplantation and molecular therapy after SCI. The current review aimed not to just extend the length of the list. Instead, the authors focused more on the aFGF which has been long applied in nerve repair-associated studies, but seldom sorted as a neurotrophic factor. A series of investigations by the authors’ team, in accordance with others, have demonstrated the promising results of aFGF in the treatment of nerve injury. Together with the utilization of autologous PNGs, aFGF enhances the axonal regrowth and promotes clinical motor function improvement in animal studies. By single use of aFGF without PNG bridging in clinical scenarios, patients obtained functional improvement and became less dependent on helpers in phases I and II in clinical trials. Most important, the oncogenic adverse effect was not observed throughout the 4-year follow-up. The positive effects of aFGF on neuroprotection and regeneration of injured neurons of the spinal cord make it a promising candidate for inclusion in treatment strategies [39]. Longer period of follow-up after the use of aFGF is also necessary in order to find out any possible adverse effects.
It seems that the aFGF is beneficial in the neural regeneration after SCI. However, the authors do not claim that the single pharmacological agent could cure the neurological deficit and immediately make the patients walk again. Rather, we emphasize that rehabilitation programs remain the gold standard to enhance possible neural plasticity, promote occupational recovery, and treat complicated neuropathic pain in chronic cases. Pharmacologically, the glial scar formed in the chronically injured spinal cord is an obstacle and inhibits neural growth. The administration of lysing molecules, such as chondroitinase, helps to lyse the fibrosis/gliosis in the injury zone and allows the axonal growth cone progress distally. Besides, biomaterial scaffolds serve as a drug release system and also promote axonal growth within the scaffolds. Combining PNGs with scaffolds has gained more acceptance. Commonly used scaffolds, including natural polymers (in vivo extracellular matrix polymers, polymers derived from blood, and polymers from marine life) and synthetic polymers (poly-hydroxy acid polymers and synthetic hydrogels), are crucial in the process of neural growth [82].
Individual therapies are unlikely to emerge as a cure for SCI. The authors, in agreement with other groups of investigators, predict that tailored combinations of strategies will lead to cumulative improvements in outcome after SCI [13,39]. An isolated therapy can be the focus of research; however, the merged strategies should be the future effort.
Notes
The authors have nothing to disclose.