INTRODUCTION
Gout is a common metabolic disease in which deposition of monosodium urate (MSU) crystals causes inflammatory arthritis of the peripheral joints. Nodules formed by the accumulation of MSU crystals in the soft tissue are called tophi. Gout affects 1-2% of adults in developed countries, where it is the most common type of inflammatory arthritis in men13). Typically, gout affects the distal joints of the appendicular skeleton. The occurrence of gout in the spine has been reported in only a limited number of cases1,2,5,6,8-12,14,16,18).
In this report, we describe a patient who complained of paraparesis. The cause was a cord-compressing mass-like lesion at the thoracic level. The presumed diagnosis according to the imaging was a neoplastic lesion, but histopathological examination confirmed the diagnosis as a gouty tophus.
CASE REPORT
A 64-year-old man was admitted to our clinic with weeklong weakness of both lower extremities. The patient also complained of back pain radiating to his left anterior chest for 4 weeks. Eight years prior, the patient had acute gout arthritis of the right knee. At that time, the symptoms spontaneously resolved, and the patient received no subsequent management for the condition. The back and chest pain had increased gradually and had become more severe over the previous month in spite of conservative treatment. A week before admission, paraparesis progressed to difficulty walking. The patient had no sphincter function impairment, fever, or weight loss.
Neurological examination showed paraparesis with grade IV motor weakness of both the hip and knee with no sensory impairment. Reflexes were normoactive and within normal ranges as assessed using the straight leg raise test. The patient's white blood cell count was slightly increased to 10,960/mm3 (normal range: 4,000-10,000/mm3); erythrocyte sedimentation rate was 5mm/h (normal range: lower than 9mm/h); C-reactive protein level was 0.9mg/dL (normal range: lower than 0.5mg/dL); and serum uric acid level was 12.2mg/dL (normal range: lower than 7.0mg/dL). No symptoms of gouty arthritis such as pain or swelling were reported for other joints of the appendicular skeleton.
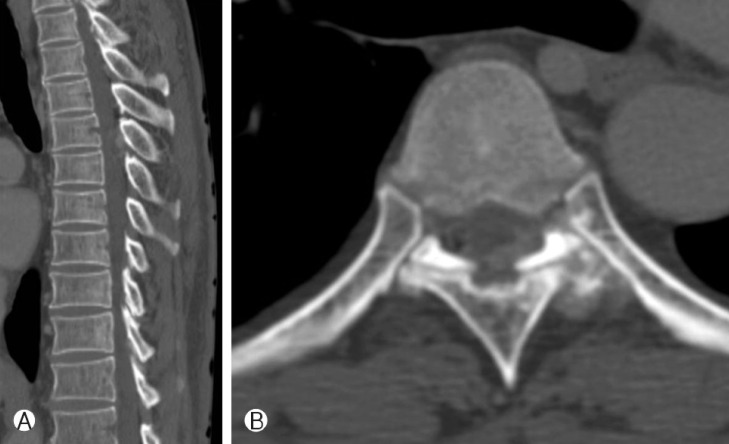
Computed tomography (CT) demonstrated a low-density mass with calcification surrounding the T6/7 left facet articulation and T6 and T7 left lamina. Bony erosion was also observed at the T6/7 left facet articulation (Fig. 1). Magnetic resonance imaging (MRI) of the thoracic spine revealed an extradural mass compressing the posterior of the spinal cord at T5-7, with heterogeneous high and low signal on T2-weighted images and low signal on T1-weighted images. The small amount of the low signal intensity of the mass on T2-weighted MRI corresponded to the calcified lesion on CT, and this lesion showed peripheral enhancement on post-contrast MRI (Fig. 2).
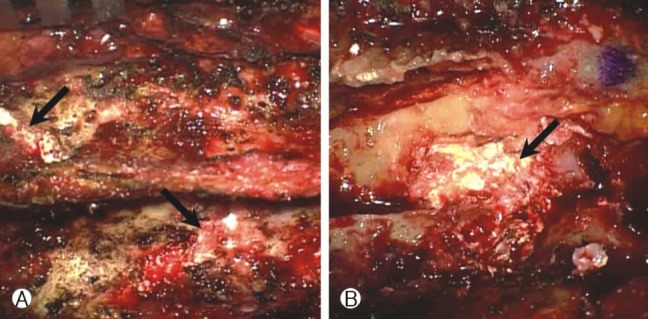
We made a tentative diagnosis based on the imaging findings of an epidural cyst or benign bone-originating tumor. Thoracic laminectomy from T5 to T7 was performed for the purpose of neural decompression and for histopathological confirmation. Intraoperatively, a chalky white material was found at the interlaminar space. This material extended to the dorsal space of the thoracic dura and the T5/6, T6/7 left foramen (Fig. 3). After left T5/6, T6/7 facetectomy, the mass that was compressing the cord and root was removed and T5, 6, and 7 both pedicle screw fixation with posterolateral fusion was performed.
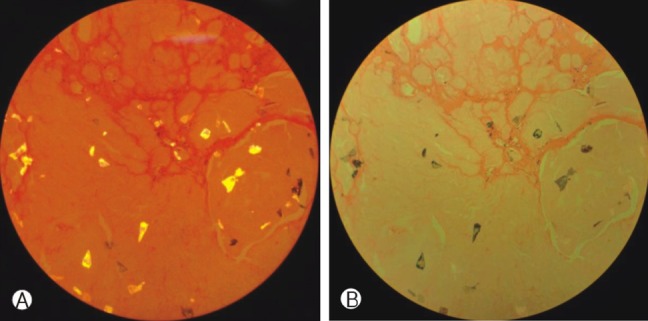
Histopathological examination of the specimen by polarized light microscope demonstrated a granular material with a needle pattern and negative birefringence (Fig. 4). This finding confirmed a diagnosis of a gouty tophus. No neoplastic tissue was found in the specimen. Postoperatively, the patient regained strength in both lower extremities, and his back and chest pain attenuated. After being discharged from the hospital, the patient has been treated with medication.
DISCUSSION
Gout, a purine metabolism disorder, is a common cause of inflammatory arthritis. Gout is caused by hyperuricemia leading to production of MSU crystals. Gout typically affects the peripheral joints of the appendicular skeleton but is also known to involve the axial joints. Although involvement of the spine is an unusual manifestation of tophaceous gout, spinal gout involvement is probably under-diagnosed because asymptomatic patients with this condition are unlikely to undergo neuroimaging of the spine8). A recent study found evidence of axial involvement, with or without symptoms, in 9 of 64 patients with gouty arthritis who had undergone a spinal CT examination10). However, only a few cases of histologically proven gout causing spinal cord compression have been reported in the literature.
Spinal involvement of gouty tophus has been observed in all spinal sections. The most frequent involvement reported in the literature is the lumbar spine10,14). However, the cervical and thoracic spine can also be affected1,4,5,15,17). Tophaceous crystal deposition in axial elements of the spine is generally found at the facet articulation, but the ligamentum flavum, pedicle, lamina, epidural soft tissue, vertebral body, and disc space can also be affected11,12,16). Chang et al. suggested that tophaceous accumulation begins at the facet joint and spreads to the ligamentum flavum and other elements9).
Patients with symptomatic gouty spine can present with spinal pain and fever associated with local and systemic inflammatory response. Because of the inflammation and the radiological findings, spinal gout is often mischaracterized as spondylitis or epidural abscess, as described in the literature16,18). Furthermore, the formation of a tophus can cause varied neurological manifestations that depend on the nature of the neural compression such as radiculopathy, claudication, and myelopathy4,5,8-10,12,15-17).
The radiological features of spinal gout are nonspecific and varied. Standard radiographs can be normal or might reveal degenerative changes such as disc-space narrowing and bony erosion. MSU crystals are not radiographically opaque so unless the urate deposits calcify, standard radiographs are not sensitive enough to detect them and therefore have limited use in diagnosing spinal gout6,10). CT and MRI are more specific imaging methods, but lack the accuracy required for differential diagnosis of spinal gout. CT can show juxta-articular masses with low-density and intra-articular and vertebral bony erosion with sclerotic or normal bony density16,18). When these features are accompanied by calcification, crystals can be seen as regions of hyperdensity on CT images6). On MRI, a tophus can have various appearances, with intermediate-to-low signal intensity on T1-weighted images and variable signal intensity on T2-weighted images. Contrast-enhanced T1-weighted MRI has two enhancement patterns: homogeneous enhancement and heterogeneous peripheral enhancement2,7,18). In our case, CT revealed a low-density tophus, including calcification surrounding facet articulation and lamina with bony erosion; MRI showed a mass with homogeneous low signal intensity on T1-weighted images and heterogeneous low and high signal intensity on T2-weighted images. The calcified portion of the tophus on the CT images corresponded to the location of the lesion that had low signal intensity on the T2-weighted image and peripheral enhancement in a gadolinium-enhanced T1-weighted image. These findings are delineating characteristics for tophaceous spinal gout, but spinal gout was not initially suspected. In the literature, many cases of spinal gout are misdiagnosed as other diseases such as spondylitis, epidural abscess, neoplastic lesion, or degenerative stenosis2,4,8,10,12,16-18). Spinal gout has diverse clinical presentations, and radiological findings are not sufficiently specific to confirm a diagnosis; therefore, clinicians should consider spinal gout as a potential diagnosis for patients with a history of gout who present with spinal pain and neural compressive symptoms.
A definitive diagnosis of spinal gout requires histopathological examination. MSU crystals are easily detected under a light microscope, but the use of a polarized light microscope is required for definitive confirmation. Under polarized light, the crystals exhibit strong negative birefringence: they are yellow when aligned parallel to the axis and blue when aligned across the direction of polarization. A finding of negatively birefringent urate crystals firmly establishes the diagnosis of gouty arthritis13,14).
Standard management of gout is stabilization of acute attacks by colchicines, non-steroidal anti-inflammatory drugs, or both, with urate-lowering therapy using allopurinol13). In spinal gout causing neural-compressive manifestations such as myelopathy or radiculopathy, however, surgical decompression in conjunction with pharmacological treatment can provide a more satisfactory outcome3,14).
CONCLUSION
Spinal gout is not easily detected because its symptoms are varied and its radiological characteristics are not specific enough to establish a definitive diagnosis. Therefore, when patients with prior episodes of gouty arthritis have neural compressive symptoms of the spine, spinal gout should be considered. For patients with neurological symptoms, neurosurgical decompression with pharmacological treatment should be performed and can provide positive outcomes.