 |
 |
- Search
|
|
||
Abstract
Objective
To analyze long-term clinical outcomes of endoscopic transforaminal foraminoplasty for foraminal stenosis.
Methods
Long-term 5-year MacNab outcomes, visual analogue scale (VAS) scores, complications, and unintended aftercare were analyzed in a series of 86 patients who underwent endoscopic transforaminal foraminoplasty for foraminal stenosis.
Results
At minimum 5-year follow-up, excellent results according to the MacNab criteria were obtained in 32 patients (37.2%), Good in 40 (46.5%), fair in 11 (12.8%), and poor in 3 (3.5%), respectively. The mean preoperative VAS was 6.15. The mean postoperative and last follow-up VAS was 3.44. Both postoperative VAS and last follow-up VAS were statistically reduced at a significance level of p<0.0001. Postoperative dysesthesia occurred in 9 patients. Another 9 patients had recurrent disc herniations (10.5%). Failure to cure with persistent pain occurred in 3 patients. Two patients developed pain postoperatively stemming from a different level. One patient experienced a postoperative hematoma which ultimately was inconsequential and did not require any additional surgery. Only 3 patients opted for revision endoscopic discectomy and another 2 for revision fusion surgery.
Spinal decompression fusion surgery is often recommended as the preferred option for patients who have lumbar spinal stenosis, with or without degenerative spondylolisthesis. However, spinal fusion remains controversial, and its clinical superiority over less aggressive outpatient endoscopic decompression surgeries has not been substantiated in controlled trials comparing the least disruptive staged employment of endoscopic procedures to the myriad of traditional fusion surgeries.
Contemporary studies on the degeneration of the lumbar spine have identified that the age-related degeneration of the intervertebral disc eventually results in instability of the spinal segment. The ensuing hypermobility is associated with thickening of soft tissues in the foramen, followed by hypertrophy of the facet joints, particularly at the superior articular process (SAP) [1-3]. Most patients who are symptomatic voice complaints that begin with benign early symptoms of subjective numbness, intermittent claudication and weakness from increased activity and exertion. Symptoms gradually progress insidiously to become painful irritations that are increasingly bothersome, and then debilitating.
When symptoms progress, patients may then seek medical consultation. The objective physical findings are often absent and subjective weakness, and intermittent claudication may limit walking endurance and other physical activities. When surgery is considered many patients shy away from the aggressive treatment options of open spine surgery whose preoperative planning is often based in the traditional image-based decision-making [4,5]. Lumbar endoscopic decompression techniques offer an alternative. The first author of this manuscript has experienced firsthand that patients' satisfaction with their clinical outcomes is highest when the final determination of the plan of care is a shared decision between patient and provider based on a complex analysis of each patient's painful patho-anatomy [6,7]. In this context, foraminal stenosis is a frequent source of pain. The focus of this study was to analyze the long-term survival of the treatment effect following endoscopic foraminal decompression using Yeung’s widely publicized techniques [8].
A review of Yeung’s extensive database identified 86 consecutive clinical cases of endoscopic foraminoplasty decompression using A. Yeung’s technique and selective endoscopic discectomy (SED) technique of endoscopic transforaminal decompression for degenerative foraminal stenosis was used [8]. Patients had surgery before 2015 and were included in this study if a minimum of 5-year follow-up data was available. Of the 86 patients, 22 (25.6%) were women, and 64 (74.4%) were men (Table 1).
The inclusion criteria were: (1) clinical signs of lumbar radiculopathy, dysesthesias, and decreased motor function; (2) imaging evidence of foraminal or lateral recess stenosis demonstrated on preoperative magnetic resonance imaging (MRI) and computed tomography (CT) scans described below; (3) unsuccessful nonoperative treatment, including physical therapy and transforaminal epidural steroid injections for at least 12 weeks.
Patients considered not suitable for the endoscopic transforaminal decompression procedure were stratified according to the following exclusion criteria: (1) severe central stenosis (less than 100 mm2) [9]; (2) metastatic disease, (3) infection; and (4) instability defined as anterolateral translation of more than 3–5 mm or rotation of more than 10° to 15° in the dynamic views [8].
These preoperative image-based patient selection criteria were consistently used throughout the study since the knowledge gained during intraoperative evaluation of the patients’ specific predominant pain generators, such as the tip of the SAP, hypertrophy of the transverse process and pars interarticularis, of soft tissues impingement and tethering of the nerve roots—to name a few examples—most likely would have introduced selection and hindsight bias particularly after the initial learning curve. All patients in this consecutive case series provided informed consent and IRB approval was obtained (CEIFUS 106-19).
Patients preoperative imaging studies including plain film studies including extension/flexion views, MRI, and CT were evaluated for the presence of foraminal and lateral recess stenosis. Well established radiographic classification systems explained elsewhere were used to classify the severity and location of the stenotic process in the neuroforamen [10-13]. Patients with severe foraminal stenosis according to Lee et al. [10] were included in this study. Lee’s classification defines foraminal zone and lateral recess stenosis within the neuroforamen by dividing it from medial to lateral into entry-, middle-, and exit zone [10]. Foraminal and lateral recess stenosis were stratified according to the main offending pathology. In the entry zone, Lee et al. [10] described hypertrophy of the superior articular process as the predominant pathology. In the mid zone, it was often due to an osteophytic process underneath the pars interarticularis and in the exit zone due to a subluxed and hypertrophic facet joint. The heights of the posterior intervertebral disc and lumbar foramina were evaluated according to Hasegawa et al. [11], who described a lumbar neuroforaminal height of 15 mm or more as normal and reduced posterior intervertebral disc height of 3 to 4 mm as suggestive of spinal stenosis. Preoperative sagittal and axial MRI images were graded by the author and used to assess the location and extent of foraminal stenosis. Only patients with stenotic lesions (whether due to bony stenosis, or contained disc bulge) producing a neuroforaminal width of 3 mm or less on the sagittal MRI cuts or lateral recess height of 3 mm or less on the axial MRI cuts were considered “stenotic” and treated surgically. These radiographic descriptors of foraminal stenosis have previously been employed by the senior author and were used in this study to stratify patients for the further diagnostic workup of the level(s) believed to be causing the patient’s symptoms (see below) [14].
The sex and frequency distribution of the stenotic levels identified on preoperative imaging studies is summarized in Table 1. The frequency distribution of the actual surgical procedures performed is listed in Table 2. As expected, foraminoplasty was most frequently performed at the L4/5 (20 of 86 patients) and the L5/S1 (15 of 86 patients) levels. This data set of the 86 identified lumbar foraminoplasty patients with a minimum of 5-year follow-up was used for the outcome analysis presented herein.
History and physical examination (H&P) are the most significant factors in establishing the diagnosis of claudication. Since the physical exam while resting may be “normal,” and abnormalities in motor and sensory function may be absent, patients were asked to walk to the pain limit. Electrodiagnostic studies were used as an adjunct H&P to help elucidate the clinical severity of symptoms with an objective test such as electromyography (EMG) and nerve conduction studies. However, their usefulness has been questioned because of their low specificity and sensitivity. Peripheral neuropathy and other comorbidities were also considered, as their existence may affect clinical results and increase the risk of complications [4]. The EMG may be “normal” or “abnormal,” but abnormal interpretation will help validate the patient’s subjective complaints [12].
Although CT and MRI imaging measurements are useful guidelines in the evaluation of lumbar stenosis conditions, the actual symptoms do not always correspond to traditional imaging studies [15]. Therefore, the patient’s response to diagnostic and therapeutic injections was used to correlate imaging findings with symptoms and provide a prognosis for endoscopic transforaminal decompression. Transforaminal diagnostic and therapeutic injections containing steroids and/or a local anesthetic were shown to be more reliable predictors of successful clinical outcomes with the transforaminal decompression procedure. This has been conclusively demonstrated in a recent study on the positive predictive value of preoperative selective nerve root blocks published by the last author of this manuscript [16]. Given the high positive predictive value of preoperative diagnostic transforaminal epidural steroid injection (TESI) of 98.4%, successful outcomes were expected with the transforaminal decompression if patients had a diagnostic response to the injection by reporting more than 50% reduction in pain on the visual analog scale for back and leg pain [17-19]. Additional auxiliary information was derived from an epidurogram, whose patterns may provide more additional imaging confirmation of foraminal compression, disc protrusion, and anatomic abnormalities.
Patients were treated employing the endoscopic transforaminal approach using the “inside-out” technique, in which the working sheath is placed into the intervertebral disc, thus, retracting and avoiding the exiting nerve root. The surgical technique used by the authors employs a foraminoplasty in patients with foraminal and lateral stenosis for the treatment of herniated disc (Fig. 1) [4]. Procedures were performed in prone position under local anesthesia and sedation in all patients. For the foraminoplasty, bone from the hypertrophied superior and inferior articular process was removed with endoscopic drills, Kerrison rongeurs, and percutaneous trephines (Fig. 2) (Supplementary video clip 1). The endoscopic drills and rongeurs were deployed inside the center working cannula of the endoscope to lessen the risk of dysesthesia and irritation of the exiting nerve root and its dorsal root ganglion (DRG). The decompression was typically performed under continuous direct videoendoscopic visualization. The foraminoplasty was facilitated by changing the trajectory of the instruments to aim for the compressive pathology identified on preoperative studies, i.e., the bony decompression was focused on removing the relevant stenotic process (Fig. 3). Contained herniations were decompressed from the inside out, and extruded disc herniations were through a small annular window. A laser was used to remove the tip of the SAP.
Plain radiographs were reviewed for the presence of any fractures due to trauma, and osteoporosis. They were also evaluated for the loss of disc height, spinal alignment, pars defects, facet hypertrophy and osteophytosis which may be indicators of subclinical instability, and, hence, be a cause for excluding the patient from the study. Advanced imaging modalities including MRI and CT were evaluated for lateral recess stenosis. CT myelography was assessed whenever available as the best measure of central and lateral canal due to any extradural cause of neural compression. It was ordered for patients with suspected dynamic stenosis, postoperative leg pain, severe scoliosis or spondylolisthesis, metallic implants, other contraindications to MRI, and, most importantly, in patients with lower extremity symptoms in the absence of plausible MRI findings [8,15]. Midline T2-sagittal and axial images may be useful in diagnosing central and lateral recess stenosis by noting narrowing neuroforaminal height of less than 15 mm and width of less than 3 mm [11]. The reduced posterior height of the intervertebral disc of less than 3 mm has also been considered an indicator of symptomatic spinal stenosis in more than 80% of patients [11]. Sagittal T1-weighted images were evaluated with particular attention focused on the foramen. The absence of normal fat around the root may indicate foraminal stenosis as evidenced by a recent study. Extraforaminal stenosis was preferably identified on axial T1-weighted images by obliteration of the regular interval of fat between the disc and nerve root. In this study, all available advanced imaging studies were carefully analyzed in correlation with the patient’s clinical symptoms since both CT and MRI are poor predictors of severity and resolution of symptoms. Both studies have been found to give an inaccurate assessment of the degree of stenosis and underestimate soft tissue impingement.
At 5 years postoperatively, primary clinical outcomes were assessed using the MacNab criteria [20]. The good/excellent results were then stratified by lessons learned with the intent of refining indications and patient selection criteria for endoscopic decompression under local anesthesia in this consecutive case series of 86 patients. In the case of fair and poor MacNab outcomes, patients’ postoperative imaging studies were scrutinized for instability, or recurrent stenosis at the index level. Patients were also asked to select a score on the visual analogue scale (VAS) preoperatively (preop VAS), within the immediate postoperative period (postop VAS), and at final follow-up (last F/U VAS) [21]. For the detailed outcome analysis, paired and 2-tailed t-test, crosstabulation statistics as measures of association between variables were computed for 2-way tables using IBM SPSS Statistics ver. 25.0 (IBM Co., Armonk, NY, USA). Descriptive statistic measures were used to calculate the mean, range, and standard deviation as well as percentages. Crosstabulation methods were used to assess for any statistically significant association between intraoperatively visualized pathology and clinical outcome data based on the modified Macnab. Pearson chi-square and Fisher exact test were employed as statistical measures of association. Expected cell counts, continuity corrections, and likelihood ratios were calculated for some analyses.
The age distribution of our patient population was normal (Fig. 4). The average age was 61.9 years (standard deviation [SD], 12.38 years) with the youngest patient being 26 years of age and the oldest patient 84 years, respectively. The average time to perform Yeung’s lumbar endoscopic transforaminal foraminoplasty surgery was 55.71 minutes (range, 23–105 minutes; SD, 18.76 minutes; Table 3). At minimum 5-year follow-up, excellent results according to the MacNab criteria were obtained in 32 patients (37.2%), good in 40 (46.5%), fair in 11 (12.8%), and poor in 3 (3.5%), respectively (Table 4). The mean preoperative VAS was 6.15. The mean postoperative and last F/U VAS was 3.44. Both postoperative VAS and last F/U VAS were statistically reduced at a significance level of p<0.0001. The minimum, maximum, and standard deviation for these numbers are listed in Table 3.
There were no major approach-, surgical- or anesthesia-related complications in this series. The vast majority of patients (62 of 86; 72.1% of study population) did not have any postoperative sequelae (unavoidable side effect of an otherwise expertly executed surgery), or complication (Table 5). Postoperative dysesthesia due to irritation of the DRG occurred in 9 patients (10.5%) and was the most common benign sequelae. It was managed with activity modification, gabapentin or pregabalin, and transforaminal epidural steroid injections. Most patient’s DRG irritation resolved with these supportive care measures within 2 to 3 weeks. Ten patients had postoperative complications; nine of which suffered from a recurrent herniation of the nucleus pulposus (HNP) at the index level (recurrence rate of 10.5%). Three patients rated their outcome as fair and poor with persistent pain due to failure to cure. Two patients developed pain postoperatively stemming from a different level. One patient experienced a postoperative hematoma which ultimately was inconsequential and did not require any additional surgery (Table 5). Thirty of the 86 patients had additional treatments postoperatively. Most of them (23 patients) were treated with TESI postoperatively to diminish their symptoms. Three of the 9 patients with recurrent HNP opted for another SED. One patient underwent L3/4 fusion and another patient an L3–5 fusion at the index levels of their respective endoscopic foraminoplasty. An additional patient required a postoperative hemilaminectomy at the index level to control his symptoms, and yet another patient underwent a foraminotomy at another than the index level. Furcal nerves were observed in 19 patients. Furcal nerves were readily recognized by the senior surgeon and did not impact outcomes. Another patient had multilevel postlaminectomy syndrome but rated the clinical outcome as good (Table 5).
Cross-tabulation of MacNab outcome data versus postoperative sequelae and complications show that 62 patients (72.1% of the study population) did not have any additional intervention postoperatively. Fifty-five of those 62 patients had excellent and good outcomes according to Macnab. Another 7 patients with fair and poor Macnab outcomes did not receive any additional treatments defining the remainder of the study population (24 patients, 27.9%) as the subgroup of patients that received some unintended postoperative aftercare. Four patients attributed their fair and poor postoperative outcome rating to the severity of the DRG irritation. None of the patients with poor outcomes opted for more surgery. Three patients with fair MacNab outcomes had additional surgery: 1 patient for recurrent HNP, 1 patient for incomplete decompression and persistent stenosis related back- and leg pain, and 1 additional patient with another symptomatic level that was not operated on during the index level decompression. The crosstabulation between unintended postoperative care measures and MacNab outcomes did not reveal any predictors of poor postoperative outcomes at a statistically significant level (Table 6).
This 5-year follow-up study on patients who underwent endoscopic transforaminal decompression for bony foraminal spinal stenosis showed favorable long-term outcomes with the procedure in the majority of patients. Sixty-four percent of patients were rated to have excellent and good outcomes using MacNab standardized outcome criteria. The VAS reductions were also statistically significant (p<0.0001). Over two-thirds of the study population (72.1%) did not require any additional intervention following their endoscopic transforaminal foraminoplasty. Approximately one-third (27.9%) had some unintended aftercare postoperatively which was not associated with fair or poor outcomes (Table 5). The majority of unintended aftercare was delivered to patients with excellent and good outcomes (17 patients). Therefore, the authors of this study conclude that longterm outcomes with endoscopic transforaminal foraminoplasty are similar to outcomes reported with microdiscectomy or laminectomy [9].
In the opinion of this team of authors, the endoscopic visualization of the patient’s lumbar foraminal anatomy, whether a variation of normal or abnormal, needs to be evaluated intraoperatively as to whether it causes pain and then correlated with preoperative imaging studies to determine the best course of action during the decompression procedure [5-7,22]. Other pain generators may exist besides the mechanical compression of the exiting or traversing nerve root created by a herniated disc or bony obstruction of the neuroforamen. As evidenced by the few patients in this study who opted for additional surgery, patients may have multiple pain generators within one motion segment that can contribute to inferior clinical outcomes postoperatively if left untreated during the index procedure. Therefore, surgical intervention should not just be based on interpretation of plain film and advanced imaging studies. Disregard for the utility of the endoscopic visualization of the symptomatic patho-anatomy seems ill-advised and may be a source of less favorable outcomes for the novice endoscopic spine surgeon unfamiliar with endoscopic spine surgery and its techniques.
In patients who fail to respond to nonoperative management, translaminar surgical treatments such as decompression or decompression with spinal fusion are considered as the surgical standards. Traditionally trained surgeons may be hesitant to advise surgery because the patient is deemed “too young,” or “too old,” or have “too many” comorbidities [22]. Even obesity as a common comorbidity may provide an additional risk to open surgery. In some patients, traditional decompression may require extensive decompression to effectively relieve the patient of lateral recess or foraminal stenosis iatrogenically creating the need for fusion, which in turn may be considered too aggressive. Hence, many patients go on without treatment with persistent and often increasing disability. Therefore, percutaneous endoscopic foraminal decompression is an attractive alternative for these types of patients as it can effectively treat foraminal stenosis without creating the need for fusion. The latter statement is corroborated by the results of this study, where only 2 of the 86 (2.32% fusion rate) patients underwent revision fusion as part of their definitive treatment. The 2 patients underwent fusion to prevent postlaminectomy instability after their midline decompression. There was no patient in this study requiring fusion due to progressive instability.
The recurrence rate of herniated disc observed in this study at first glance perhaps seems somewhat higher at 10.5% than what is reported in the literature [23-25]. However, when adjusted for the advanced age of the patients of this study group it is more realistic to expect a 10% recurrence rate [26] than the published recurrence rate of around 5% [19,20]. Compared to established complication rates [25-30] with open lumbar spine surgery or other forms of translaminar or transforaminal minimally invasive spinal surgery our postoperative complications where almost exclusively comprised of recurrent HNP. One patient had an inconsequential postoperative hematoma. There were no dural tears, infections, wrong level surgeries, or patients with motor dysfunction or foot drop, pulmonary emboli or any other complications due to poorly managed medical comorbidities. Nearly all of the unintended aftercare was due to benign postoperative DRG irritation or postoperative pain nearly all of which was treated successfully with TESI.
Perhaps the most significant limitation of this retrospective study may have been the impact of affective (unconscious emotional reaction) and cognitive (distortions of thinking) biases in the clinical diagnostic and surgical decision-making process [31,32]. Cognitive biases, such as hindsight or outcome bias, are virtually unavoidable in retrospective studies as knowledge of the clinical outcome by the surgeon has been recognized to inflate the predictability of an event after it happened [33-36]. Hindsight cognitive biases may have been less relevant since the individual patient-specific pain generators ascertained during awake intraoperative evaluation under local anesthesia were not known throughout the 8-year study period. Intuition bias [36] may have played a role in patient selection for surgery after the initial learning curve. Stratifying patients according to the individual patient-specific pain generators seems appropriate for the nextgeneration follow-up study which this team of authors intends to perform. This more advanced analysis of the role on patientspecific pain generators on long-term outcomes may focus on the impact of rostral migration of the cranial tip of the SAP into the axilla of the exiting nerve root, hypertrophy of the transverse process and pars interarticularis, soft tissues impingement caused by hypertrophy of the ligament flavum and facet cysts or by apophyseal ring osteophytes of the upper vertebral body within the axilla of the exiting root, and on the impact of nerve root tethering and chronic inflammation of the DRG caused by far lateral extraforaminal disc herniations. This current study simply lacked statistical power to cross-tabulate these factors with any statistical significance.
Patients with symptomatic foraminal stenosis may present with a combination of axial back pain, sciatica, numbness, and neurogenic claudication. Foraminal stenosis can be treated favorably with early transforaminal endoscopic decompression. Delaying surgical decompression often recommended with open spinal surgery seems unwarranted given the direct access to the stenotic neuroforamen with the endoscopic transforaminal approach and its low propensity to destabilize the lumbar spinal motion segment. New “disruptive” endoscopic concepts are currently under the radar of traditionally trained surgeons because it may be difficult to accept new evidence until the concepts and illustrations are validated with high-grade clinical studies.
SUPPLEMENTARY MATERIAL
Supplementary video clip 1 can be found via https://doi.org/10.14245/ns.1938038.019.v1.
Supplementary video clip 1: This video shows the key steps of the visualized endoscopic transforaminal treatment of lumbar stenosis using the YESSTM technique.
Fig. 1.
(A-C) Sagittal and axial T2-weighted magnetic resonance imaging images of an 85-year-old male specifically referred for open translaminar decompression because of the severe central, lateral recess, and foraminal stenosis. The patient insisted on endoscopic transforaminal decompression since a right-sided transforaminal epidural steroid injection gave him 70% pain relief. Yeung’s technique specifically uses diagnostic and therapeutic injections as a predictor of the surgical outcome of his trademarked technique. (D, E) Intraoperative chromodiscography and epidurography confirmed concordant pain, severe central and foraminal stenosis and rendered an outline of the exiting nerve root.
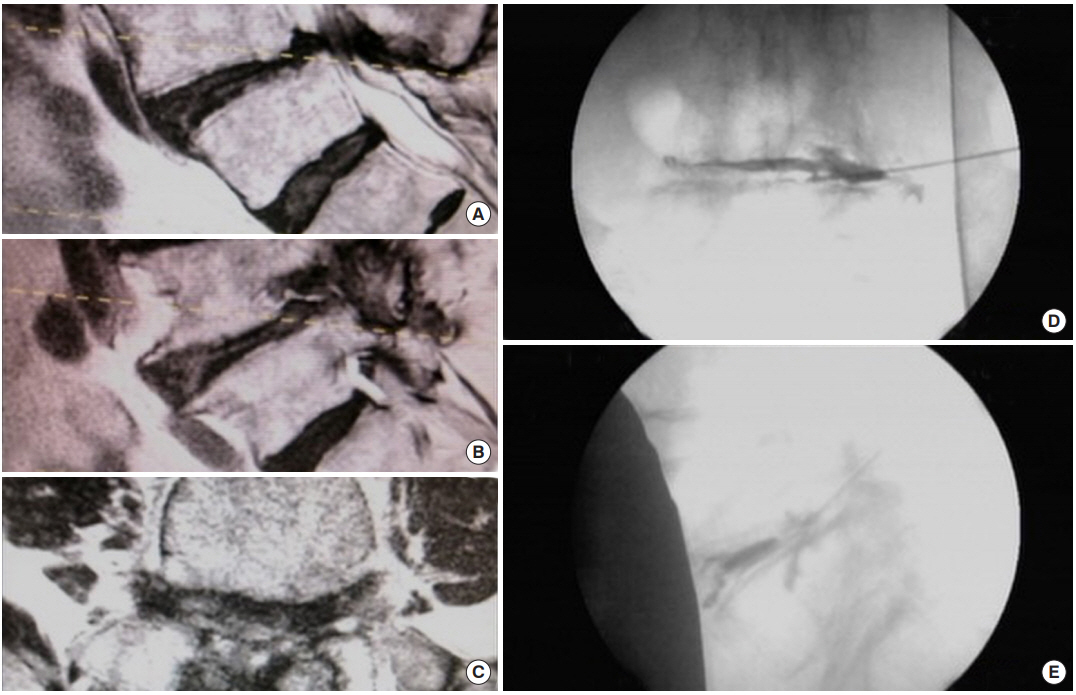
Fig. 2.
(A-D) Intraoperative endoscopic images of the same 85-year-old male patient illustrated in Fig. 1. After placement of the working cannula and removal of capsular tissue, the superior articular process (SAP) was exposed. (A) Direct visualization, SAP decompression was facilitated with the use of trephines. (B) An articulating burr was used to further the decompression from lateral to medial and to further resect the tip of the SAP. Alternatively, a laser can be used to remove the tip of the SAP (Supplementary video clip 1). Disc tissue and disc annulus was removed with pituitary rongeurs and cutting forceps (C), which allowed decompression of the traversing nerve root by removing both disc and annular tissue (D). The intradiscal position of the 5.0- or 6.0-mm trephines during the inside-out decompression in the posterior-anterior projection (E), and at the recess in the lateral projection (F) confirm adequate foraminal and central stenosis decompression.
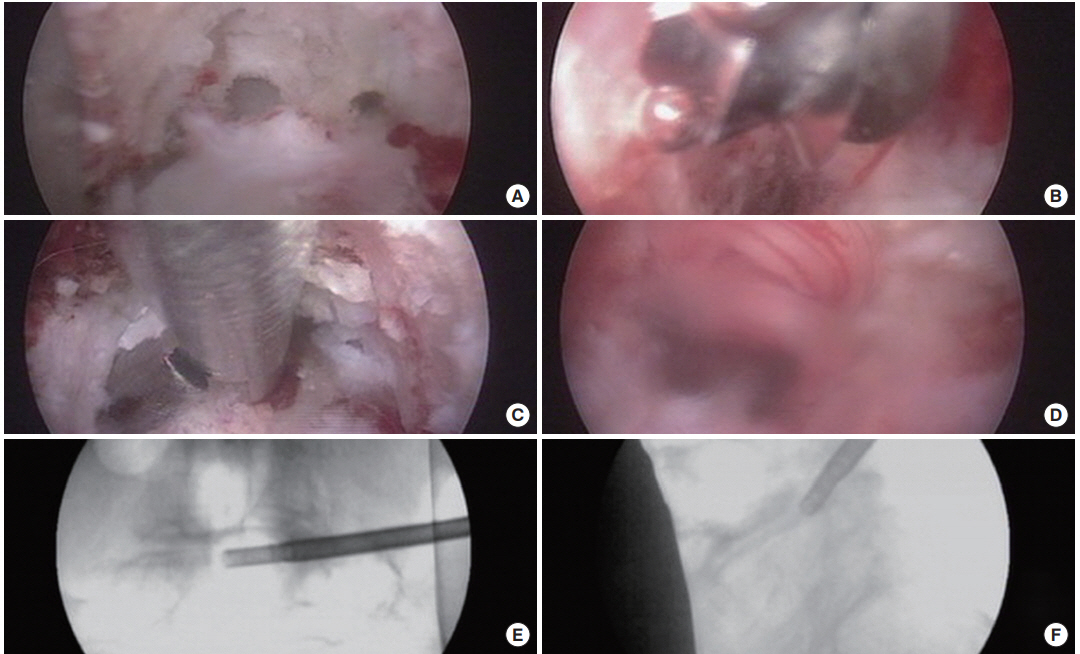
Fig. 3.
(A-D) Intraoperative endoscopic images of the same 85-year-old male patient illustrated in Fig. 1. The final position of the working cannula is shown (A, B) posteriorly and at the medial interpedicular line confirming adequate foraminal and central stenosis decompression. (C) The traversing nerve root (red arrow) is adequately decompressed. A large amount of bone cut by the trephines (D) and disc annulus tissue (E) was removed during the decompression.
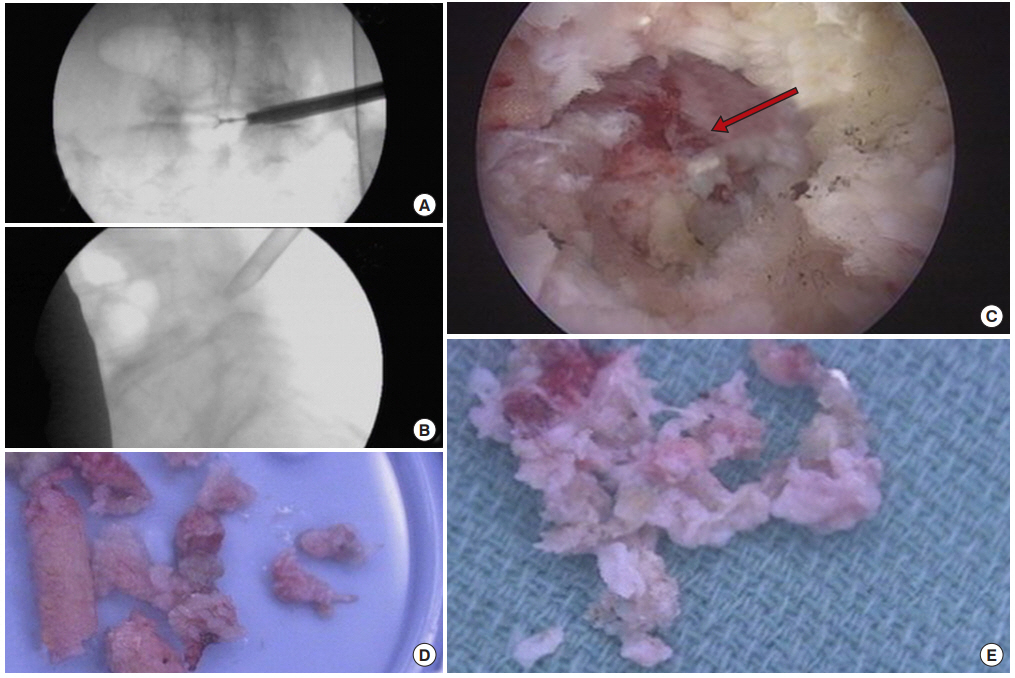
Fig. 4.
Age distribution of patients with 5-year follow-up after endoscopic transforaminal foraminoplasty for foraminal stenosis with the superimposed expected normal distribution (black line). Patient’s age ranged from 26 to 84 years of age and averaged 61.9 years. SD, standard deviation.
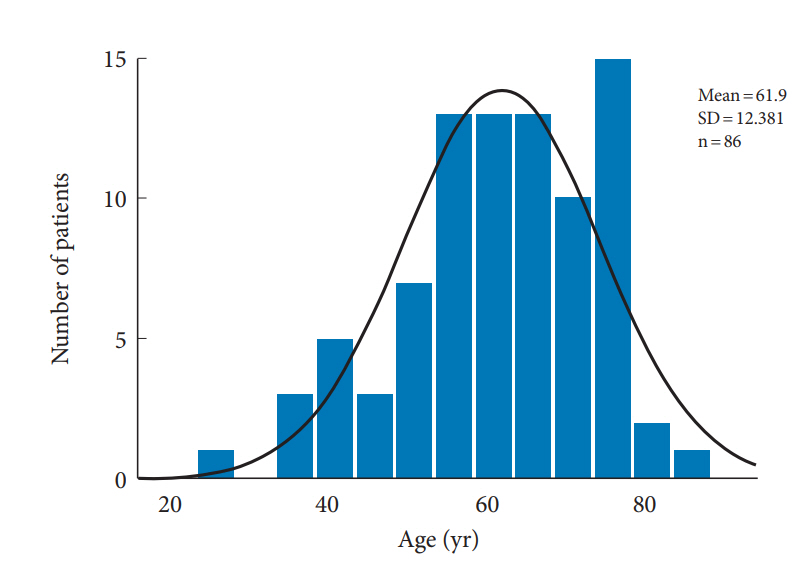
Table 1.
Sex and level distribution of foraminoplasty patients
Table 2.
Type of endoscopic transforaminal foraminoplasty by level
Table 3.
Descriptive statistics age, surgery time, preoperative VAS (pre-VAS), postoperative VAS (post-VAS), and last F/U VAS (n=86)
| Variable | Mean±SD (range) |
|---|---|
| Age (yr) | 61.90 ± 12.38 (26–84) |
| Surgery time (min) | 55.71 ± 18.76 (23–105) |
| Pre-VAS | 6.15 ± 2.07 (1–10) |
| Post-VAS | 3.44 ± 1.92 (0–8) |
| Last F/U VAS | 3.44 ± 2.29 (0–10) |
Table 4.
MacNab clinical outcomes in foraminoplasty patients
| Outcome | No. of patients (%) |
|---|---|
| Excellent | 32 (37.2) |
| Good | 40 (46.5) |
| Fair | 11 (12.8) |
| Poor | 3 (3.5) |
| Total | 86 (100) |
Table 5.
Sequelae & complications, additional postoperative treatment, and intraoperative findings
Table 6.
Postoperative sequalae and complications by MacNab outcomes
| Complication |
MacNab outcomes |
Total | |||
|---|---|---|---|---|---|
| Excellent | Good | Fair | Poor | ||
| Patients without complication | 27 | 28 | 5 | 2 | 62 |
| Dysesthesia | 0 | 5* | 3* | 1* | 9 |
| Hematoma | 0 | 1 | 0 | 0 | 1 |
| Other level pain | 0 | 1 | 1 | 0 | 2 |
| Persistent pain & stenosis | 1 | 1 | 1 | 0 | 3 |
| Recurrent HNP | 4 | 4 | 1 | 0 | 9 |
| Total | 32 | 40 | 11 | 3 | 86 |
REFERENCES
2. Rauschning W. Pathoanatomy of lumbar disc degeneration and stenosis. Acta Orthop Scand Suppl 1993 251:3-12.


3. Rauschning W. Normal and pathologic anatomy of the lumbar root canals. Spine (Phila Pa 1976) 1987 12:1008-19.


4. Yeung AT. Endoscopic decompression, foraminal-plasty and dorsal rhizotomy for foraminal stenosis and lumbar spondylosis: a hybrid procedure in lieu of fusion. J Neurol Disord 2016 4:322.

5. Yeung A, Yeung CA. Endoscopic identification and treating the pain generators in the lumbar spine that escape detection by traditional imaging studies. J Spine 2017 6:369.
6. Yeung AT, Gore S. In-vivo endoscopic visualization of pathoanatomy in symptomatic degenerative conditions of the lumbar spine II: intradiscal, foraminal, and central canal decompression. Surg Technol Int 2011 21:299-319.

7. Yeung AT. In-vivo endoscopic visualization of pain generators in the lumbar spine. J Spine 2017 6:385.

8. Yeung AT. The Yeung percutaneous endoscopic lumbar decompressive technique (YESSTM). J Spine 2018 7:408.

9. Sengupta DK, Herkowitz HN. Lumbar spinal stenosis. Treatment strategies and indications for surgery. Orthop Clin North Am 2003 34:281-95.


10. Lee CK, Rauschning W, Glenn W. Lateral lumbar spinal canal stenosis: classification, pathologic anatomy and surgical decompression. Spine (Phila Pa 1976) 1988 13:313-20.


11. Hasegawa T, An HS, Haughton VM, et al. Lumbar foraminal stenosis: critical heights of the intervertebral discs and foramina. A cryomicrotome study in cadavera. J Bone Joint Surg Am 1995 77:32-8.


12. Kirkaldy-Willis WH, Wedge JH, Yong-Hing K, et al. Lumbar spinal nerve lateral entrapment. Clin Orthop Relat Res 1982 (169):171-8.


14. Lewandrowski KU. “Outside-in” technique, clinical results, and indications with transforaminal lumbar endoscopic surgery: a retrospective study on 220 patients on applied radiographic classification of foraminal spinal stenosis. Int J Spine Surg 2014 Dec 1;8. https://doi.org/10.14444/1026. eCollection 2014.

15. Lewandrowski KU. Retrospective analysis of accuracy and positive predictive value of preoperative lumbar MRI grading after successful outcome following outpatient endoscopic decompression for lumbar foraminal and lateral recess stenosis. Clin Neurol Neurosurg 2019 179:74-80.


16. Lewandrowski KU. Successful outcome after outpatient transforaminal decompression for lumbar foraminal and lateral recess stenosis: The positive predictive value of diagnostic epidural steroid injection. Clin Neurol Neurosurg 2018 173:38-45.


17. Botwin KP, Gruber RD, Bouchlas CG, et al. Fluoroscopically guided lumbar transformational epidural steroid injections in degenerative lumbar stenosis: an outcome study. Am J Phys Med Rehabil 2002 81:898-905.


18. el-Khoury GY, Ehara S, Weinstein JN, et al. Epidural steroid injection: a procedure ideally performed with fluoroscopic control. Radiology 1988 168:554-7.


19. Bogduk N, Aprill C, Derby R. Epidural spinal injections. In: White AH, Schollerman J editors. Spinal care: diagnosis and treatment. St. Louis (MO): Mosby; 1995. p.322-43.
20. Macnab I. Negative disc exploration. An analysis of the causes of nerve-root involvement in sixty-eight patients. J Bone Joint Surg Am 1971 53:891-903.


21. Huskisson EC, Jones J, Scott PJ. Application of visual-analogue scales to the measurement of functional capacity. Rheumatol Rehabil 1976 15:185-7.



22. Yeung A. Failed back surgery syndrome: endoscopic documentation of common causes by visualization of painful patho-anatomy in the hidden zone of the axilla containing the dorsal root ganglion and salvage treatment of neuropathic pain with DRG neuromodulation [abstract]. In: Regular poster of ISASS17 conference; 2017 Apr 12-4; Boca Raton (FL), USA.

23. O'Lynnger TM, Zuckerman SL, Morone PJ, et al. Trends for spine surgery for the elderly: implications for access to healthcare in North America. Neurosurgery 2015 77 Suppl 4:S136-41.



24. Hernandez-Perez PA, Prinzo-Yamurri H. Analysis of the lumbar discectomy complications. Neurocirugia (Astur) 2005 16:419-26.


25. Lewandrowski KU. Incidence, management, and cost of complications after transforaminal endoscopic decompression surgery for lumbar foraminal and lateral recess stenosis: a value proposition for outpatient ambulatory surgery. Int J Spine Surg 2019 13:53-67.



26. Ramirez LF, Thisted R. Complications and demographic characteristics of patients undergoing lumbar discectomy in community hospitals. Neurosurgery 1989 25:226-30.


27. Asch HL, Lewis PJ, Moreland DB, et al. Prospective multiple outcomes study of outpatient lumbar microdiscectomy: should 75 to 80% success rates be the norm? J Neurosurg 2002 96(1 Suppl):34-44.


28. Tafazal SI, Sell PJ. Incidental durotomy in lumbar spine surgery: incidence and management. Eur Spine J 2005 14:287-90.


29. Kraemer R, Wild A, Haak H, et al. Classification and management of early complications in open lumbar microdiscectomy. Eur Spine J 2003 12:239-46.


30. Schizas C, Neumayer F, Kosmopoulos V. Incidence and management of pulmonary embolism following spinal surgery occurring while under chemical thromboprophylaxis. Eur Spine J 2008 17:970-4.



31. Bakeman R, McArthur D, Robinson BF. Detecting sequential patterns and determining their reliability with fallible observers. Psychological Method 1997 2:357-70.

32. Sibbald M, Cavalcanti RB. The biasing effect of clinical history on physical examination diagnostic accuracy. Med Educ 2011 45:827-34.


33. Zwaan L, Monteiro S, Sherbino J, et al. Is bias in the eye of the beholder? A vignette study to assess recognition of cognitive biases in clinical case workups. BMJ Qual Saf 2017 26:104-10.



- TOOLS
- Related articles in NS
-
Journal Impact Factor 3.2



























