 |
 |
- Search
| Neurospine > Volume 19(2); 2022 > Article |
|
|
Abstract
Objective
Spinal reconstruction after resection of invasive craniocervical junction malignancies is fraught with technical and management considerations as well as a paucity of data in the existing literature. In this study, we describe our experience with craniocervical junction malignancies, especially the influence of radiation on the need for revision spinal instrumentation.
Methods
We performed a retrospective chart review of all patients who underwent occipitocervical fixation between 2011 and 2019 at The University of Texas MD Anderson Cancer Center.
Results
Twenty-five patients had primary malignancies and 12 (30%) had metastatic tumors. Thirteen (33%) underwent a staged resection in multiple operations during their hospital stay. Tumor resection was performed in 19 patients (48%), while only stabilization was performed in 21 patients (52%). Nine patients (23%) underwent expanded endoscopic transclival approaches for tumor resection, 10 patients (25%) an extreme lateral approach, and 2 patients (5%) an anterior open approach. Eleven patients underwent early postoperative radiation therapy (within 3 months) and 8 underwent delayed radiation therapy (between 3 months and 1 year in 7 patients). The revision rate was 8%, with a median time to revision surgery of 42 months. The administration and timing of adjuvant radiation therapy relative to surgery had no significant effect on the need for instrumentation revision on log-rank and Cox regression analyses (p < 0.05).
The craniocervical junction (CCJ) is a complex anatomic region that encompasses the lower clivus, foramen magnum, occipital condyles, and vertebrae of the atlas and axis, with all of their associated ligaments and vascular and neural structures [1]. Primary or metastatic tumors that affect the CCJ can affect the stability of this region, resulting in pain, lower cranial nerve dysfunction, cerebral vascular insufficiency, and myelopathy. It is estimated that only 0.5% of all spinal metastases occur in the CCJ, with the most common histologies being breast, renal, lung, and prostate cancer [2]. The most common primary tumors of the CCJ are chordomas, chondrosarcomas, giant cell tumors, and nasopharyngeal carcinomas [2].
The surgical approach to tumor resection in the CCJ varies according to the underlying pathology, patients’ symptoms, and extent of the disease. Modern endoscopic techniques that are associated with improvements in intraoperative navigation are frequently used to resect ventrally located tumors that extend from the level of the soft palate to the anterior arch of C1 [3]. A transoral extension allows an approach from the lower clivus to up to the C3–4 disc. The far lateral transcondylar approach, with or without mobilization of the vertebral artery, is used to resect dorsal and lateral lesions, as well as tumors located from the mid portion of the dens to the lower body of C2 [4]. These approaches are associated with significant disruption of the ligaments and bony structures, requiring CCJ fixation.
Craniocervical reconstruction is mandatory in the setting of the CCJ malignant disease and extensive adjuvant therapy often is required. Additionally, there are concerns regarding the impact of adjuvant therapy on the achievement of mature arthrodesis, which drives the wide variability in the spectrum of reconstruction strategies reported [5]. It is also unclear whether radiation therapy should be delayed to allow the development of arthrodesis, especially in slow-growing low-grade chondrosarcomas, where the time to the initiation of radiation therapy does not clearly impact long-term outcomes. In addition to possible hindrance of the fusion process, high dose radiation therapy required for local control of chordomas can induce muscle atrophy and subcutaneous scars as well as reducing the blood supply to discs and cartilage. These pathophysiological processes can accelerate degenerative changes within the radiation field. Additionally, there are few outcome data on whether the need for and timing of radiation therapy affect the need for revision spinal instrumentation in CCJ reconstruction patients.
In this study, we evaluated the effect of postoperative radiation therapy on the durability of occipitocervical stabilization in a retrospective review of patients who underwent stabilization for malignant disease involving the CCJ at a large tertiary cancer center. We also describe our experience with CCJ malignanices, especially the influence of radiation on the need for revision spinal instrumentation.
A retrospective review was performed with the following inclusion criteria: all patients who underwent occipitocervical stabilization at The University of Texas MD Anderson Cancer Center (Houston, TX, USA) for a CCJ malignancy between 2011 and 2019 with a greater than 6-month follow-up. The study was approved by the Institutional Review Board (IRB) of The University of Texas MD Anderson Cancer Center (IRB No. PA17-0906), in compliance with regulations set by our institution for the study of human subjects, and it met all Health Insurance Portability and Accountability Act standards. A waiver of informed consent was granted. Patients were identified via a search of a prospectively collected departmental registry. Their clinical charts were reviewed to collect baseline characteristics, imaging and pathology findings, radiation treatments, and chemotherapy regimens. Preoperative imaging (computed tomography [CT] and magnetic resonance imaging [MRI]) results were assessed to determine the extent of tumor invasion of the bony and ligamentous structures of the CCJ.
Decisions pertaining to tumor resection were made in a histology-specific fashion in which radical resection was attempted for primary malignancies. In cases of metastatic disease, the decision to pursue resection versus limited debulking or stabilization was tailored on the basis of the feasibility of adequate tumor control with adjuvant conventional external-beam radiation therapy or spinal stereotactic radiation surgery [6]. The decision to perform occipitocervical fixation was based on the following factors: intractable neck pain, mechanical instability, and resection of critical bony and ligamentous structures because of tumor involvement or as part of the surgical approach. In transcondylar approaches (lateral open, ventral open, or endoscopic), > 70% resection of a single condyle or > 50% of both condyles were the thresholds for fusion. In staged surgical strategies in which the fusion was performed separately from the resection, patients were kept in a halo vest in the interim.
The occipitocervical fixation technique has been well described [5,6]. The construct length was decided based on bone quality, osteoporosis, and additional sites of subaxial disease with the intent of performing the shortest segment fixation feasible. All spinal instrumentation was performed with a midline buttress plate on the occiput, pedicle, or pars screws at C2 and lateral mass screws in the subaxial spine. Arthrodesis was performed with decortication and the use of demineralized bone matrix and local autologous bone when available. Complex plastics closure with rotational flaps was performed in reoperative cases with previous irradiation or a need for extensive soft tissue dissection.
The primary outcome was time to hardware revision, defined as the time from occipitocervical fixation to instrumentation failure requiring revision surgery, stratified by a binary measure of radiation therapy status (received/did not receive) as well as a binary measure of radiation therapy timing (early > 3 months and delayed < 3 months). Reasons for delayed radiation therapy include worry of hurting the durability of spinal instrumentation, wound complications such as dehiscence or infection, or lack of need due to complete tumor resection. Instrumentation failure was identified on the basis of imaging findings (CT or MRI) that demonstrated hardware failure and clinical notes that confirmed the need for revision surgery. We also analyzed the overall survival period of enrolled patients.
The frequency distribution and summary statistics were calculated for all variables. Kaplan-Meier estimates of the primary outcome were calculated, and survival curves, stratified by radiation therapy status, were compared using the log-rank test at a maximum significance of p< 0.05. A Cox regression analysis was used to generate the hazard ratio between the radiation therapy and no radiation therapy cohorts, and chi-square was used to evaluate the statistical significance of the model at a significance level of p < 0.05. All statistical analysis were performed using IBM SPSS Statistics ver. 23.0 (IBM Co., Armonk, NY, USA).
Forty patients who had undergone instrumented occipitocervical fusion at our institution were included. The baseline characteristics of the study cohort are shown in Table 1. Twenty-five patients (63%) had primary malignancies, and 12 (30%) had metastatic tumors; the remaining 3 patients (8%) had osteoradionecrosis. Eighteen patients (45%) had undergone previous radiation therapy, 9 (23%) had undergone previous chemotherapy, 11 (28%) had undergone previous surgery for the same lesion, and 2 (5%) had undergone previous spinal instrumentation. Median follow-up was 18.8 months.
The degrees of preoperative disease involvement of the bony and ligamentous structures of the CCJ are shown in Table 2. The most common bony site affected by tumor was the C1 lateral masses/anterior arch in 22 patients (55%), followed by the occipital condyles 21 (53%) and the C2 body/dens in 21 individuals (53%). Twelve patients (30%) had integrity of all CCJ ligaments compromised by the tumor invasion. Individual CCJ ligament involvement occurred as follows: apical ligament, 6 (15%); transverse ligament, 14 (35%); alar ligament, 11 (28%), 8 (20%) had no significant ligamentous insufficiency suspected. Baseline characteristics were not significantly different between the postoperative radiation and nonpostoperative radiation groups using binary logistic regression analysis, shown in Table 3.
Table 4 shows patients’ surgical details and postoperative data. Tumor resection was performed in 19 patients (48%), while stabilization alone was performed in 21 (52%). Thirteen patients (33%) underwent a staged resection in multiple operations during their hospital stay. Nine patients (23%) underwent an expanded endoscopic transclival approach for tumor resection, 10 (25%) an extreme lateral approach, and 2 (5%) an anterior open approach. Eleven patients (28%) had a halo vest placed between surgical stages. Thirty-two patients (80%) had 3 to 6 levels fused. Seven patients (18%) had significant swallowing dysfunction requiring a percutaneous endoscopic gastrostomy (PEG) before surgery, while 5 (13%) had developed postoperative dysphagia requiring placement of PEG tubes for postoperative nutritional support. Twelve patients (30%) experienced a complication within 30 days after surgery. Postoperative radiation therapy was given as early (within 3 months) after surgery to 19 patients (48%), and in delayed fashion (between 3 to 12 months) in 8 patients. The most common radiation therapy modalities were proton therapy and stereotactic radiation therapy, with average doses of 70 GyRBE (radiation biological equivalents) and 27 Gy to the clinical target volume, respectively.
The median overall survival was 20.3 months. The revision rate was 8% (3 patients), with a median time to revision surgery of 42 months. Fig. 1 shows the Kaplan-Meier curve when using hardware failure and revision surgery as the primary outcome. The Kaplan-Meier analysis stratified by postoperative radiation therapy did not show statistically significant difference of survival rate (Fig. 2, p= 0.54). No statistically significant difference was found between the 2 cohorts (p= 0.54). Similarly, no statistically significant difference was found (p= 0.55) in a Cox regression analysis (Fig. 2). In the Kaplan-Meier analysis of revisions surgery in the postoperative radiation therapy cohort, stratified by the timing of adjuvant radiation therapy, no statistically significant difference of survival was noted between patients undergoing early (less than 3 months postoperatively) versus delayed (greater than 3 months) adjuvant radiation therapy (Fig. 3).
We could not find a correlation between presence and timing of adjuvant radiation therapy influencing the occurrence of revision surgery. The patient who received early radiation had a fall 5 years after surgery and developed traumatic disconnection of the spinal hardware. The patient who received delayed radiation experienced tumor recurrence 3 years after surgery leading to a pathologic fracture of C2 and bilateral rod breakage. The patient who had undergone no postoperative radiation therapy experienced loosening of the lateral mass screws approximately 2 years after surgery.
CCJ malignancies pose unique surgical and management challenges. Given the unique biomechanical function of this region and the contributing bony and ligamentous structures, the indications for and technique of occipitocervical fixation require special consideration. A critical management decision is the need for and ideal timing of postoperative radiation therapy to minimize its negative impact on the wound healing and durability of instrumented spinal stabilization. The results of our study indicate that in general, independent of the timing of radiation therapy, revision surgery after occipitocervical instrumentation was extremely infrequent. The administration and timing of adjuvant radiation therapy did not significantly impact instrumentation outcomes.
The stability of the CCJ stems from its cup-shaped configuration and its ligamentous structures that connect the clivus, dens, and atlas. Instability can be induced by tumor invasion of these critical structures or the surgical approach employed. Table 2 describes the severity of bony and ligamentous involvement in our patient cohort, which are key variables used to guide surgical planning. Any disruption involving the transverse ligament of the atlas, anterior arch of the atlas, or bilateral disruption of the alar ligaments was considered high risk for craniocervical instability and we performed occipitocervical fusion in these cases. The only cases that we did not consider high risk for instability was resection of the lower clivus with isolated disruption of the apical ligament.
For example, the far lateral transcondylar approach has been shown to result in instability, with 50%–75% condylar resection, that is compounded by disruption of the posterior atlantooccipital membrane, along with the alar ligaments [7,8]. Beyond bony and ligamentous resection, the resection of the adjacent muscular structures that are responsible for CCJ mobility and stability also contributes to postresection instability as a result of the extensive extracompartmental resection outside of the CCJ. These surgical considerations factor into determining the indications for postoperative fixation. Champagne et al. [9] recognized that there are indeterminate situations in the need for fixation; in an effort to avoid the consequences of instrumentation on radiation therapy fields, they reported a protocol in which instrumentation can be delayed until the need for fixation is clearly demonstrated on radiographic follow-up in patients with CCJ chordomas.
Spinal reconstruction in the setting of cancer surgery and the need for adjuvant therapy is fraught with the obstacles highlighted above. Because of the rarity of malignancies in this CCJ, the data regarding reconstruction strategies across all spinal sites are limited to retrospective studies; the data regarding CCJ instrumented fixation are even more limited. To this effect, Glennie et al. [10] performed a systematic review to identify optimal strategies in the cancer setting. Regardless of the location along the spine, instrumentation revision rates of 7.7%–10% were reported. Arthrodesis techniques included the use of vascularized autograft, morcelized allograft, and bone graft substitutes. The reported spectrum of techniques highlights the lack of a consensus approach to fixation strategies after cancer ablative surgery. Despite the unique challenges of the CCJ, our revision rate compares well to the results of Glennie et al. [10] and supports the techniques used in our cohort.
Anterior reconstruction was not attempted in any of our cases. We believe that modern spinal instrumentation is adequate for the stabilization of the CCJ junction, if there is at least 50% of one occipital condyle left. As most of our patients require immediate postoperative radiation therapy or chemotherapy, we do not perform harvesting of iliac crest, as the viability of this autograft may be compromised by adjuvant therapy. As we observed a low incidence of hardware failure, we prefer to have an iliac crest autograft available for eventual hardware revision, where radiation therapy will have less chance to negatively affect the newly implanted iliac crest graft. All hardware failures in our study occurred at the distal end of the construct or with rod breakage.
We did not observe the presence of a solid bony fusion that bridged the occipital bone to the subaxial spine in any of our patients. A longer follow-up period than 18.8 months is needed to clarify whether a lack of arthrodesis affects functional outcomes. We did not observe increased hardware failure or woundrelated complications in patients treated with adjuvant radiation therapy. We attribute the lack of wound-related complications to our frequent collaboration with plastic surgeons to perform muscle advancement flaps or complex free flap reconstructions in cases that were considered high risk, which included prior radiation therapy, prior extensive surgery, severe malnutrition, and skin-related trophic changes.
The patients in our series underwent occipitocervical instrumented fusion for the treatment of craniocervical instability due to malignant disease. Patients with primary tumors underwent attempted maximal cytoreduction, which required a staged anterior and posterior approach. It is our practice to perform the anterior operation first, in which the patient’s head is positioned in extension and rotation. Upon the completion of this stage, we place the patient in a halo vest if there is disruption of the transverse and alar ligaments, resection of the anterior arch of C1 or odontoid process, or compromise of more than 50% of the occipital condyle. Upon completion of the posterior approach, spinal instrumentation is applied. We do not use cervical braces in the postoperative period. All 12 patients in our series with metastatic disease were treated with a single-stage posterior approach. In radiation-resistant histologies, the goal was tumor resection to allow adequate spinal cord decompression and delivery of tumoricidal doses of postoperative spinal stereotactic radiation therapy. In radiation-sensitive tumors, we performed posterior instrumented fusion and subsequent external-beam radiation therapy.
The incidence of dysphagia requiring PEG placement highlights the importance of achieving optimal craniocervical alignment. As expected with extensive CCJ malignancies, all patients in this cohort who required percutaneous gastrostomy had preexisting dysphagia due to tumor involvement of the lower cranial nerves. However, swallowing difficulty as a result of suboptimal alignment of the CCJ is well-documented in the medical literature, with rates of up to 24% in patients even without tumor pathology [11]. Several techniques have been reported that use preoperative radiographic parameters to assist in optimizing cervical alignment [12,13]. Consistent with Bagley et al. [14] we found that it is easier to achieve a functionally comfortable alignment in patients undergoing staged surgical strategies with postresection halo vests. The technique allows patients to report their comfort level and be evaluated by speech pathologists in the halo prior to being fused permanently into that position. As a result, in patients with lower cranial nerve deficits, optimal alignment can be used to maintain the ideal function of the muscles responsible for deglutition.
We recognize several limitations of this study, including its retrospective nature, the relatively short follow-up, the small sample size, the lack of standardization in postoperative radiation therapy regimens, the different surgical approaches, the heterogeneous tumor histology, and the variability in the number of levels instrumented. However, we believe that this series represents a real-world sample of rare cases that were managed in a tertiary cancer center. Our data suggest that radiation therapy administered in a short interval following instrumented occipitocervical stabilization does not affect the durability of spinal reconstruction. Longer follow-up and additional studies are needed to clarify the durability of spinal constructs in the absence of documented solid arthrodesis between the occiput and the axial spine.
We report a retrospective series of CCJ malignancies treated with occipitocervical fixation; we found a low incidence of hardware failure and need for revision surgery. Postoperative radiation therapy did not impact long-term fixation outcomes, providing evidence that radiation therapy can be postoperatively administered without delay.
NOTES
Fig. 1.
Kaplan-Meier curve of overall need for revision surgery in patients undergoing occipitocervical fixation for malignant disease.
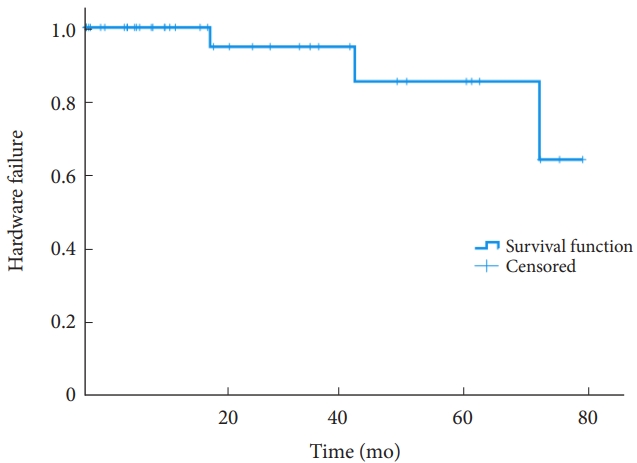
Fig. 2.
Kaplan-Meier curve demonstrating impact of adjuvant radiation therapy on the need for revision surgery after occipitocervical fixation.
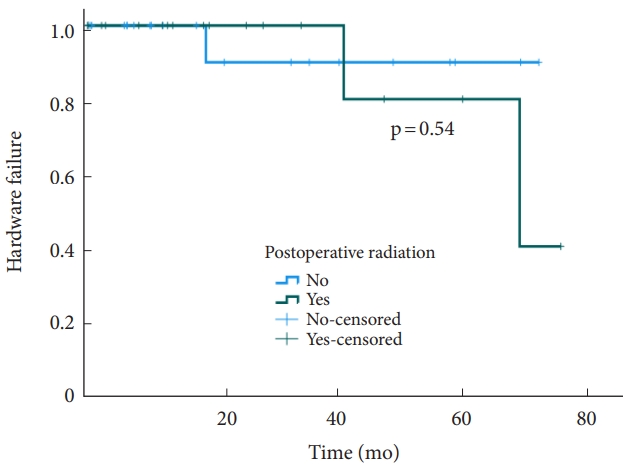
Fig. 3.
Kaplan-Meier curve demonstrating impact of adjuvant radiation therapy timing on the need for revision surgery after occipitocervical fixation.
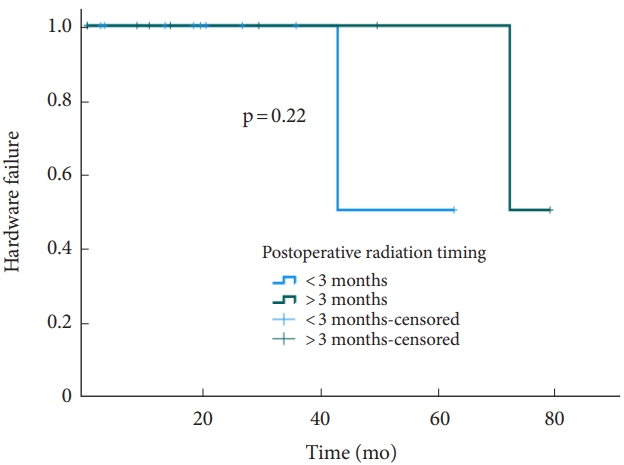
Table 1.
Baseline characteristics of study cohort
Table 2.
Degree of bony and ligamentous involvement
Table 3.
Overview of surgical outcomes
REFERENCES
2. O’Sullivan MD, Lyons F, Morris S, et al. Metastasis affecting craniocervical junction: current concepts and an update on surgical management. Global Spine J 2018;8:866-71.




3. Baird CJ, Conway JE, Sciubba DM, et al. Radiographic and anatomic basis of endoscopic anterior craniocervical decompression: a comparison of endonasal, transoral, and transcervical approaches. Neurosurgery 2009;65(6 Suppl):158-63. discussion 63-4.

4. Kawashima M, Tanriover N, Rhoton AL Jr, et al. Comparison of the far lateral and extreme lateral variants of the atlanto-occipital transarticular approach to anterior extradural lesions of the craniovertebral junction. Neurosurgery 2003;53:662-74. discussion 674-5.



5. Zuckerman SL, Kreines F, Powers A, et al. Stabilization of tumor-associated craniovertebral junction instability: indications, operative variables, and outcomes. Neurosurgery 2017;81:251-8.



6. Fourney DR, York JE, Cohen ZR, et al. Management of atlantoaxial metastases with posterior occipitocervical stabilization. J Neurosur 2003;98(2 Suppl):165-70.

7. Bejjani GK, Sekhar LN, Riedel CJ. Occipitocervical fusion following the extreme lateral transcondylar approach. Surg Neurol 2000;54:109-15. discussion 115-6.


8. Vishteh AG, Crawford NR, Melton MS, et al. Stability of the craniovertebral junction after unilateral occipital condyle resection: a biomechanical study. J Neurosurg 1999;90(1 Suppl):91-8.


9. Champagne PO, Voormolen EH, Mammar H, et al. Delayed instrumentation following removal of cranio-vertebral junction chordomas: a technical note. J Neurol Surg B Skull Base 2020;81:694-700.



10. Glennie RA, Rampersaud YR, Boriani S, et al. A systematic review with consensus expert opinion of best reconstructive techniques after osseous en bloc spinal column tumor resection. Spine (Phila Pa 1976) 2016;41 Suppl 20:S205-11.


11. Wang X, Chou D, Jian F. Influence of postoperative O-C2 angle on the development of dysphagia after occipitocervical fusion surgery: results from a retrospective analysis and prospective validation. World Neurosurg 2018;116:e595-601.


12. Kaneyama S, Sumi M, Takabatake M, et al. The prediction and prevention of dysphagia after occipitospinal fusion by use of the S-line (Swallowing Line). Spine (Phila Pa 1976) 2017;42:718-25.






























