INTRODUCTION
The craniocervical junction (CCJ) is composed of the skull base, atlas, and axis. Schwannomas in the CCJ are classified into 3 types. Type A is primarily intracranial, with minimal extension into the bone. Type B is primarily within the bone, with or without an intracranial component. Type C is primarily extracranial [1]. The location of type C schwannomas is a deep-seated craniocervical space including the internal carotid artery, internal jugular vein, lower cranial nerves, cervical sympathetic chains, and glomus bodies. When accessing type C tumors, care should be taken regarding the deep location and surrounding anatomic structures. Several surgical approaches have been developed to overcome these difficulties. The transcervical approach provides a direct route, but has a limited working space with a vertical extension [2,3]. The endoscopic endonasal approach provides a surgical window [4,5], but there remains a risk of damage to the vessels and cranial nerves.
The anterolateral approach provides a presternomastoid retrojugular route to the CCJ [6,7]. It has been reported that the main indication is an intra-extradural lesion located lateral to the CCJ [8-13]. Although the anterolateral approach exposes the intraextradural spaces and bony structures of the lateral parts of the CCJ, this approach has the advantage of reaching the inner part of the CCJ, which is located in the parapharyngeal space. In this study, we presented 3 cases of surgical treatment of deep-seated extracranial schwannomas in the CCJ through the anterolateral approach, which provided a shallow, sufficient rostrocaudal surgical window in the inner part of the CCJ.
SURGICAL PROCEDURE
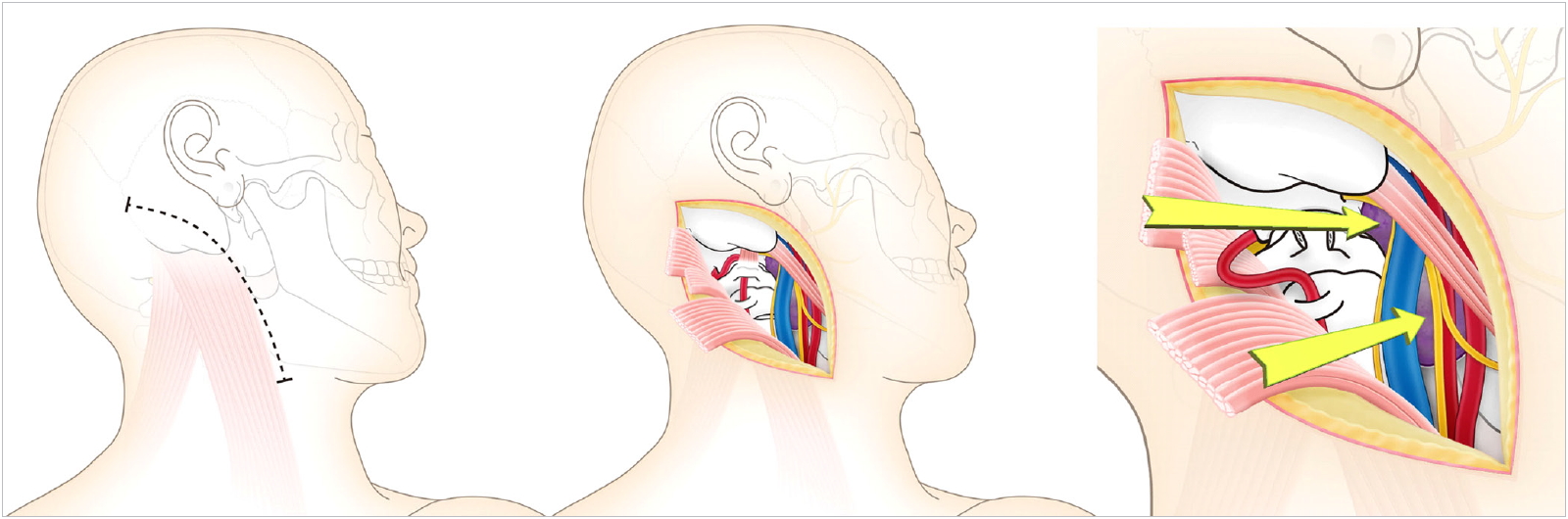
The patient was placed in the supine lateral position with his or her neck turned. The facial and lower cranial nerves were monitored (Nerve Integrity Monitor, Medtronic Xomed Inc., Jacksonville, FL, USA). A curved skin incision was made transversely over the proximal sternocleidomastoid (SCM) muscle, distally along the anterior border of this muscle, and posteriorly to curve across the mastoid process (Fig. 1). We dissected medially to the SCM muscle and laterally to the carotid sheath. The insertions of the SCM muscle and the splenius capitis muscle were divided at the base of the skull. The combination of neck rotation and reflection of the SCM yielded a sufficient surgical field for the CCJ. We could identify the internal jugular vein and accessory nerve, which runs through fat. The tip of the transverse process of C1 could be identified by palpation.
In order to expose the vertebral artery and C1, the superior oblique muscle, rectus capitis posterior major muscle, inferior oblique muscle, rectus capitis lateral muscle, and levator scapulae muscle were divided from the attachment of the C1 transverse process and the occipital bone. After the vertebral artery sheath was exposed in the C1–2 portion and above C1, the C1 posterior arch was partially resected. Vertebral artery transposition was performed to resect the tumors, because it was straightforward to insert and remove microscissors and forceps. We could identify tumors along the accessory nerves and internal jugular veins, and had sufficient working space to resect the tumors (Fig. 1).
Schwannomas are encapsulated and histologically distinct from the nerve itself. We tried to perform subcapsular enucleation of the tumors. Preoperative angiographic examinations demonstrated the patency of the bilateral sigmoid sinus and jugular veins, as well as anastomoses to the anterior, lateral, and posterior condylar veins. All cases showed low flow or no flow of the internal jugular veins on the tumor side, because the tumors compressed the jugular veins. The carotid sheath was displaced anteromedially. Vascular Doppler ultrasonography was used to localize the internal carotid artery in the deep surgical portion.
RESULTS
1. Case 1
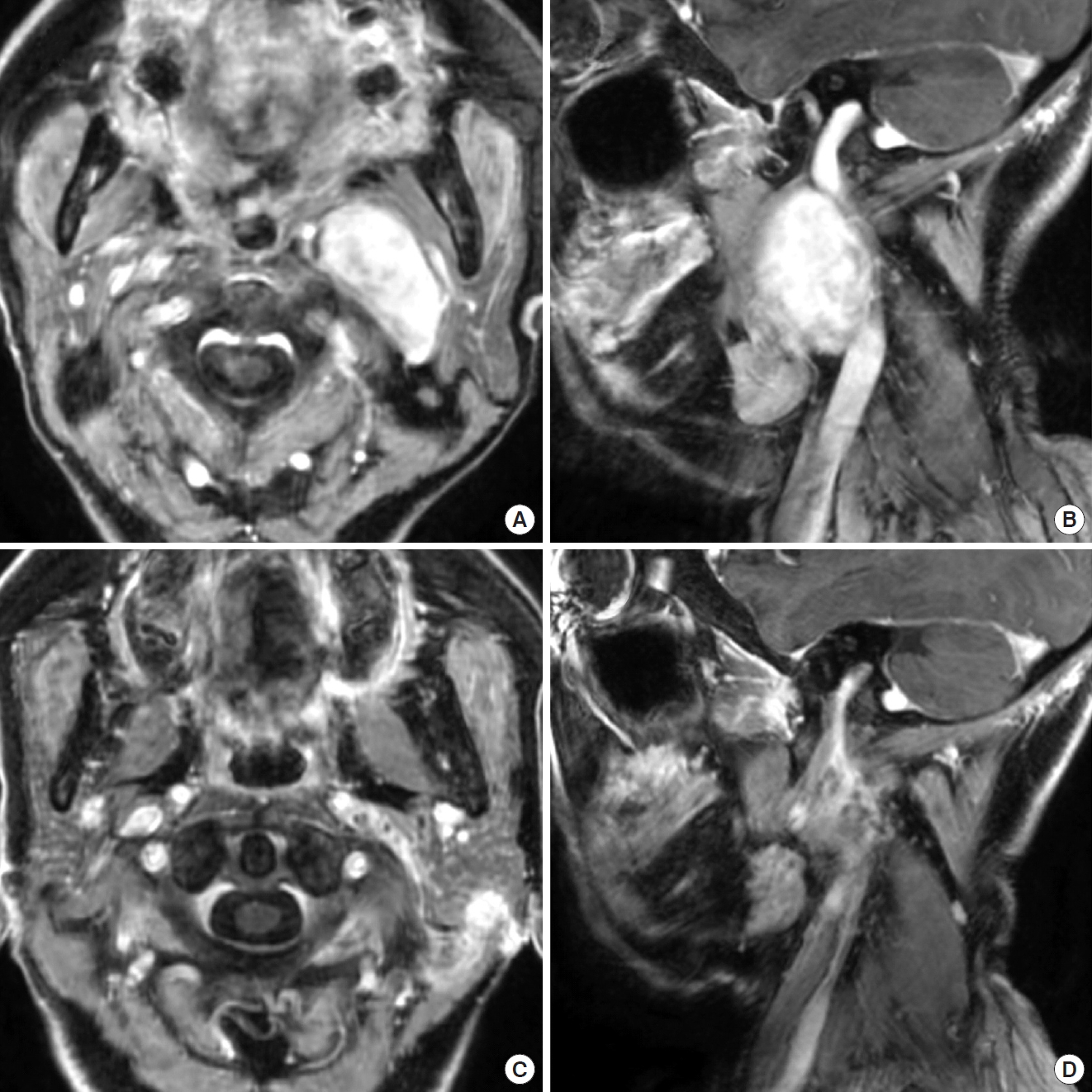
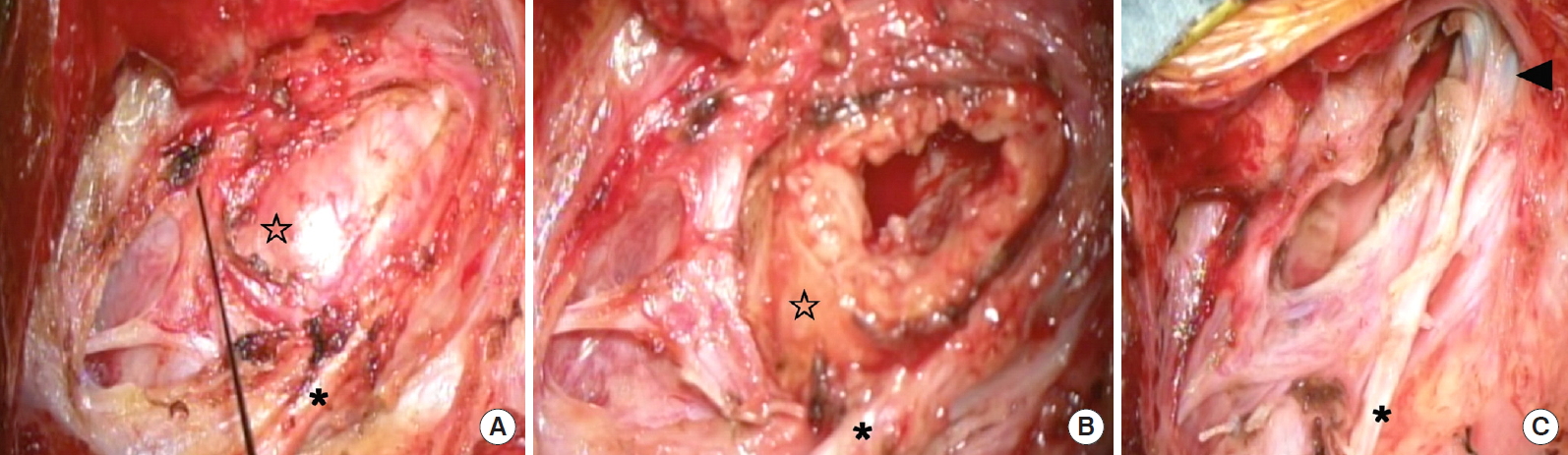
A 58-year-old woman had gradually developed cervical pain and dysphagia. She had systemic lupus erythematosus, and took immunosuppressant and corticosteroid medications orally. Magnetic resonance imaging (MRI) revealed a slightly enhanced mass in the ventral middle to lateral section from the left carotid bifurcation to the jugular foramen (Fig. 2). Angiography demonstrated low flow of the internal jugular vein due to tumor compression, the patency of the contralateral transverse sinus, and collateral flow through the anterior and lateral condylar veins. The carotid sheath was displaced anteromedially. Computed tomography angiography revealed a normal vertebral artery and no neurological symptoms when the neck was rotated. To access the jugular foramen, we sacrificed the internal jugular veins, and approached upward along the tumor. We performed total subcapsular enucleation of the tumors through the anterolateral approach (Fig. 3). The laryngeal nerves crossed the front of the tumors in the surgical field (Fig. 2). Intraoperative monitoring indicated that surgical manipulation did not disturb these nerves. However, she postoperatively presented with transient left vocal cord partial paralysis, and recovered within 1 month. The postoperative diagnosis was schwannoma.
2. Case 2
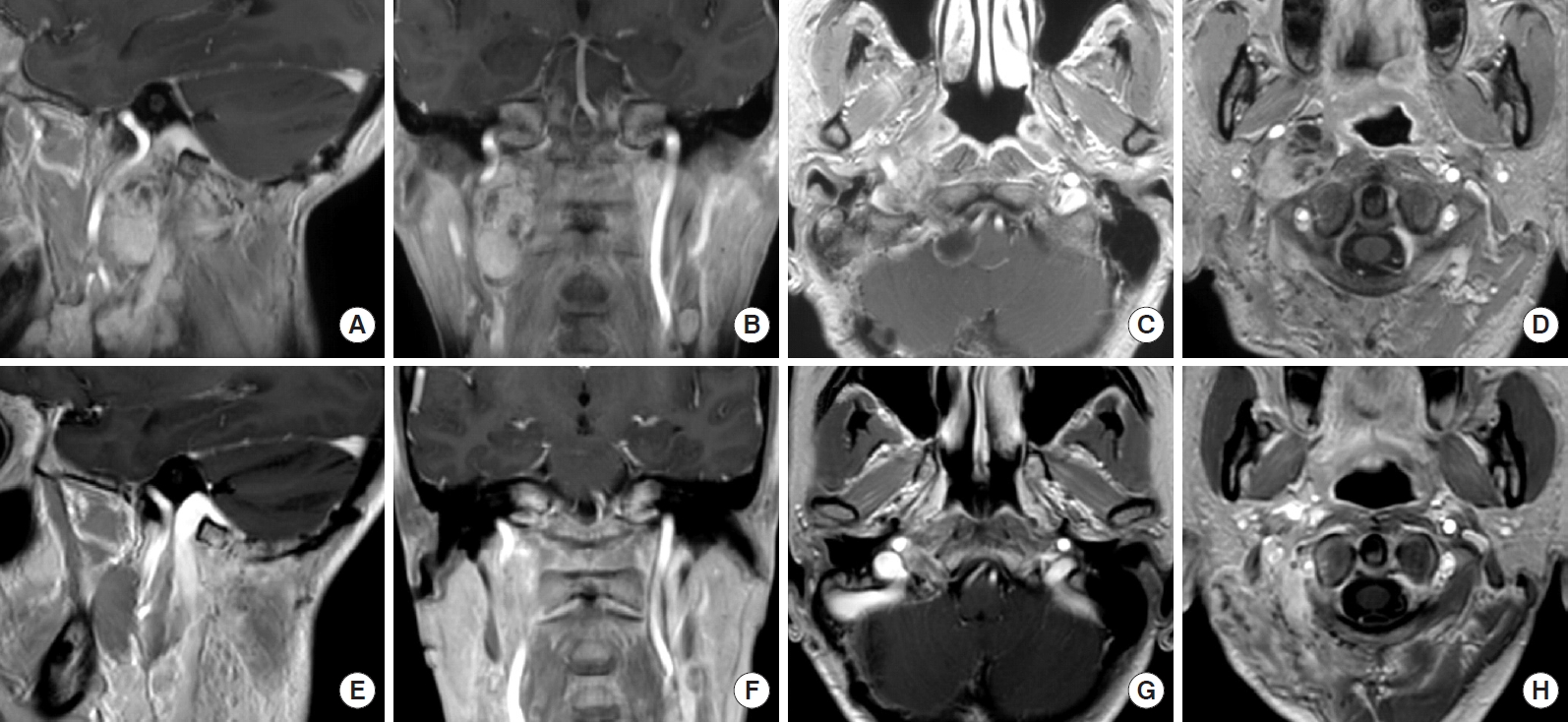
A 43-year-old woman was referred to Osaka University Hospital. She presented with right hypoglossal nerve paralysis. MRI revealed that a mass with slight enhancement was present from the right jugular foramen extending to the ventrolateral portion on the atlantoaxial level, and compressed forward to the pterygoid muscles (Fig. 4). The mass anteromedially displaced the carotid sheath. The internal jugular veins exhibited low flow. The collateral vessels drained into the condylar veins. We performed subtotal resection of the tumors because intraoperative monitoring indicated that surgical manipulation disturbed the vagus nerve and caused action potentials to disappear. We did not dissect the rectus capitis lateralis or transpose the vertebral artery. We could not adequately expose the upper side of the tumors. She postoperatively presented with right vocal cord partial paralysis and dysphagia. Her dysphagia recovered, and vocal cord paralysis and hypoglossal paralysis improved within 1 month. The postoperative diagnosis was schwannoma.
3. Case 3
A 58-year-old woman presented with hoarseness, right vocal cord and right hypoglossal nerve paralysis, and tongue atrophy. She previously had undergone resection of intracranial tumors through the transcondylar approach, and presented with recurrence. MRI revealed intracranial and extracranial tumors passing through the right hypoglossal canal. The intracranial tumors compressed the medulla. The extracranial tumors were present in the ventrolateral portion at the atlantoaxial level, compressed the internal jugular vein, and anteriorly displaced the carotid sheath (Fig. 5). We performed total resection of the tumors through the transcondylar and anterolateral approach (Fig. 6). Postoperatively, she experienced no neurological deterioration. Her hoarseness and vocal cord paralysis improved, but she had almost no change of hypoglossal nerve paralysis. The postoperative diagnosis was schwannoma.
DISCUSSION
Different surgical approaches have been used to facilitate the resection of tumors involving the CCJ and to reduce postoperative morbidity [4,5,14-16]. There are anterior, posterior, and lateral approaches to the CCJ [17]. The lateral approach involves 2 routes: one is posterior to the SCM muscle, and the other is anterior to the SCM muscle [8-10]. The anterolateral approach is also called the extreme lateral approach [18,19]. The indications for this approach are pathologies located in the intra-extradural spaces and bony structures of the lateral parts of the CCJ [20-22]. The anterolateral approach provides exposure of the jugular foramen, vertebral artery, lower cranial nerves, jugular-sigmoid junction, entire odontoid process, the inferior aspect of the lower clivus, and the medial surface of the contralateral atlanto-occipital joint without retraction of the dural sac [12,13]. The posterolateral approach can be divided into the lateral suboccipital approach [23], the far lateral approach [24], and the dorsolateral, suboccipital, and transcondylar approaches [25]. The posterolateral approach easily accesses the lateral and anteromedial parts of the CCJ, and provides excellent exposure of the lower brain stem. However, extensive drilling poses the risk of instability of the CCJ.
The anterior approach to the CCJ includes the transcervical approach, the transparotid approach, the transmandibular approach, the transoral approach, the endonasal endoscopic approach. The transcervical approach allows the surgeon to directly reach the anterior part of the CCJ. However, this approach provides very limited surgical exposure and requires resection of soft tissues. Accessing tumors with vertical extension is difficult. The transparotid approach allows wide access to the anterior part of CCJ, but requires extensive dissection and retraction of facial nerves, which can cause facial nerve injury [13]. The transmandibular approach ensures wide exposure of the anterior part of the CCJ, but often results in nerve injury, malunion, and malocclusion [14]. The transoral approach affords direct access to the anterior part of the CCJ. However, this technique carries a risk of postoperative wound infection and limits the exposure of critical neurovascular structures [26]. The endonasal endoscopic approach has been developed with anatomical studies [15,27]. Endoscopic access to the CCJ utilizes a transmaxillary and transpterygoid approach and provides a sufficient surgical window [6,7,28-30]. Surgeons can extend the surgical corridor laterally and superiorly by removing the lateral plate of the pterygoid process and the posterior wall of the maxillary sinus. However, tumors originating in the post-styloid part of the CCJ pose a greater challenge for endoscopic endonasal approach, as the internal carotid artery, internal jugular vein, and lower cranial nerves obstruct dissection.
The anterolateral approach could provide a sufficient rostral and caudal surgical window for the CCJ. The rotation of the neck moves deep lesions in the CCJ close to the surface. The reflection of the SCM uncovers the posterolateral side of CCJ. The dissection of the superficial and deep layer of the fascia is clear. Although care should be taken regarding the accessory nerves in this approach, we can easily identify these nerves by neuromonitoring. Tumors appeared aside the jugular vein, and created surgical corridors. To open the surgical field to the jugular foramen and hypoglossal canals, it is necessary to control the jugular veins and vertebral artery.
In this study, all cases showed low flow or no flow of the internal jugular veins on the tumor side. In case 1, we confirmed the patency of the contralateral transverse sinus and collateral flow through the anterior and lateral condylar veins, and sacrificed the internal jugular veins to access the jugular foramen without dissecting the rectus capitis lateralis from the occipital bone, drilling the occipital condyle, and manipulating the vertebral artery. In cases 2 and 3, we could not sacrifice the internal jugular veins. In case 2, we did not dissect the rectus capitis lateralis or transpose the vertebral artery. Therefore, we could not adequately expose the upper side of the tumors. In case 3, to approach upward along the tumors, we dissected the rectus capitis lateralis and transposed the vertebral artery. The internal jugular vein is often patent even if a preoperative radiological study shows occlusion or near occlusion of the internal jugular vein. Surgeons should carefully consider the risk of increased intracranial pressure when sacrificing the internal jugular veins.
CCJ schwannomas are encapsulated and histologically distinct from the nerve itself. Treatment is by enucleation, and preservation of the nerve of origin is usually possible. Application of subcapsular enucleation for cervical schwannomas has been reported to result in a reduced incidence of postoperative neurological deficits [31,32]. However, every patient should be cautioned about the possibility of postoperative paralysis.