 |
 |
- Search
| Neurospine > Volume 17(2); 2020 > Article |
|
|
See commentary "Are Surgically Remediable Headaches Associated With Cervical Spondylosis Equivalent to “Cervicogenic Headaches”?" in Volume 17 on page 374.
Abstract
Objective
To evaluate whether anterior cervical spine surgery offers sustained (7 years) relief in patients with cervicogenic headaches (CGHs), and evaluate the difference between cervical disc arthroplasty (CDA) and anterior cervical discectomy and fusion (ACDF) for 1 and 2-level surgeries from a multicenter randomized clinical trial.
Methods
A post hoc analysis was performed of 575 patients who underwent one or 2-level CDA or ACDF for symptomatic cervical spondylosis as part of a prospective randomized clinical trial. Assessment of pain and functional outcome was done with the Neck Disability Index (NDI) in the trial. We used the NDI headache component to assess headache outcome.
Results
For both 1- and 2-level CDA and ACDF groups, there was significant headache improvement from preoperative baseline out to 7 years (p < 0.0001). For 1-level surgeries, headache improvement was similar for both groups at the 7-year point. For 2-level treatment, CDA patients had significantly improved headache scores versus ACDF patients at the 7-year point (p = 0.016).
Conclusion
The headache improvement noted at early follow-up was sustained over the long-term period with ACDF and CDA populations. In the case of 2-level operations, CDA patients demonstrated significantly greater benefit compared to ACDF patients over the long-term. Sinuvertebral nerve irritation at the unco-vasculo-radicular junction and anterior dura may be the cause of CGH. Therefore, it is possible that improved cervical kinematics and preservation of range of motion at adjacent uncovertebral joints in CDA may contribute to the observed difference between the groups.
Cervicogenic headache (CGH) is a common symptom in patients with cervical spondylosis. The primary clinical feature is pain radiating from the neck to the frontal, parietal, and/or orbital regions of the head. CGH is described as a dull, aching, nonthrobbing headache which is often unilateral. The prevalence of CGH in the general population is estimated to be from 1% to 4%, and it accounts for up to 22% of the patients with headaches [1]. In addition, it is a significant issue in patients with cervical radiculopathy or myeloradiculopathy as it occurs in greater than 85% of these patients [2,3].
Though upper cervical spine pathology was traditionally thought to be associated with CGH, spondylotic changes in the lower cervical spine have been shown to be associated with CGH as well. In the treatment of CGH, outcomes of cervical disc arthroplasty (CDA) and anterior cervical discectomy and fusion (ACDF) have been compared, for both single-level and 2-level operations, with significant improvement in headaches, both in the immediate postoperative period and over the short-term postoperative period [3-5]. However, long-term period studies are lacking in this area. Therefore, we analyzed the 7-year follow-up data from a prospective randomized clinical trial to investigate the temporal trend in headache scores after surgery and to identify any difference between CDA and ACDF patient populations over the 7-year period. In addition, we also reviewed the pathophysiology of CGH and describe a likely patho-anatomic explanation for CGH resulting from pathology at the lower cervical spine.
For the initial investigational device exemption study used for the post hoc analysis, the patient demographics, full inclusion/exclusion criteria have been reported previously [4,6]. As part of the aforementioned randomized clinical trial, 575 patients were randomized in a 2:1 (CDA to ACDF) ratio at 24 approved clinical sites in the United States of America between April 2006 and March 2008. The primary inclusion criteria were patients aged 18 to 69 with cervical spine degenerative disc disease with associated radiculopathy or myeloradiculopathy at 1 or 2 contiguous subaxial cervical (C3–7) levels [7].
Assessment of pain and functional outcome in the patient population was performed with the Neck Disability Index (NDI). The NDI is a 10-item self-reported questionnaire with each question scored on a scale from 0 to 5. In our study, the NDI headache component was used to assess headache outcome. The NDI headache component response scale was reported as follows: 0, no headaches; 1, slight headaches that occur infrequently; 2, moderate headaches that occur infrequently; 3, moderate headaches that occur frequently; 4, severe headaches that occur frequently; 5, headaches occurring almost all the time.
A global assessment was performed to compare the mean headache scores over the entire 7-year study period (inclusive of all previous time points from 6 months through 84 months). Global p-values for differences in absolute headache score between CDA and ACDF with 1- and 2-level operations were calculated using repeated measures, mixed-effects analysis of variance (ANOVA), across all postoperative time points. To evaluate the change in outcomes at the 84-month time point, p-values for differences in improvement from baseline in headache scores were calculated using repeated measures, mixed effects ANOVA, adjusted for multiplicity.
ANOVA was used to test the mean improvement across different demographic groups (age, sex, body mass index [BMI]) at 84 months. p-values for categorical endpoints were calculated using a generalized chi-square or Fisher exact test where appropriate. Statistical analyses were performed using SAS ver. 9.4 (SAS Institute, Cary, NC, USA). All patients that were discontinued (e.g., withdrawn, lost-to-follow-up, device removal) were censored at their last follow-up prior to study withdrawal.
At 84 months, NDI headache scores were available for 78.0% (128) of 1-level CDA patients (total 164) and 70.4% (57) of 1-level ACDF patients (total 81). The distribution of headache scores at baseline was similar between the CDA and ACDF groups (p = 0.86), with a median headache response of 3 in both groups (Fig. 1A). At 84 months, the median headache score was 1 in both the CDA and ACDF groups, and there was no difference in the distribution of headache scores (p = 0.83) (Fig. 1B). The percentage of patients with a score of 3 or higher decreased from 53.0% at baseline to 21.1% at 84 months in the CDA group and from 50.6% to 17.5% in the ACDF group. At 84 months, 16.4% (21 of 128) of 1-level CDA patients had worse headache scores from baseline compared with 7.0% (4 of 57) of 1-level ACDF patients (Fig. 1C).
The mean headache scores at 84 months remained significantly improved from baseline in both the 1-level CDA and ACDF groups (p < 0.0001). Globally, there was no difference in headache scores between 1-level CDA and ACDF patients averaged across all follow-up periods (p = 0.85) (Fig. 1D). Specifically, the improvement in NDI headache score was similar between CDA and ACDF (patients 1.2 vs. 1.1, p = 0.94) at the 7-year time point (Table 1).
At 84 months, data was available for 81.8% (184) of patients with 2-level CDA (total 225) and 64.8% (68) of patients with 2-level ACDF (total 105). At baseline, the distribution of headache scores was similar between the 2 groups (p = 0.90): the median headache score at baseline was 3 (frequent moderate headaches) for both CDA and ACDF patients (Fig. 2A). At 84 months, the median headache score was one (infrequent slight headaches) in the CDA group and 1.5 in the ACDF group (Fig. 2B). The distribution of headache scores was significantly different between CDA and ACDF patients (p = 0.028) at 84 months. The percentage of patients with a score of 3 or higher (more than frequent moderate headaches) decreased from 55.1% at baseline to 20.1% at 84 months in the CDA group and from 51.4% to 32.4% in the ACDF group. At 84 months, 12.0% (22 of 184) of patients with 2-level CDA had worse headache scores from baseline compared with 22.1% (15 of 68) of 2-level ACDF patients (Fig. 2C).
The 2-level CDA group demonstrated consistently greater improvement in headache scores from baseline at all follow-up periods compared with the ACDF group. As with earlier follow-up periods, the mean headache scores at 84 months remained significantly improved from baseline for both the 2-level CDA and ACDF groups (p < 0.0001). Globally, 2-level CDA patients had significantly lower headache scores than 2-level ACDF patients averaged across all follow-up periods (p = 0.025) (Fig. 2D). Specifically, the CDA group also had greater improvement in NDI headache scores than the ACDF group (1.3 vs. 0.6, p = 0.016) at the 7-year time point (Table 1).
In both the 1- and 2-level cohorts, the mean improvement in headache score was not significantly different between CDA and ACDF patients stratified by age, sex, and BMI (Table 2).
CGH is described as a dull, aching, nonthrobbing, often unilateral pain experienced in the distribution of first trigeminal branch, generally thought to arise from pathology in the cervical spine. It may be accompanied by sympathetic symptoms such as conjunctival injection, runny nose and tinnitus [8]. Severe attacks in some patients have been described with “migrainous” phenomena, such as nausea and vomiting. The headache can be provoked in most patients by certain movements of the neck, coughing, sneezing, bowel movements, and/or Valsalva maneuvers [8,9]. Nearly all patients with CGH tend to have myofascial trigger points on the symptomatic side [10].
While there does not appear to be a consistent consensus among all the headache specialists regarding the definition of CGH, we found the International Headache Society’s classification of headaches (ICHD-3) to be helpful. They note the diagnostic criteria of CGH include clinical and/or imaging evidence of cervical spine disorder and at least 2 of the following characteristics: A, temporal relation of origination of headache with cervical spine disorder; B, resolution or improvement in cervical spine disorder correlates with resolution or improvement of headache; C, headache can be provoked by certain neck movements and/or Valsalva maneuvers; D, headache eliminated after diagnostic cervical spine injections [11].
The reported prevalence of CGH varies from one to 4.1% in the general population [1]. A recent study in the Japanese population using ICHD-3 criteria estimated prevalence of CGH to be approximately 22% of all headaches [10]. Greater than 85% of patients with cervical radiculopathy or myeloradiculopathy are noted to have associated CGHs [2,3]. The high prevalence, significant morbidity, and decreased quality of life with CGH underscore the importance of understanding its pathogenesis [12].
CGH is a distinct entity that needs to be differentiated from other causes of headache. Chronic paroxysmal headache (CPH) and C2 neuralgia can closely mimic CGH [13]. Whereas CGH patients typically have 1 or 2 episodes every day, CPH patients are differentiated by having usually five to 15 episodes per day, that are usually amenable to indomethacin treatment [8]. Separately, patients suffering from C2 neuralgia typically present with lancinating occipital pain associated with lacrimation and ciliary injection. The underlying cause of C2 neuralgia is the involvement of the C2 nerve root in inflammation and/or fibrosis in conditions such as meningioma, neuroma, or anomalous vertebral arteries. In addition, greater occipital neuralgia is a clinical condition characterized by pain in the occipital region, thought to be mediated through greater occipital nerve entrapment and/or affliction. However, no conclusive evidence has been found in favor of that particular theory, and it is now widely considered to be a referred pain from upper cervical joints. Similarly, the diagnosis of cervical migraine, also known as Barre-Lieou Syndrome, was initially attributed to vertebral ischemia mediated by cervical sympathetic nerve compression/stimulation but was later discredited as it was not supported by experimental evidence [14-16]. Overall, given the ICHD-3 diagnostic criteria, the main symptoms, when experienced a by a patient, that should raise suspicion for CGH are unilateral pain, often in the ipsilateral shoulder diffusely as well, reduced range of motion of the neck, and pain relieved by anesthetic blocks [17].
The diagnosis and underlying pathogenesis of CGH have been a subject of much controversy and speculation [9]. CGH is believed to be mediated through the convergence of nociceptive fibers from the upper cervical nerves (C1, C2, and/or C3) to the trigeminocervical nucleus, leading to referred pain through the trigeminal afferents. A similar mechanism is postulated in bifrontal or orbital headaches in patients with posterior fossa tumors. It is thought that the downward displacement of the cerebellar tonsils leads to stretching of dorsal C1 rootlets resulting in frontal area referred pain through trigeminal convergence [18].
Despite this theory, CGH has been clinically noted in patients with lower cervical spine pathology as well [19,20]. While the trigeminocervical convergence theory explains the origin of CGH from the upper cervical spine, it does not provide a direct neuroanatomic basis for the causation of CGH from the lower cervical spine. Several hypotheses have been proposed by investigators to explain CGH etiology from the lower cervical spine. One hypothesis postulates indirect or intermediate mechanisms, such as abnormalities with muscle tension and kinematics, in causing CGH [21]. Other proposed mechanisms are pain mediated by limited range of motion due to spondylosis leading to over mobility of the upper cervical segments or a possible communication between the spinothalamic tract and the trigeminal complex [10]. However, none of these theories has substantial evidence supporting them, so a prevailing theory for how lower cervical spine pathology can cause CGH is still lacking.
Paramount to understanding the mechanism of CGH in cervical spondylosis is appreciating the intricacies of the innervation of the cervical spine. The dura mater and extradural structures of the cervical spine are innervated by the anterior and posterior primary rami of that corresponding cervical level’s nerve root. The posterior primary rami innervate the portions of the cervical spine dorsal to the vertebral foramen, i.e., the zygohypophyseal joints and the posterior portion of the cervical vertebrae. The anterior primary rami innervate the portion of the cervical spine anteromedial to the neural foramen, including the intervertebral discs and the anterior dura mater, primarily through the sinuvertebral nerve (SVN).
The SVN was thoroughly detailed in the 1960s by Drs. Edgar and Nundy [22] through cadaveric studies, ending several controversial ideas associated with its course and innervation. The SVN, also known as the ramus meningismus or recurrent meningeal nerve of Luschka, is described as a group of 5 to 6 perivascular filaments with one usually being more prominent than the other filaments. It originates primarily from the anterior primary ramus of the cervical nerve root with contributing fibers from the sympathetic trunk in the form of the gray ramus communicantes (Fig. 3A). At certain levels, it is also joined by branches from the vertebral nerve, a sympathetic nerve coursing along the vertebral artery [23]. After its origination, the SVN travels superomedially in the vertebral foramen, anterior to the cervical nerve root to enter the spinal canal. The SVN divides into 3 main branches inside the spinal canal at each segment (Fig. 3A, B). The superior segment interconnects with the descending branches of the SVN from the superior levels. The middle branch runs horizontally and innervates the intervertebral disc, posterior longitudinal ligament, and the ventral dura mater. The inferiorly traversing branch extends inferiorly to join with ascending fibers of the lower segments. At the lateral recess of the spinal canal, in the extradural space posterior to the uncovertebral joint, it is closely associated with the rich venous plexus and arterioles. It then crosses over the vasculature medially, diving into smaller branches to innervate the dura mater and the intervertebral disc. The SVN also innervates the cervical dural sheath, predominantly in the ventral aspect extending up to the dentate ligament laterally.
The small size of the nerve fibers of the SVN and the presence of naked nerve endings are indicative of its nociceptive function. Because the inferior branch of the SVN can reach up to 3 segments below its origin, nociception from the lower cervical segment, as far as C6, can be mediated through the third cervical nerve, which will eventually project to the trigeminocervical nucleus and, thereby, potentially cause CGH. Even SVN branches from C7 can ascend up to the adjacent level and mediate nociception through communication with the aforementioned inferior descending SVN branches of C3. It is also conceivable that SVNs from C1/C2/C3 levels (upper cervical spine) would innervate their corresponding levels more densely than the lower cervical spine (C4/C5/C6/C7) through their descending branches. This may be the explanation for the predilection of CGH associated with lower cervical spine pathology to be less severe than CGH symptoms arising from upper cervical levels [3].
In regard to neuroforaminal compression seen in cervical spondylosis, the entry zone to the foramen is typically much narrower compared to the exit, thus making it a more likely source of neural compression and, subsequently, radicular symptoms. The ventral border of this foraminal entry zone is formed by the uncovertebral joint, and the dorsal border is formed by the medial facet joint. This narrow corridor (dark shaded area, Fig. 3A) houses the SVN plexus, the cervical vasculature (anterior interior portion of venous ring and the segmental radicular artery), and the cervical nerve root, forming an unco-vasculoradicular (UVR) junction (Fig. 3A, B).
In the pathologically spondylotic cervical spine, the narrow UVR zone is usually further constricted by intervertebral disc protrusion and/or a hypertrophied uncovertebral joint. This can lead to indirect or even direct compression of SVN and sympathetic neural elements resulting in CGH. Additionally, a phenomenon most often noted intraoperatively and less so on radiographic studies, engorgement of the epidural venous plexus, which lie in the UVR zone, could lead to SVN compression and cause CGH. This would be akin to the neurovascular trigger mechanism described in trigeminal neuralgia.
Other possible mechanisms leading to neural compression in the UVR zone may include stretching of the dura mater due to cervical kyphotic deformity or segmental cerebrospinal fluid (CSF) entrapments resulting from central canal stenosis. Since the dural investment and CSF surrounds the nerve root up until the lateral border of the neural foramen, it is possible that mechanisms that increase CSF pressure (such as any type of Valsalva maneuver) can trigger nociception, which is frequently noted clinically in CGH patients.
While these possibilities can explain the actual headache of CGH, the autonomic symptoms commonly seen in CGH patients are not directly accounted for in the above theories. However, autonomic connections are known to exist between the cervical plexus and the hypoglossal and vagal nerves through the C1 and C2 nerve roots. Furthermore, the superior cervical sympathetic ganglion directly connects with the C1, C2, C3, and C4 nerve roots through the vertebral nerves. Thus, the irritation of the ventral dural nerve endings and/or impingement of the cervical nerve roots seen in cervical spondylosis could aggravate the sympathetic afferents, and explain the autonomic symptoms frequently seen in association with CGH.
It has also been shown that treating neuroforaminal compression, whether temporarily or more long-term, leads to decreased CGH symptoms. In regard to the more temporary treatment, Bogduk et al. [23] showed that in 161 patients undergoing selective nerve root block for cervical radiculopathy, in both upper and lower cervical spine, 59% patients had a reduction or complete relief of headache symptoms. A more long-term solution to neuroforaminal compression is uncoforaminectomy with foraminal decompression. This has been shown to be effective in treating CGH symptoms, and the headache relief is noted to be both immediate and durable in these cases [8]. Recently, Liu et al. [5] reported a consecutive series of 34 patients treated with anterior decompression and fusion, leading to headache improvement in all patients. Posterior cervical laminectomies have also been shown to provide positive results in regards to CGH relief and/or reduction (12 of 15 patients, 80% success rate), specifically in patients with a narrow cervical canal [10]. However, the headache relief with a posterior approach is noted to be less durable than the improvement seen in anterior approach surgeries [10]. Though the overall severity of headaches in patients treated with a posterior approach was reduced significantly, a small number of patients who were initially relieved of headaches had recurrences at 1-year follow-up [10]. Yet, none of the patients in that study relapsed to severe headaches after posterior laminectomies [10]. The authors postulated that upright posture and mobility over time could be the reason for CGH exacerbation [10]. We theorize that performing posterior laminectomy provides an indirect decompression of the UVR zone and anterior dura mater. This would explain the overall decrease in headache severity at early follow-up and recurrence of a low-grade headache over the long-term.
In contrast, the ACDF and CDA operations provide direct elimination of the anterior underlying pathogenesis and thus provide more durable results. Furthermore, it has been shown that performing an adequate uncoforaminectomy and removal of the posterior longitudinal ligament are critical to headache relief with these anterolateral approaches [8]. An anterior cervical approach with discectomy provides an additional advantage of eliminating a potentially concomitant discogenic nociception.
In our 7-year follow-up results, the 2-level CDA group showed significantly greater improvement in headaches compared to the 2-level ACDF group. Improved kinematics, CSF flow, and possibly lower stress and less arthropathy at adjacent segments, in contrast to fusion operations, might contribute towards better outcomes observed with CDA over the long-term. It will be of interest and value to compare the results between the 2 groups with even longer follow-up.
This is a post hoc study with inherent limitations from relying on the headache component of the NDI. Numerical value from 0 to 5 assigned by the patient was used to derive headache scores, and no further quantification was performed or possible in retrospect. Further details about the headaches such as the character, location, and duration were not able to be obtained. Understandably, not all headaches that the patients reported were cervicogenic in nature, even though it has been shown that, in patients with cervical spine disorders, patients with CGH had significantly higher NDI scores than compared to patients without CGH [10]. However, based on the limitations of the data, we did assume any headache that improved after cervical spine surgery was deemed to be CGH. Given the above information, we made the assumption that other types of headache are distributed evenly among the study arms and would not affect the outcomes substantially. This is, of course, a noteworthy limitation, however.
The follow-up rates for the groups are also different and the study was not blinded so patient perception may have also biased the study. Minimally clinically important difference for NDI total has been established for patients with radiculopathy to be about 15 points on the 100-point scale (15% improvement) [24]. As each NDI component is measured on a 5-point scale, it seems reasonable to propose that a 1-point difference (20%) in headache score is clinically significant. However, a great precaution must be taken in drawing the wrong conclusion: headache should not be a primary indication for cervical spine surgery.
In this study, we showed that the headache improvement, as noted at early follow-up, was sustained over the long-term (7 years - the longest duration documented to date) with both ACDF and CDA populations. In the case of 2-level operations, CDA patients demonstrated greater benefit compared to ACDF patients over the long-term. Improved cervical kinematics and preservation of range of motion with favorable mechanics at adjacent uncovertebral joints in case of CDA may contribute to the observed difference between the CDA and the ACDF groups. The nociception in CGH due to spondylosis may likely be mediated by neurovascular triggers arising from congestion in the unco-vasculo-radicular region and/or the anterior cervical dura mater. Irritation of the SVN may play a key role in CGH.
ACKNOWLEDGEMENTS
We would like to acknowledge and thank Bill Dolman from Zimmer Biomet for assistance with statistical analysis. A portion of the work submitted herein was presented at The Annual Meeting of the AANS/CNS Section on Disorders of the Spine and Peripheral Nerves- Spine Summit 2019.
Fig. 1.
(A) The distribution of headache scores at baseline in patients with 1-level cervical disc arthroplasty (CDA) or anterior cervical discectomy and fusion (ACDF). p-value from chi-square test comparing headache score distribution of CDA vs. ACDF. (B) The distribution of headache scores in 1-level group at 84 months. p-value from chi-square test. (C) The distribution of changes in headache score from baseline to 84 months in 1-level CDA and ACDF. (D) The mean Neck Disability Index (NDI) headache score (± standard error) from baseline to 84-month follow-up in patients with 1-level CDA or ACDF. p-value from repeated-measures analysis of variance test of CDA vs. ACDF.

Fig. 2.
(A) The distribution of headache scores at baseline in patients having 2-level cervical disc arthroplasty (CDA) or anterior cervical discectomy and fusion (ACDF). p-value from chi-square test comparing headache score distribution of CDA vs. ACDF. (B) The distribution of headache scores in the 2-level group at 84 months. p-value from chi-square test. (C) The distribution of changes in headache score from baseline to 84 months in 2-level CDA and ACDF. (D) The mean NDI headache score (± standard error) from baseline to 84-month follow-up in patients with two-level CDA or ACDF. p-value from repeated-measures ANOVA test of CDA vs. ACDF.
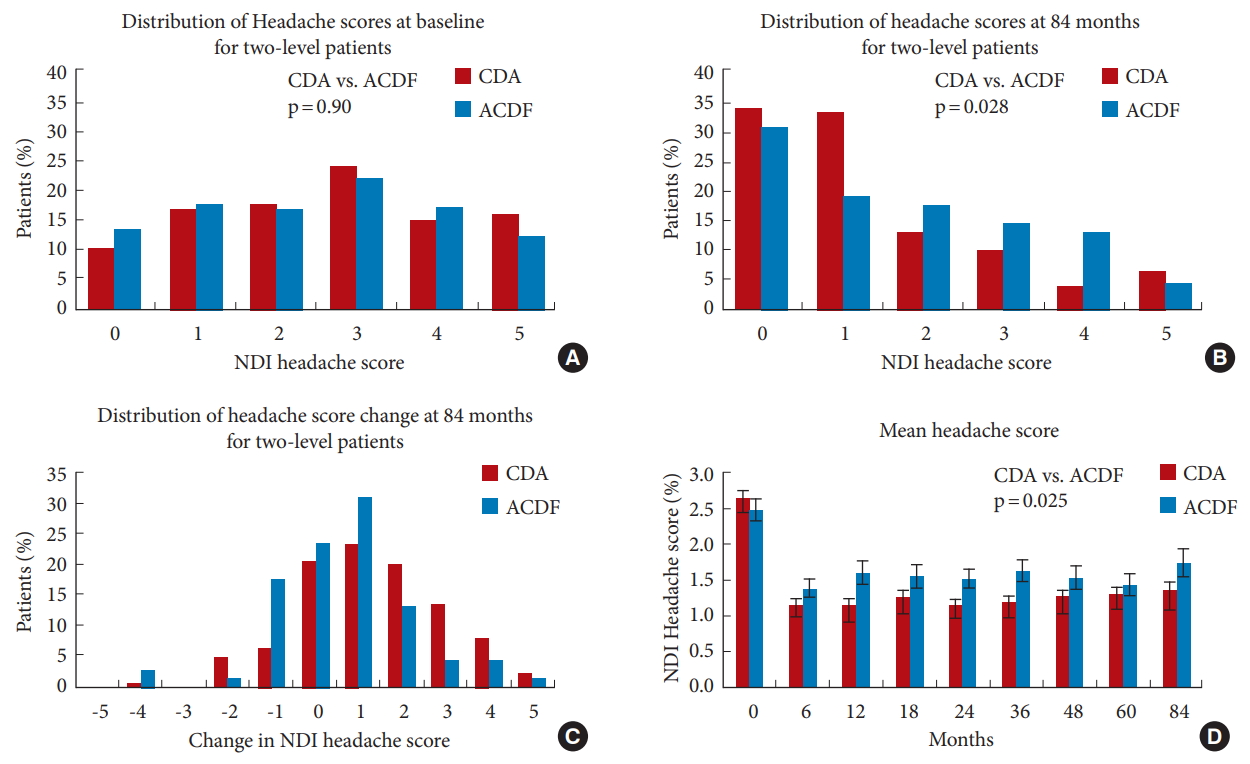
Fig. 3.
(A) Axial schematic illustration of the cervical spine demonstrating the origin and course of the sinuvertebral nerve (SVN). The SVN (green) travels from lateral to medial direction, starting outside the vertebral foramen, posterolateral to the uncovertebral joint, in close anatomic association with the sympathetic and vascular plexus. The rich SVN innervation to the cervical disc is also illustrated. (B) Schematic illustration of the subaxial cervical spine innervation. Demonstrates the longitudinal course of the SVN branches (green), descending up to 3 levels below and anastomosing with branches at the corresponding levels (as deduced from findings in Edgar and Nundy, 1966).22 Note the close association of the epidural venous plexus, SVN, and the arterial branches at the foraminal entry zone.
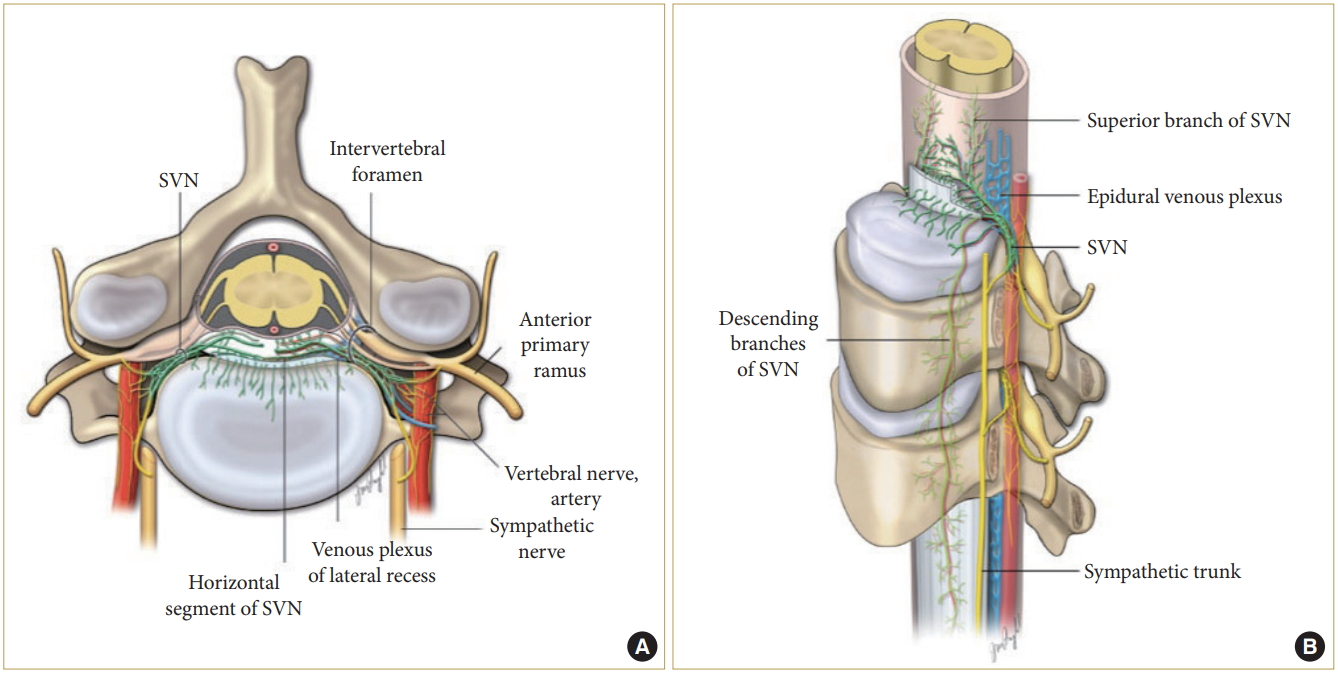
Table 1.
Mean NDI headache scores and change from baseline to 7 years
| Study | Treatment |
NDI headache score |
p-value† | ||
|---|---|---|---|---|---|
| Baseline | 7 Years | Mean Δ at 7 years* | |||
| 2-Level | CDA | 2.6 ± 1.6 | 1.4 ± 1.5 | 1.3 ± 1.7 | 0.016 |
| ACDF | 2.5 ± 1.6 | 1.7 ± 1.6 | 0.6 ± 1.6 | ||
| 1-Level | CDA | 2.5 ± 1.5 | 1.3 ± 1.4 | 1.2 ± 1.6 | 0.94 |
| ACDF | 2.4 ± 1.6 | 1.2 ± 1.3 | 1.1 ± 1.3 | ||
Table 2.
Improvement of NDI headache scores at 84 months in patients with 1- or 2-level treatment stratified by age, sex, and BMI
| Variable |
Mean improvement in headache score |
|||||||
|---|---|---|---|---|---|---|---|---|
|
1-Level |
2-Level |
|||||||
| No. | All patients | CDA | ACDF | No. | All patients | CDA | ACDF | |
| Age (yr) | ||||||||
| 21–49 | 136 | 1.1±1.6 | 1.1±1.7 | 1.1±1.4 | 175 | 1.1±1.8 | 1.4±1.8 | 0.6±1.7 |
| 50–67 | 49 | 1.1±1.4 | 1.2±1.5 | 1.0±1.2 | 77 | 1.0±1.3 | 1.1±1.3 | 0.8±1.4 |
| p-value | 0.98* | 0.64 | ||||||
| Sex | ||||||||
| Female | 97 | 1.2±1.6 | 1.4±1.7 | 1.0±1.3 | 135 | 1.2±1.6 | 1.3±1.7 | 0.9±1.4 |
| Male | 88 | 1.0±1.4 | 0.9±1.5 | 1.2±1.3 | 117 | 1.0±1.8 | 1.3±1.7 | 0.2±1.9 |
| p-value | 0.31 | 0.28 | ||||||
| BMI (kg/m2) | ||||||||
| <30 | 132 | 1.1±1.5 | 1.1±1.6 | 1.2±1.3 | 176 | 1.1±1.7 | 1.3±1.7 | 0.6±1.7 |
| ≥30 | 53 | 1.2±1.5 | 1.4±1.7 | 0.7±0.9 | 76 | 1.1±1.7 | 1.2±1.7 | 0.7±1.5 |
| p-value | 0.71 | 0.82 | ||||||
REFERENCES
2. Riina J, Anderson PA, Holly LT, et al. The effect of an anterior cervical operation for cervical radiculopathy or myelopathy on associated headaches. J Bone Joint Surg Am 2009 91:1919-23.


3. Schrot RJ, Mathew JS, Li Y, et al. Headache relief after anterior cervical discectomy: post hoc analysis of a randomized investigational device exemption trial: clinical article. J Neurosurg Spine 2014 21:217-22.



4. Hisey MS, Bae HW, Davis R, et al. Multi-center, prospective, randomized, controlled investigational device exemption clinical trial comparing Mobi-C Cervical Artificial Disc to anterior discectomy and fusion in the treatment of symptomatic degenerative disc disease in the cervical spine. Int J Spine Surg 2014 Dec 1 8:https://doi.org/10.14444/1007.eCollection 2014.

5. Liu H, Ploumis A, Wang S, et al. Treatment of cervicogenic headache concurrent with cervical stenosis by anterior cervical decompression and fusion. Clin Spine Surg 2017 30:E1093-7.


6. Radcliff K, Davis RJ, Hisey MS, et al. Long-term evaluation of cervical disc arthroplasty with the Mobi-C© cervical disc: a randomized, prospective, multicenter clinical trial with seven-year follow-up. Int J Spine Surg 2017 11:31.



7. Liu JJ, Cadena G, Panchal RR, et al. Relief of cervicogenic headaches after single-level and multilevel anterior cervical diskectomy: a 5-year post hoc analysis. Global Spine J 2016 6:563-70.


8. Sjaastad O, Saunte C, Hovdahl H, et al. “Cervicogenic” headache. An hypothesis. Cephalalgia 1983 3:249-56.


9. Sjaastad O. Cervicogenic headache: the controversial headache. Clin Neurol Neurosurg 1992 94 Suppl:S147-9.


10. Shimohata K, Hasegawa K, Onodera O, et al. The clinical features, risk factors, and surgical treatment of cervicogenic headache in patients with cervical spine disorders requiring surgery. Headache 2017 57:1109-17.


11. Headache Classification Committee of the International Headache Society (IHS). The international classification of headache disorders, 3rd edition. Cephalalgia 2018 38:1-211.


12. Persson LC, Carlsson JY, Anderberg L. Headache in patients with cervical radiculopathy: a prospective study with selective nerve root blocks in 275 patients. Eur Spine J 2007 16:953-9.



13. Bogduk N, Govind J. Cervicogenic headache: an assessment of the evidence on clinical diagnosis, invasive tests, and treatment. Lancet Neurol 2009 8:959-68.


14. Bogduk N, Lambert GA, Duckworth JW. The anatomy and physiology of the vertebral nerve in relation to cervical migraine. Cephalalgia 1981 1:11-24.


16. Lambert GA, Duckworth JW, Bogduk N, et al. Low pharmacological responsiveness of the vertebro-basilar circulation in Macaca nemestrina monkeys. Eur J Pharmacol 1984 102:451-8.


17. Al Khalili Y, Jain S, Murphy PB. Cervicogenic headache. Treasure Island (FL): StatPearls Publishing; 2020.
18. KERR RW. A mechanism to account for frontal headache in cases of posterior-fossa tumors. J Neurosurg 1961 18:605-9.


19. Michler RP, Bovim G, Sjaastad O. Disorders in the lower cervical spine. A cause of unilateral headache? A case report. Headache 1991 31:550-1.


20. Gondo G, Watanabe T, Kawada J, et al. A case of cervicogenic headache caused by C5 nerve root derived shwannoma: Case report. Cephalalgia 2017 37:902-5.


21. Bogduk N. Cervicogenic headache: anatomic basis and pathophysiologic mechanisms. Curr Pain Headache Rep 2001 5:382-6.



22. Edgar MA, Nundy S. Innervation of the spinal dura mater. J Neurol Neurosurg Psychiatry 1966 29:530-4.



- TOOLS
- Related articles in NS
-
Journal Impact Factor 3.2



























