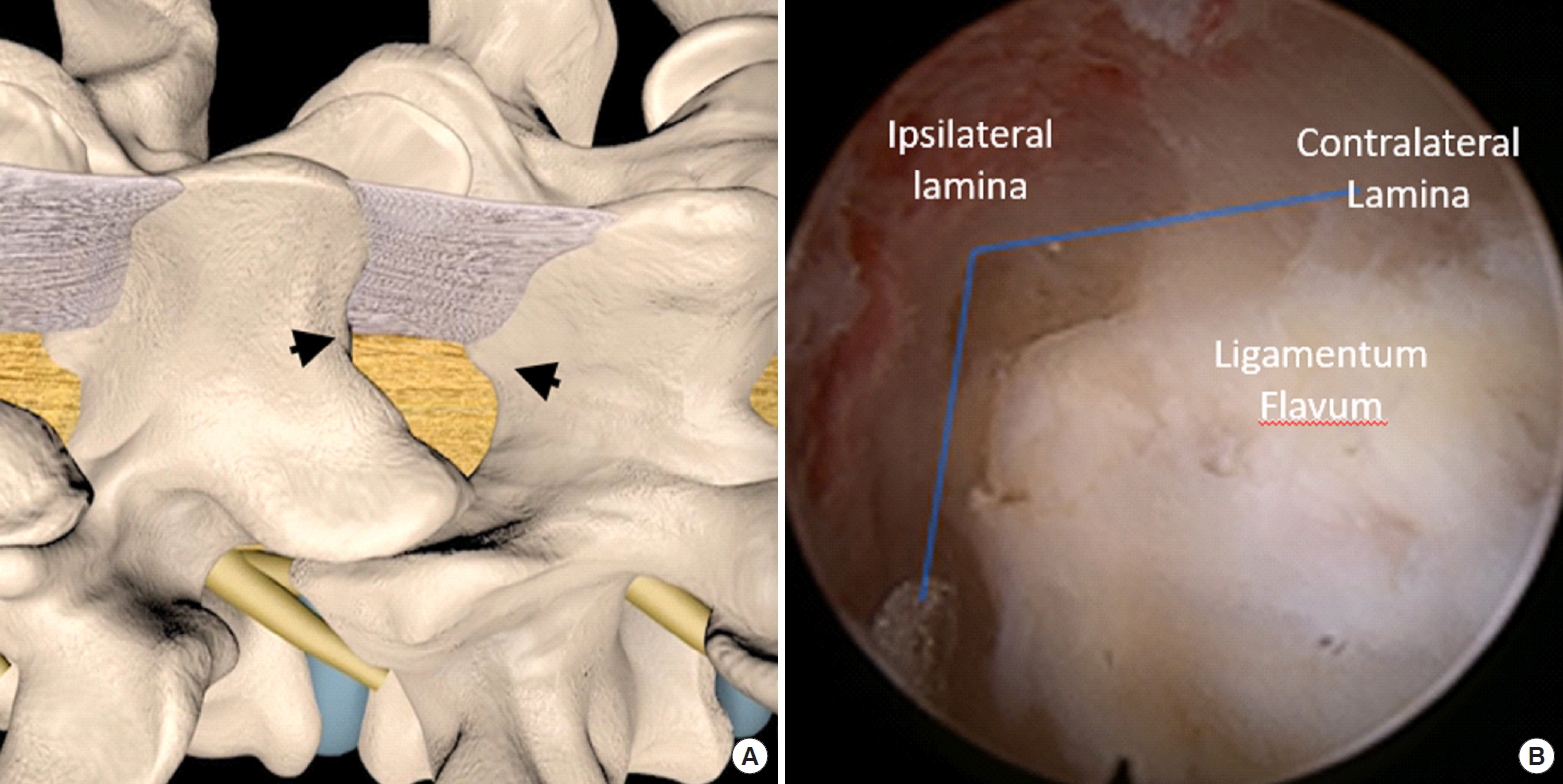
Lumbar Endoscopic Unilateral Laminotomy for Bilateral Decompression Outside-In Approach: A Proctorship Guideline With 12 Steps of Effectiveness and Safety
Article information
Abstract
Minimally invasive spinal surgery in particular lumbar endoscopic unilateral laminotomy with bilateral decompression becomes popular as it can be performed with regional anesthesia, soft tissue damages are minimized as endoscopic visualization and instruments can be brought close to operating area bypassing much of the intervening soft tissues for sufficient spinal decompression with ligamentum flavum resection despite less bony resection compared to open surgery. Overall, when well executed, it preserves spinal stability. Outside-in technique of decompression is also known as over the top decompression in minimally invasive literature. It involves maintaining deep layer of ligamentum flavum integrity till satisfactory bony decompression is achieved. Deep layer of ligamentum flavum is removed as final step of decompression. Preservation of the deep layer of ligamentum flavum protects the neural elements, allowing drills and sharp equipment to be used safely to perform bony decompression.In this study, we demonstrate the technical details of outside-in approach lumbar endoscopic unilateral laminotomy with bilateral decompression (LE-ULBD). LE-ULBD Outside-in Technique is an effective and safe procedure in relieving lumbar spinal stenosis with favorable results with a follow-up for more than 1 year.
INTRODUCTION
As the life expectancy increases globally, aging population leads to increasing prevalence of degenerative conditions [1]. Back pain and disability is the leading cause of musculoskeletal pathology in years of disability [2]. Lumbar spinal stenosis is the most common cause of degenerative spinal pathology in older population [3,4]. There is an increasing need for surgical treatment of lumbar spinal stenosis, yet surgical management in an older population is often complicated by their comorbidities, polypharmacy, increase frailty related with immobility, lower wound healing potentials, and adverse reaction to blood loss and transfusion [3,5]. With the evolution of techniques and indications for endoscopic spine surgery, full endoscopic spine surgery has shown significant benefits for patients with spinal stenosis as the most minimally invasive method of decompression [6,7]. Full endoscopic lumbar decompression for stenosis can be performed by uniportal endoscopy [8,9]. Uniportal full endoscopic stenosis lumbar decompression is recently renamed as lumbar endoscopic unilateral laminotomy for bilateral decompression (LE-ULBD) by AO (Arbeitsgemeinschaft für Osteosynthesefragen) Minimally Invasive task force. In the authors’ opinion, the style used in the literature for interlaminar endoscopic decompression for spinal stenosis can be further divided into inside-out, outside-in technique and contralateral approach [8-10]. The outside-in technique of decompression is what is described as over the top decompression in minimally invasive literature for both microscopic and endoscopic surgery [11-13]. The authors defined outsidein technique of endoscopic stenotic decompression as bony decompression of cephalad lamina, caudal lamina, inferior articular process, and superior articular process (“outside”) to the extent that is sufficient for complete release of ligamentum flavum prior to removal of ligamentum en bloc with blunt instrument in the last part of the procedure to expose the spinal canal (“in”). The inside-out technique, on the other hand, involved bony decompression of lamina, inferior articular process, and superior articular process with early splitting of ligamentum flavum to get into spinal canal (“inside”) prior to releasing ligamentum flavum from within the spinal canal with concurrent bony decompression (“out”) [10]. Contralateral approach involved sparing ipsilateral ligamentum flavum and bony structure, creating sublaminar window working space in the contralateral lamina and performing contralateral ligamentum flavum resection and foraminotomy [8,9,14,15]. This review focus on LE-ULBD outside-in approach which is commonly known as over the top decompression technique. The primary goals are sufficient decompression of spinal canal, removal of sufficient ligamentum flavum, and minimal facet injury in the process. We present the technical details, figures, and a step by step video guide with the results of a retrospective case series of this technique applied in 92 patients who underwent LE-ULBD for 124 levels of lumbar degenerative conditions.
MATERIALS AND METHODS
1. Protocol
The protocol follows the guidelines of Nanoori Hospital’s Human Research Ethics Committee as exempt paper. Written informed consent was obtained from all the patients with neurogenic claudication due to lumbar spinal stenosis.
2. Indication, Inclusion, and Exclusion Criteria
Uniportal LE-ULBD, outside-in approach is indicated in patients with spinal central canal and lateral recess stenosis due to central and paracentral disc protrusion, facet hypertrophy, ligament flavum hypertrophy with Schiaz grade B and above stenosis [16].
The exclusion criteria were the presence of segmental instability, more than grade 1 spondylolisthesis, patients with associated infection, tumor, and fracture in the region of the spinal segment. We also excluded conditions which may require additional procedural steps on top of LE-ULBD such as prolapsed intervertebral disc, foraminal stenosis, ossification of yellow ligaments, and calcified discs.
We collected baseline demographics, performed radiological classification of severity of stenosis by pre- and postoperative Schiaz classification, a comparison was made on radiological grade at preoperative and postoperative 1-year MRI [16]. Visual analogue scale and Oswestry Disability Index at preoperative, 1 week postoperative, 6 months postoperative, and final follow-up were analyzed. MacNab criteria were evaluated at final follow-up. Early and late perioperative complications were documented.
3. Preoperative Preparation
The authors perform anteroposterior (AP), lateral and dynamic view of lumbar spine as well as 3 feet roentgenogram to assess for the sagittal balance, coronal balance, and spinal segmental stability as part of evaluation for surgical indication. Magnetic resonance imaging (MRI) was performed to assess for degree of central, lateral recess, foraminal and extraforaminal stenosis as well as thickness and extent of ligamentum flavum. Computer tomography (CT) scan was performed for the detailed facet analysis such as the facet orientations, size, and shape of the facet as well as the amount of bony osteophytes. This provided the idea of the amount of medial facet resection required for decompression while preserving stability of the facet joint. Three-dimensional reconstruction CT scan was helpful to review the size of interlaminar window narrowing and gave the surgeon a visual impression of the amount of bone resection overall in surgical planning. The operative side was decided based on the collective data of clinical side of symptoms and the amount of stenosis on the preoperative scan.
4. Anesthesia and Skin Incision
The procedure was performed under epidural anesthesia with sedation or general anesthesia, the authors did not need neuromonitoring for the procedure. We placed the patient in the prone position on the radiolucent operating table with the spine in slight flexion using a Wilson Frame. We used a single dose of antibiotics prior to skin incision. The endoscopic procedure was performed with 25–40 mmHg of irrigation fluid pressure. We used irrigation pump to achieve this pressure, the amount of pressure required can be adjusted to provide optimal clarity of endoscopic view. Skin marking was drawn on midline of the spine, as well as the extent of the iliac crest, with a marking pen under an image intensifier. A 1-cm to 1.6-cm skin incision was made vertically at the facet “V” point which was the intersection of (1) most medial inferior point of inferior articular process, (2) medial superior aspect of superior articular process of the caudal lamina, and (3) intervertebral disc line on AP view of intraoperative fluoroscopy (Fig. 1A). The fascia incision was made to 3 cm to allow mobility of the working channel.
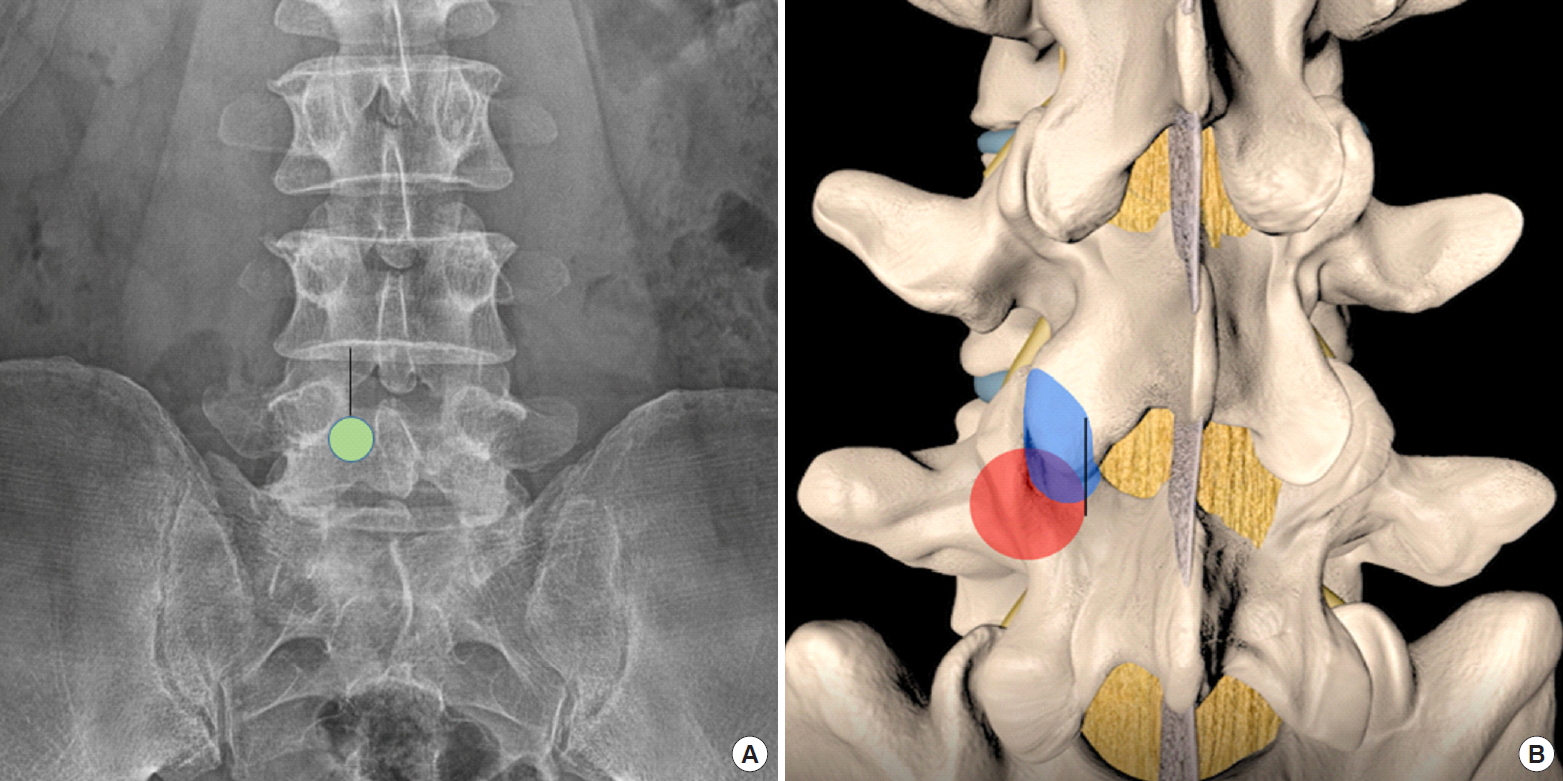
(A) Skin incision (black line) and working channel docking point (green circular area) on anteroposterior view of roentgenogram, the same points can be cross referenced to intraoperative fluoroscopy. (B) Anatomical relation of pedicle (red region) and superior articular process (blue region). The superomedial border of superior articular is adjacent to the perpendicular line of the medial pedicle (medial pedicle line) is the starting point of facet joint. If we keep the decompression lateral to medial pedicle line, we can save sufficient amount of the facet joint.
5. Insertion of Endoscope
Guidewire was placed on the upper part of caudal lamina adjacent to the facet “V” point, serial dilation was made with obturators through which a 13.7-mm beveled tip working cannula was inserted. We perform an intraoperative AP and lateral view at this point of time to confirm the correct level of decompression. We then inserted a 15° viewing angle, outer diameter 10 mm, working channel diameter of 6 mm, and working length 125-mm endoscope to begin surgical procedure (Fig. 2).
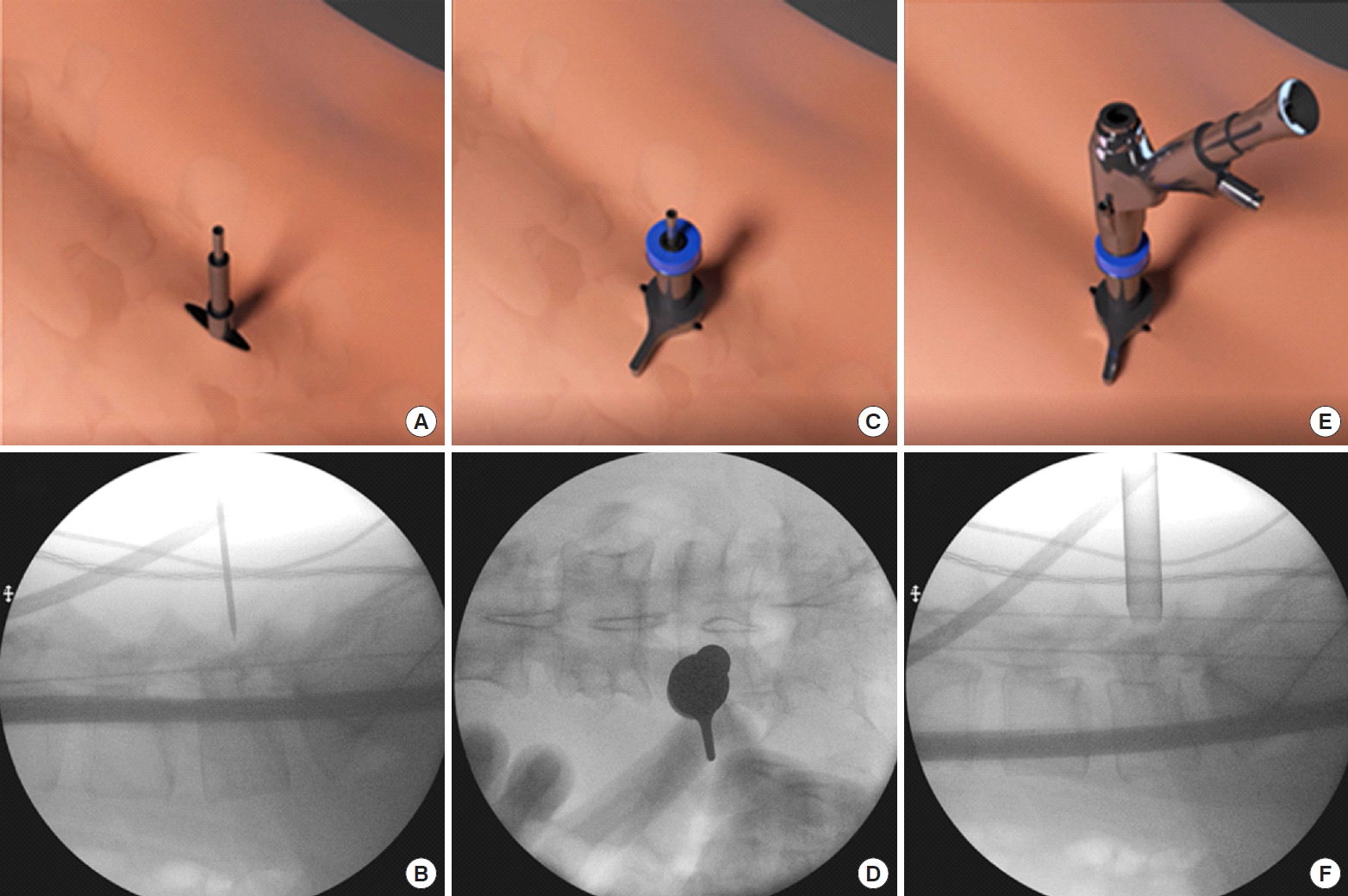
Serial dilation toward the docking point on a mannequin. Skin incision, docking with guidewire, and serial obtruator dilation. Working cannula inserted on the obtruator and endoscope introduced through the working cannula. Panels A and B showed corresponding external and intraoperative flurosocopic position of the docking wire which was on the caudal lamina adjacent to the “V” point described in the text. Panels C and D showed the external and corresponding anteroposterior view and panels E and F showed the external and corresponding lateral view of the docked working channel on the caudal lamina adjacent to the “V” point.
6. Surgical Procedure
After insertion of endoscope, hemostasis was performed with radiofrequency ablator. Soft tissue was dissected off the caudal lamina to expose the inferomedial border of inferior articular process. Inferior articular process was drilled on its medial border in caudal to cranial direction to expose the underlying superior articular process. We were careful as far as possible not to drill beyond the line bisecting medial pedicle to save as much of the medial facet as possible to remove sufficient medial ligamentum flavum (Fig. 1B). The ipsilateral cephalad lamina was drilled to cephalad free margin of deep layer of ligamentum flavum (Fig. 3A) and caudal lamina was drilled to expose caudal free margin of deep layer of ligamentum flavum (Fig. 3B). We typically separated the superficial layer of ligamentum flavum from the deeper layer and pulled out the superficial layer by forceps for better exposure at this point of time. We left the deep layer of ligamentum flavum and its free margin to be removed at the last step of the procedure. We would proceed to decompress the base of spinous process at spinolaminar angle (Fig. 4) and identified the cephalad lamina “V” point which was the landmark of the midline and undersurface of contralateral cephalad lamina (Fig. 3C) followed by a translaminar over the top decompression by drilling the ventral aspect of the spinous process and interspinous ligament to create a sublaminar working space for resection of contralateral ligamentum flavum. This was followed by drilling on contralateral caudal lamina and contralateral medial superior articular facet resection to the lateral margin of contralateral ligamentum flavum which was located at the dorsal entrance of the foramen region in the cephalad lamina and tip of superior articular process (Fig. 3D). Both the deep layer of ipsilateral and contralateral ligamentum flavum was kept intact to protect underlying neural elements till bony decompression was deemed satisfactory. Once the bony decompression was complete, where the ligamentum flavum attachments at (1) laterally at the tip of the superior articular facet, (2) superiorly on the lower half of the cephalad lamina, and (3) inferiorly on the top of caudal lamina are released, ligamentum flavum would be released. At the final step, the ligamentum flavum was removed en bloc exposing the underlying neural elements. This forms the basis of uniportal LE-ULBD, outside-in approach.
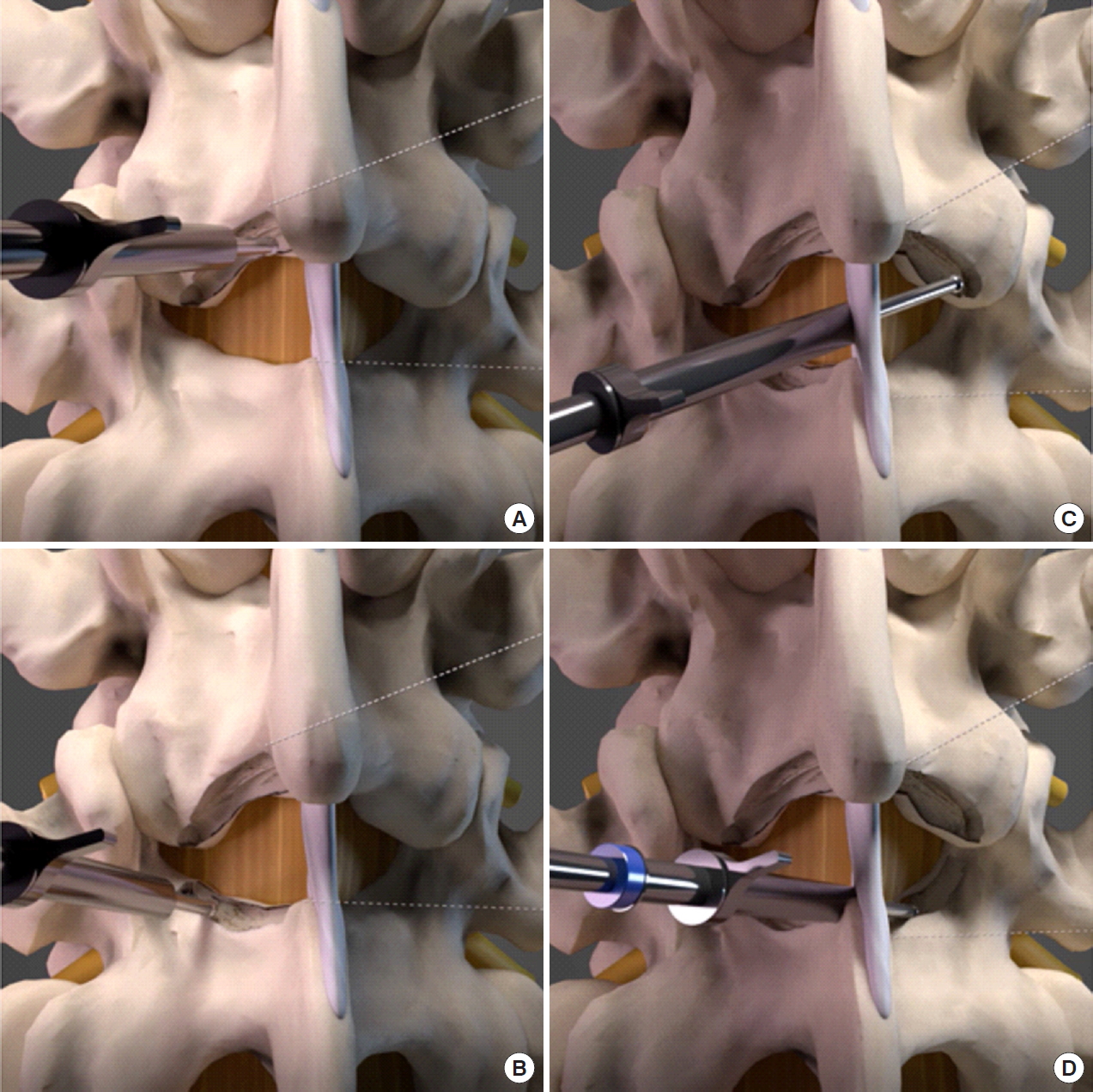
Surgical steps in lumbar endoscopic unilateral laminotomy and bilateral decompression (LE-ULBD) of left L4/5. (A) Ipsilateral cephalad laminotomy. (B) Ipsilateral caudal laminotomy. (C) Contralateral cephalad ventral laminotomy. (D) Contralateral caudal laminotomy.
7. Final Checking Point
Pulsating traversing and exiting nerve roots of both ipsilateral and contralateral side was seen. Both the ipsilateral and contralateral pedicle were both visualized and palpated, lateral recess was assessed to be well decompressed with lateral edge of traversing dura clearly seen on both sides. If necessary, further trimming and decompression of ligamentum flavum and its attached bony structure can be performed with endoscopic Kerrison punches. Disc herniation if seen indenting, compressing or displacing neural elements are removed on a case by case basis. Disc bulge was treated with thermal radiofrequency coagulator shrinkage. Drain was inserted under direct endoscopic vision and anchored and typically it will be removed on postoperative day 1. Skin was closed in layers (Fig. 5).
RESULTS
1. Baseline Demographics
In January to December 2018, a total of 92 patients with 124 levels of lumbar spinal stenosis who met inclusion and exclusion criteria underwent lumbar endoscopic unilateral laminotomy with bilateral decompression. Their mean age was 64.7 (range, 33–87 years) years old with a follow-up of 16.8 months (range, 13–23 months). There were 55 males and 67 females in this group of patients. Sixty-five patients had 1 level, 25 patients had 2 levels, and 3 patients had 3 levels of decompression. Three L1/2, 11 L2/3, 26 L3/4, 72 L4/5, and 20 L5/S1 levels were decompressed with 67.7% of the levels involving L4 to S1 levels. Among the 92 patients, 12 underwent general anesthesia and 80 underwent epidural anesthesia for the surgery.
2. Radiological and Clinical Outcomes
In terms of radiological evaluation, 29 out of 31 levels with Schiaz grade B improved to grade A1–3. Of the 2 patients who had maintained Schiaz grade B pre- and postoperatively, their ligamentum flava were adequately removed and had relieved their symptoms significantly with excellent grade at final MacNab criteria. Fifty-one out of 52 levels of Schiaz grade C improved to grade A 1–4, one patient had Schiaz grade C improved to B with good final MacNab criteria. All of 41 Schiaz grade D levels improved to grade A1–4. We had 9 incidental durotomies during the procedure in 124 levels (7.2%) which required dural patch blocking repair, 3 patients developed facet cyst on the operated level on subsequent follow-up MRI. Two of the patients were asymptomatic and their facet cysts were found as incidental findings on follow-up MRI. There was 1 patient with mild symptomatic neurogenic claudication with facet cyst found on the MRI on his right leg (Fig. 6). He was treated conservatively with occasional pain medications, he achieved a good MacNab criteria on his final follow-up. There was statistically significant improvement of visual analogue scale score with preoperative, 1 week postoperative, 6 months postoperative, and final follow-up mean of 7.88 (6–10), 3.34 (2–5), 2.52 (1–4), and 2.29 (1–4), respectively. As for Oswestry Disability Index, there was statistically significant improvement with preoperative, 1 week postoperative, 6 months postoperative, and final follow-up mean of 74.02 (56–86), 30.56 (22–42), 25.92 (18–42), and 24.62 (16–38), p < 0.05. MacNab criteria showed 3 patients with fair, 99 patients with good and 22 patients with excellent scores.
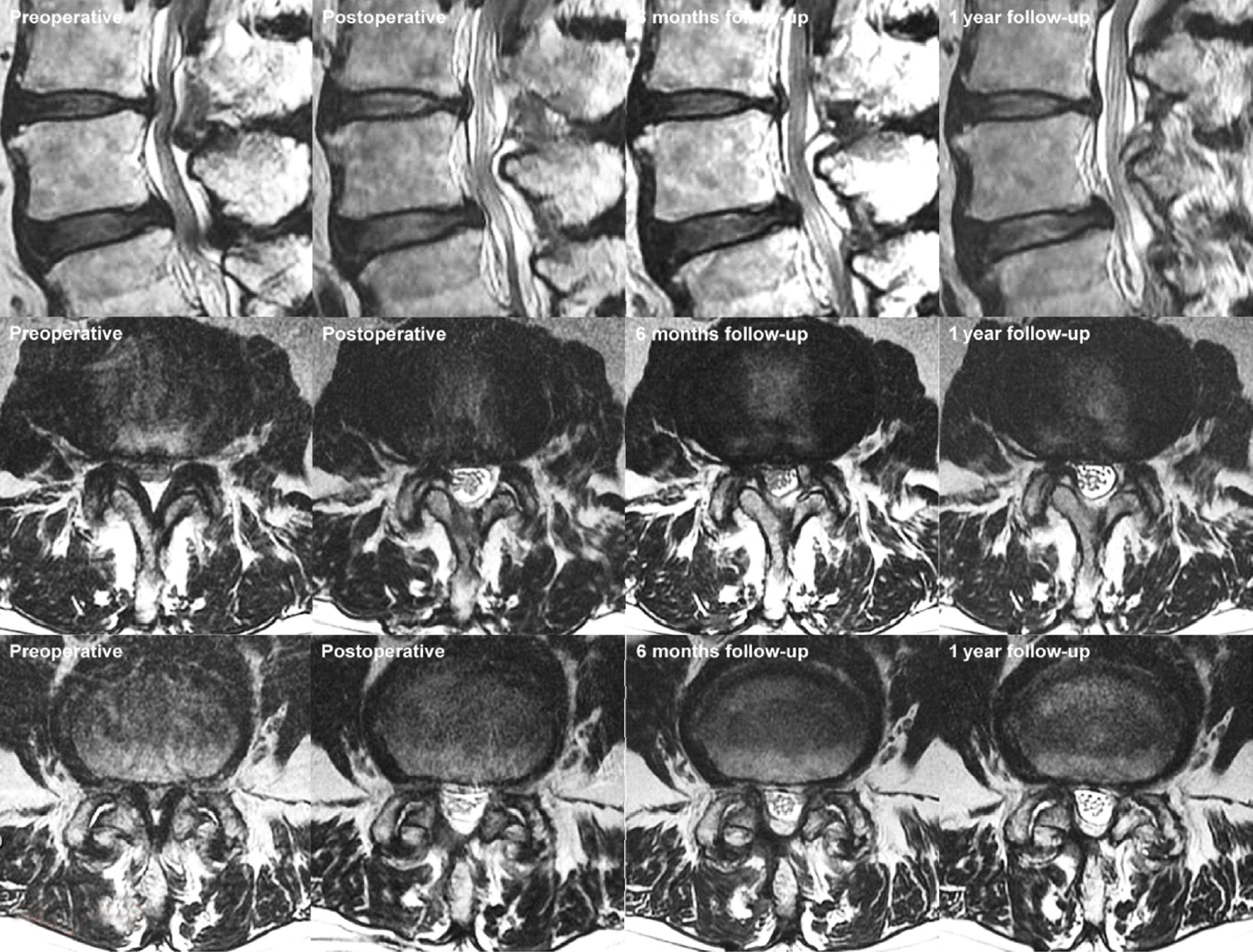
Pre- and postoperative magnetic resonance imaging (MRI) series of L3/4 and L4/5 of an 83-year-old man who presented with bilateral lower limb neurogenic claudication and underwent left L3/4 and L4/5 lumbar endoscopic unilateral laminotomy and bilateral decompression. Left most figures are sagittal and axial preoperative MRI images of Schiaz grade C L3/4 and L4/5 levels spinal stenosis. Corresponding postoperative day 1 images showed Schiaz grade A2 decompression of neural elements at the L3/4 and L4/5 levels. Follow-up scans in 6 months and 1 year showed similar spinal canal patency with good soft tissue and muscle preservation. There was an MRI finding of high signal intensity tissue at the right facet joint which was possible facet cyst developed at L3/4 over time, he was treated conservatively, no intervention nor histology required to confirm the diagnosis.
Figs. 6 and 7 showed 2 clinical case examples of LE-ULBD performed.

A 56-year-old woman with neurogenic claudication, from left to right serial magnetic resonance imaging mid sagittal and axial L4/5 images showed preoperative spinal canal stenosis of Schiaz grade C L4/5 which was decompressed after lumbar endoscopic unilateral laminotomy and bilateral decompression of L4/5 with corresponding postoperative day 1 images demonstrating Schiaz grade A1 decompression of neural elements at the L4/5 levels. Follow-up scans in 6 months and 1 year showed similar spinal canal patency with good soft tissue and muscle preservation.
DISCUSSION
With evolution of endoscopic technique, therapeutic indication of endoscopic spinal surgery had broadened from disc herniation requiring discectomy to current state with stenosis decompression, contralateral decompression, and fusion [6,8,17-19]. The results of endoscopic decompression were promising with comparable or better results than microscopic decompression in various studies [7,20,21].
Unlike the tubular microscopic decompression, endoscope is mobile, there is a significant difference in operative and endoscopic optical angle which can work in the surgeon’s favor by rotating and tilting maneuver to view the hidden areas difficult to be seen by tubular and microscope which is a straight-on view. The optical angle however helped in limiting bony resection required to visualize the edge of ligamentum flavum attachment, preserving the excess bony resection required for viewing in tubular or open microscopic cases.
However, the magnified and narrow operative view can be confusing for the endoscopic surgeon. The optical and working angle is also different with an optical angle typically 15°–30° while working angle of instrument is straight ahead at 0°. This can lead to confusion while performing decompression in the early phase of endoscopic practice. Operating surgeon needs to pay constant attention to key magnified anatomical landmarks which can be confusing after burr and Kerisson rongeur decompression which changed the size and shape of the anatomical landmarks. If in doubts, intraoperative fluoroscopy and/or navigation can help in orientation.
Outside-in technique or over the top endoscopic decompression has the advantage of maintaining the deep layer of ligamentum flavum to offer neural elements protection during bony decompression till the last stages of decompression. Drilling performed superficial to ligamentum flavum protects it from inadvertent durotomy. Learning curve of this technique is significant with a decrease in timing and better results achieved in the more experienced surgeon [22]. Despite significant steep learning curve, clinical results had been favorable [3,7,12,22-24]. Complications rate ranges from 8.3%–16%. Most of the complications are temporary. Overall revision rate is 1.9%, incidental durotomy rate around 0%–8.6%, transient paresthesia around 2.6%, epidural hematoma and headache each around 1.9% [3,7,12,22-26]. These complications are comparable in some and favorable in other studies as compared to open and minimally invasive spinal decompression [27-31]. Infection rate is extremely low in minimally invasive surgery [32]. While limited literature is available for infection rate in endoscopic decompression which is the least invasive version of minimally invasive surgery, the authors felt the rate is comparable if not lower than tubular and mini-open decompression. Part of the reason is constant saline irrigation which helps to flush out any possible bacteria and prevents debris collection, being the most minimally invasive procedure also means smaller incision and less soft tissue damage leading to less necrotic tissue, decreasing risk of infection. In our series, there was 7.2% dura tear and 3% facet cysts formation of which only one patient was symptomatic. There was no infection. This complication rate was comparable to other similar studies [25]. None of the complications required conversion or revision surgery. Incidental durotomy in our LE-ULBD series was small and only required patch blocking dural repair [33].
Keeping the deep layer of ligamentum flavum intact till last part of surgery also keeps the epidural bleeding till the last part of procedure. As epidural bleeding obscures the endoscopic viewing, which may require higher irrigation pressure, causing unwanted side effects of headache, neckache, and even seizure [34]. Hence leaving the removal of deep layer of ligamentum flavum en bloc in final stages has significant advantages in prevention of surgical complications.
One disadvantage of outside-in technique is possibility of over resection of facet joint [22]. Care needed to be taken in preoperative planning as well as intraoperative execution of facet resection. As facet is resected prior to release of ligamentum flavum. Lee et al. [22] found to have excessive facet resection of 5% in his earlier series of cases which dropped to 1.6% in his later series. The author prevents overresection by preoperatively evaluating the size, shape, and orientation of facet as well as presence of facet cyst. Care should be taken in identification of extent of lateral margins of ligamentum flavum. Medial pedicle is a useful landmark to prevent over resection of facet joint. The authors use a uniform size drill bit of 3.5 mm which helps in measurement of the amount of facet resection by using the drill bit as a gauge. Operating surgeon can rotate the endoscope such that the optical angle is favorable to preservation of the facet by looking more medially rather than laterally may aid in facet preservation. On the other hand, it is interesting to see that with facet preservation, there is a possibility of subsequent development of facet cyst in follow-up MRI which happened in 3 of our 92 patients. One of the 3 was having mild symptoms treated with conservative management. It is hence important to remind the patient that progression of degeneration is possible in the arthritic joint with its associated cyst development in LE-ULBD procedure.
The data was obtained as a retrospective study with patients who had undergone LE-ULBD only. There would be inherent selection and performance bias in the study set. Preoperative data such as comorbidities, Charlson Morrison Index, and length of operations time were not collected which might introduce confounders in the study. We limited these confounding factors by having the same team of anesthetists and surgeons for all the operations performed in the data set.
CONCLUSION
LE-ULBD is a safe and effective procedure in relieving spinal stenosis with favorable results.
POINTS OF SURGICAL TECHNIQUE
• Docking accurately on the facet “V” point allows identification of the ipsilateral medial inferior margin of inferior articular facet caudal lamina.
• Drilling of the ipsilateral medial inferior margin of the inferior articular process reveals underlying superior articular process.
• Drilling medial and superior aspect of ipsilateral superior articular process just enough to expose lateral extent of ligamentum flavum.
• Cephalad lamina is drilled to expose cephalad margin of ligamentum flavum.
• Caudal lamina is drilled carefully to expose caudal margin of ligamentum flavum.
• Superficial ligamentum flavum is removed to expose contralateral structures.
• Drilling is continued across midline in contralateral cephalad ventral lamina dorsal to deep layer of ligamentum flavum to the lateral margin of the contralateral ligamentum flavum attachment at contralateral superior articular facet and the entrance of contralateral foramen.
• Contralateral overhanging osteophytes of superior articular process is drilled till the lateral attachment is seen.
• Contralateral caudal lamina is drilled to expose contralateral caudal margin of ligamentum flavum.
• Removal of contralateral ligament flavum followed by ipsilateral ligamentum flavum.
• Final check shows free and pulsatile underlying neural elements in spinal canal.
• Hemostasis and check for any intraoperative complications which require intervention.
Notes
The authors have nothing to disclose.
SUPPLEMENTARY MATERIALS
Supplementary video clips can be found via https://doi.org/10.14245/ns.2040078.039.v.1.