 |
 |
- Search
| Neurospine > Volume 17(3); 2020 > Article |
|
|
Abstract
Craniovertebral junction (CVJ) deformity is a challenging pathology that can result in progressive deformity, myelopathy, severe neck pain, and functional disability, such as difficulty swallowing. Surgical management of CVJ deformity is complex for anatomical reasons; given the discreet relationships involved in the surrounding neurovascular structures and intricate biochemical issues, access to this region is relatively difficult. Evaluation of the reducibility, CVJ alignment, and direction of the mechanical compression may determine surgical strategy. If CVJ deformity is reducible, posterior in situ fixation may be a viable solution. If the deformity is rigid and the C1ŌĆō2 facet is fixed, osteotomy may be necessary to make the C1ŌĆō2 facet joint reducible. C1ŌĆō2 facet release with vertical reduction technique could be useful, especially when the C1ŌĆō2 facet joint is the primary pathology of CVJ kyphotic deformity or basilar invagination. The indications for transoral surgery are becoming as narrow as a treatment for CVJ deformity. In this article, we will discuss CVJ alignment and various strategies for the management of CVJ deformity and possible ways to prevent complications and improve surgical outcomes.
Craniovertebral junction (CVJ) is the region around the skull base and the upper cervical spine (atlas and axis), and its alignment is unique regarding the segmental motion and biomechanical perspective [1-4]. The stability of CVJ is dependent on a robust ligamentous complex and the shape of the bony structures, which are also responsible for much of the axial rotation (C1ŌĆō2 joint) and flexion-extension movements (C0ŌĆō1 and C1ŌĆō2 joint) [5-7].
Although deformity or malalignment around CVJ is a rare, CVJ deformity results in sagittal and coronal imbalances, which causes significant pain due to arthritis, instability, and C2 foraminal stenosis. Moreover, malalignment can be a potentially life-threatening due to swallowing difficulty and cervicomedullary compressive myelopathy.
Patients presenting intractable pain, functional disability, or myelopathy caused by the CVJ deformity usually require surgical treatment. Decompression of neural structures, sagittal and coronal spinal realignment, and stabilization are the primary goals of surgical intervention.
Surgical management of CVJ pathology could be complicated for various reasons; given the discreet relationships involved in the surrounding neurovascular structures and intricate biomechanical issues, access to this region is relatively tricky, and there are many risks involved [8-14].
Recently, there have been tremendous advances in CVJ surgery. Understanding normal alignment, segmental motion, compensatory reaction, and anatomical variation around CVJ is critical not only to decide optimal surgical options but also to prevent complications and improve surgical outcomes.
CVJ deformity has various etiologies, such as congenital anomaly, tumor, infection, rheumatoid arthritis (RA), and trauma [15]. One of the most common CVJ anomalies is basilar invagination (BI), either congenital or degenerative, as represented by the upward migration of the odontoid process into an already limited space of the foramen magnum, causing compressive myelopathy around the cervicomedullary junction [16,17]. Many bony anomalies are associated with BI, such as clivus and condyle hypoplasia, occipital assimilation, os odontoideum, bifid C1 arch, and Klippel-Feil syndrome [18,19].
RA is the leading cause of secondary BI, which is also called basilar impression, atlantoaxial impaction, vertical settling, and cranial settling [20]. Symmetrical rheumatoid destruction of the C0ŌĆō1 and C1ŌĆō2 joints allow for the cranium to settle on the cervical spine and for the dens to enter the foramen magnum. However, only one lateral mass involvement may result in a fixed rotational tilt of the head. Secondary BI often results in a reduction of atlantodental interval (ADI) due to the conical shape of dens and reduced motion, producing a false impression of anatomic improvement known as ŌĆ£pseudostabilizationŌĆØ [21].
The human cervical spine is located inferior to the foramen magnum. These characteristics ensure an energy-saving balance of the head and neck when at rest, allowing humans to effectively maintain horizontal vision without the significant effort of the neck extensor muscle [22]. However, quadrupeds with posteriorly positioned foramina magna require well-developed nuchal musculature and ligaments to bear the headŌĆÖs weight to maintain horizontal gaze. When humans are at rest in standing position, the C0ŌĆō1 articulation is held rather flexibly, whereas the C1ŌĆō2 joints are held slightly in an extended position.
So, the C1 slope is backwardly slanted in the neutral position, and kyphotic angulation of the C0ŌĆō1 segment allows some degree of freedom for neck extension as the space between the occiput and C1 posterior arch allows for upper cervical extension (Fig. 1A).
When patients have a kyphotic deformity in the lower cervical spine or thoracolumbar spine, the C0ŌĆō1 segment can hold up the head to compensate for distal kyphosis, maintaining the sagittal balance and horizontal gaze. Therefore, a normal kyphotic angulation of the C0ŌĆō1 segment could be referred to as a ŌĆ£tertiary curvatureŌĆØ to characterize human CVJ alignment, to differentiate between upper cervical spine alignment and primary/secondary curvature of the human spine as well as differentiate between the cervical spine curvature of humans and that of quadrupeds.
Although the asymptomatic population may have different types of cervical alignment, such as ŌĆ£straightŌĆØ or ŌĆ£kyphoticŌĆØ cervical spines ranging from 7% to 40%, it has been well accepted that cervical lordosis is a physiological posture in asymptomatic subjects, and a large percentage (about 75%ŌĆō80%) of cervical lordosis is localized to the C1ŌĆō2 segment. Only a small percentage (15%) of cervical lordosis exists in the lower cervical levels. On average, the C0-1 segment is kyphotic [23-28]. The loss of subaxial lordosis has been reported in CVJ fixation in which excessive hyperlordosis is created at the C0ŌĆō2 segment (Fig. 1B) [29]. Moreover, craniometrics studies revealed that excessive CVJ kyphosis could cause the subaxial compensatory lordosis (Fig. 1C) [30-32]. The cervicothoracic junction has been shown to be the main determining factor for cervical lordosis. However, the CVJ angle has also been shown to be related to the subaxial cervical spineŌĆÖs angle significantly.
The OŌĆōC2 angle was significantly greater in the patients with a straight or kyphotic subaxial cervical spine than those with lordotic lower cervical spine. When patients have a kyphotic deformity in the lower cervical spine, positive sagittal imbalance occurs in the cervical spine, and the hyperextended C0ŌĆō2 segment holds up the head to compensate for distal kyphosis, sagittal imbalance, and maintaining horizontal gaze.
In the patients with CVJ kyphotic deformity, lateral radiographs show hyperlordotic compensation in the subaxial cervical spine and negative sagittal imbalance to maintain gaze (Fig. 1B, C).
These findings suggest that reciprocal interaction may likely affect not only global balance but also regional balance [33].
Another interesting finding is that neck flexion, and extension movement is initiated primarily by motions between the head and C1 [34]. As the head approaches full flexion and extension, contributions from the upper cervical segments decrease, whereas the lower cervical segmentsŌĆÖ contribution increases.
Understanding the normal CVJ alignment and segmental motion is paramount to understand CVJ deformity better and to decide on a more appropriate surgical strategy according to the pathologies of CVJ.
Irreducibility was defined as nonalignment of C1ŌĆō2 after neck extension (determined on lateral x-ray) or after cervical traction. Lateral radiography with a dynamic study is necessary to evaluate ADI, degree of vertical subluxation, and reducibility. Furthermore, bony abnormalities, such as occipital assimilation, os odontoideum, bifid C1 arch, and C2ŌĆō3 fusion, can be evaluated on lateral radiography.
Traction can be a useful tool in evaluating the reducibility and predicting intraoperative neurological worsening by surgical position. Preoperative cervical traction may correct the atlantoaxial dislocation (AAD) and vertical subluxation in some reducible deformity cases. This could especially be important for managing fragile patients because they can be treated with only a stabilization procedure postreduction, reducing the level of fixation. Gardner cervical traction was applied, depending on age and weight, starting with 2ŌĆō5 kg for one or 2 days. The head of the bed was elevated to provide counter traction. Serial radiographs were assessed for reduction. These patients who demonstrated a reduction were classified as reducible AAD or BI.
Magnetic resonance imaging (MRI) was studied to evaluate cord compression, T2 signal change, Chiari malformation, and syringomyelia. Computed tomography (CT) scans can confirm bony abnormalities, the densŌĆÖ exact location, C1ŌĆō2 joint destruction, and abnormal C1ŌĆō2 facet angle. CT scans are also used to determine the extent of facet fusion and osteophytic bridging to assess the need for C1ŌĆō2 osteotomy.
Computed tomography angiography (CTA) screening is a useful tool for getting 3-dimensional complex deformity information and identifying vertebral artery (VA) anomaly around CVJ [14,35]. The incidence of V3 segment anomalies has been reported to be as high as 10% [9,14]. In V3 segment anomalies, a more optimal entry point for C1 fixation should be selected to avoid significant morbidities associated with VA injury [36]. Although there could be a racial difference in the incidence of V3 segment anomaly, existing literature indicates that the V3 segment anomaly is more common in the group with the congenital bony anomaly. Quadrupeds, including humans, are more likely to have other CVJ bony abnormalities, such as atlantooccipital assimilation or dens anomalies, if atlantoaxial instability is present.
Therefore, we suggest that preoperative CTA might be informative for deciding on a surgical technique, especially in the high-risk group of the V3 segment anomaly, such as Asians and those with C1ŌĆō2 instability with congenital bony anomaly [9].
Fig. 2 provides decision-making pathways to treat CVJ deformity. The treatment goal of CVJ deformity is to restore normal alignment, relieve neural compression, and stabilization without neurological complications. Factors that influence the specific treatment of CVJ deformity are as follows: (1) the reducibility of deformity (i.e., restoring anatomic alignment, thereby relieving neural compression); (2) the direction of mechanical compression; (3) the presence of abnormal craniocervical angle and alignment; and (4) the presence of hindbrain herniation, syrinx, and vascular abnormalities.
Irreducible lesions can be divided into 2 types: The ventral compressive lesion and the dorsal compressive lesion. Doral compression cases need either a posterior or posterolateral decompression procedure, and if there is instability after decompression, posterior fixation is mandatory to gain stability. Irreducible CVJ deformity may require decompression at the site of compression or may require an osteotomy procedure to make the lesion reducible and relieve neural compression [33].
Transoral surgery is a procedure carried out through the oral cavity to gain access to the ventral compressive lesion of CVJ from the lower rim of clivus cranially to C23 disc space caudally. Transoral surgery could be necessary in case of irreducible CVJ deformity, where ventral compression to neutral structures is present [37]. Other indications for transoral surgery include CVJ tumor and infection, in which a simple posterior reduction [38] cannot decompress the anterior compression. Although the combined anteriorposterior approach may provide adequate neural decompression and fusion, it poses several disadvantages, such as the higher risk of surgical morbidities, prolonged postoperative intubation/nasogastric feeding, phonation difficulty, and potential infection [39-41]. The risk of infection is a big concern, mainly when the subarachnoid space had to be opened, which may cause catastrophic sequelae. Another issue is that transoral decompression can induce significant instability of CVJ, inevitably requiring a subsequent posterior stabilization procedure. These issues are why the initial enthusiasm vanished, and an indication of transoral decompression became less evident in the recent literature.
A C1ŌĆō2 joint distraction technique, as introduced by Goel, has been gaining popularity recently as a possible treatment modality for selected cases of AAD and BI [42-45]. This technique seems to have several advantages over conventional transoral surgery and occipitocervical (OC) fixation. First, direct posterior reduction and fixation are possible for cervicomedullary decompression. Hence, a transoral surgery and its related complications could be avoided. Second, the fusion bed dimension could substantially be increased because C1ŌĆō2 facet surface is rather large. Third, it can avoid occipital fixation and conserve C0ŌĆō1 segmental motion. Fourth, C1ŌĆō2 joint manipulation can make a fixed subluxation or deformity reducible, minimizing the need for head traction, before and during surgery.
Preoperative cervical traction may correct AAD and vertical subluxation in some patients with reducible deformity. This could especially be important for managing fragile patients because they can be treated with only a stabilization procedure after reduction and decreasing the numbers of fixation levels.
The direction of the mechanical compression was the critical factor in deciding a surgical approach. With recent advancements in surgical techniques and instruments, the results of posterior C1ŌĆō2 release and reduction have been satisfactory. However, difficult or insufficient reduction and loss of reduction have been observed after posterior reduction and fixation [37]. Thus, anterior release reduction and posterior fixation could still be available options for irreducible BI with AAD, especially in patients with severely deformed bony mass anteriorly compressing the cervicomedullary junction.
CVJ alignment is another crucial factor to decide surgical strategy. Clivus-canal angle and cervicomedullary angle is tended to be kyphotic in congenital CVJ anomaly, which is caused by the short clivus, platybasia, angulated odontoid process, or abnormal sagittal inclination of deformed C1ŌĆō2 facet joints [30-32]. Craniocervical realignment through screws and rod systems may be an efficacious surgical technique for treating congenital craniocervical anomaly with abnormal angulation of the clivus. However, craniocervical realignment procedure, including distraction, compression, and skull extension, need additional decompression/fixation up to the cranium, and it should be cautiously performed, especially in cases with severe canal stenosis [46].
Reducible AAD can be defined as C1ŌĆō2 alignment on extension or cervical traction. If the dislocation cannot be reduced on extension position and preoperative traction, it is labeled as an irreducible or fixed AAD and rotatory subluxation (Fig. 3). Management becomes difficult with this type of fixed deformity. Since Goel introduced the C1ŌĆō2 distraction method, the focus in CVJ pathology treatment has shifted to the dislocated C1ŌĆō2 facets rather than the C2 dens, and the C1ŌĆō2 joint manipulation has been the preferred method of treatment for irreducible AAD [21,43]. It is because the cause of irreducibility is in the C1ŌĆō2 facets. In cases of irreducible AAD or rotatory fixation, C1ŌĆō2 joint mutilation or osteophyte formation is so severe that dislocation cannot be reduced despite cervical traction. Therefore, C1ŌĆō2 facet mobilization is necessary to make this fixed deformity reducible using facet osteotomies (Fig. 3C).
After releasing the C1ŌĆō2 facet joint capsule and removing osteophytes, the mobility between C1 and C2 is usually identifiable intraoperatively. The cartilage endplate was curetted and drilled to increase mobility further and remodel the C1ŌĆō2 facet joint plane. The rotated C1ŌĆō2 facet joint could be mobilized and reduced in a neutral position after releasing the C1ŌĆō2 facet joint (Supplementary video clip 1).
In the horizontal subluxation cases, the surgical assistant pressed down firmly on the C2 spinous process or pulled the C1 arch using cables to reduce horizontal subluxation. Adjusting the patientsŌĆÖ neck to allow for a slight extension should help the process of reduction. Bone block or cage filled with cancellous bone can be placed inside the decorticated C1ŌĆō2 joint.
rheumatoid BI is caused by C1ŌĆō2 joint destruction and subsequent upward migration of the odontoid process. These findings suggest that the primary pathology of rheumatoid BI is the C1ŌĆō2 joint, not the C0ŌĆō1 joint. Hence, spine surgeons should focus on C1ŌĆō2 joint reduction to treat rheumatoid BI to spare the innocent C0ŌĆō1 segmental motion. Rheumatoid destruction of the C1ŌĆō2 joint could easily be confused with spontaneous fusion mass on CT scan due to narrowing the joint space and destruction of joint cartilage. However, the C1ŌĆō2 joint of rheumatoid BI is unstable and easily reducible in vertical distraction.
The posterior edge of the foramen magnum, posterior arch of C1 and the C2ŌĆō3 lamina were exposed through a midline skin incision. For patients with posterior compressive lesions around the foramen magnum, the posterior margin of the foramen magnum and C1 arch were removed. Intentional sacrifice of bilateral C2 ganglions was performed to obtain enough C1 screw entry point, joint manipulation, and bone fusion (Fig. 4). Subsequently, by applying the Harms technique, polyaxial screws were inserted to the bilateral C1 lateral masses and the C2 pedicle. The C1 and C2 screws were partially loosened. Distraction between the C1 and C2 screws was performed using a distractor to achieve vertical reduction; the nuts were subsequently locked. Vertical reduction results in the migration of the odontoid process inferiorly, reducing the neural compression around the ventral cervicomedullary junction. The C1ŌĆō2 articular cartilages were removed widely using a curette and a high-speed burr. The facet cage or tricortical iliac bone autograft was inserted bilaterally as joint spacers to maintain the odontoid process vertically. Then compression between the C1 and C2 screw head increases the graft contact inside the C1ŌĆō2 joint, creating adequate C1ŌĆō2 lordosis. Subsequently, interlaminar fusion could be performed using autograft iliac bone blocks and cables.
A substantial number of rheumatoid BI cases involve the spinal cordŌĆÖs compression ventrally by the retroodontoid pannus, which needs transoral decompression. However, posterior atlantoaxial fixation with vertical reduction technique has been reported to provide an early reduction of retroodontoid rheumatoid pannus, as well as a reduction of BI and AAD [47-51]. This finding suggests that rheumatoid pannus could be a buckling of the ligamentous complex around CVJ caused by BI rather than granulation tissue caused by the inflammation.
Congenital BI is usually irreducible. Irreducibility results from the combined bony malformation and the abnormal inclination of the C1ŌĆō2 facet joints.
In C1ŌĆō2 subluxation with oblique C1ŌĆō2 facet joint orientation, the joint space is not visible even after C2 root resection. The drilling of the superior posterior corner of the C2 facet allows the C1ŌĆō2 joint space visibility. Then the small osteotome could be inserted into the joint space to open it. Then drilling, reaming, and curettage was continued to release the fixed C1ŌĆō2 joint space. Drilling and osteotome were focused on the inferior anterior corner of the C1 facet and the superior posterior aspect of the C2 facet to make the C1ŌĆō2 joint flat. Osteotome was used as a fulcrum to open the C1ŌĆō2 joint space and reduce the subluxation both for the vertical and horizontal translation. The reduction was then maintained using the C1ŌĆō2 spacers (bone block or PEEK cage filled with bone), and C1ŌĆō2 was fixed using the polyaxial screw system. In those who needed C1 laminectomy for the posterior decompression, C1 laminectomy bone could also be used as the C1ŌĆō2 facet spacer.
If BI and CVJ kyphosis are not severe, C1ŌĆō2 facet joint distraction and C1ŌĆō2 joint remodeling may be sufficient to reduce and decompress the cervicomedullary compression (Fig. 5). However, in cases with a severe degree of CVJ kyphotic angulation and ventral cord compression, C1ŌĆō2 joint vertical reduction and fixation may not be sufficient to obtain good CVJ alignment and decompression. Therefore, occipital fixation, distraction, compression, and head extension are mandatory to obtain a craniocervical realignment in CVJ kyphotic deformity (Fig. 6).
After placing the screw for occiput, C1 lateral mass, and C2 pedicle, C2 screw heads were tightened to ensure that the screws, C2 body, and the odontoid process became an integrated unit. The C1 and occipital screw heads were not tightened yet. The C1 screw placement was not mandatory in cases with occipital assimilation and in cases where the space was not sufficient for C1 screw placement or rod application. The rod bent more than the patientŌĆÖs CVJ alignment, and the head was extended using the Mayfield head holder to approximate the rod to the occipital screw head. The occipital screw and C1 screw head were only tightened loosely at this stage. Then, the longitudinal distraction between the screws, which pulled the odontoid process downward and anteriorly. Once the joint space was distracted, facet cages or bone blocks were placed bilaterally, and the occipital screws and C1 screws were tightened after head extension using the Mayfield skull pin, which reduced the AAD and CVJ kyphosis. For patients who need more horizontal reduction for AAD and forward movement of the odontoid process, the C2 pedicle screws were loosened, and vertical compression was performed between C1ŌĆō2 screw heads after placement of facet spacers. Excessive vertical reduction and CVJ realignment may pose a higher risk of postoperative neurological impairment. Therefore, the C1-2 distraction and craniocervical realignment should be performed very carefully under intraoperative neuromonitoring (IONM) to avoid excessive distraction and subsequent stretching injury of the spinal cord or VA. Although we do not fully know the amount of distraction required for neurological recovery and prevention of neurological complications, our published data showed that the change rate of Modified Ranawat index after surgery was correlated with postoperative Hirabayashi recovery rate [52]. It is impossible to simplify clinical outcomes into a linear curve. However, the amount of radiological change has a substantial correlation with the clinical outcomes. After satisfactory reduction and confirmation were provided by fluoroscopy, the posterior bony surface was decorticated to prepare for the fusion bed. The bone grafts were placed over the decorticated fusion surface.
Mechanical failure, various kinds of neurovascular complications, and postoperative swallowing difficulty can occur after the surgery in patients with CVJ deformity.
Despite recent advancements in the posterior cervical instrument, high biomechanical loading around this particular junctional area poses a risk for instrument failure or nonunion [10,12]. Fusion bed preparation and graft material selection are essential for increasing the fusion rate. We usually use iliac autograft as much as possible. Although the posterior surface of the occipital bone and C1ŌĆō2 interlaminar space is a popular fusion bed, the C1ŌĆō2 facet joint is the most critical area for fusion and malalignment correction for CVJ kyphosis patients, especially when C1 laminectomy is inevitable.
Moreover, the long-term fate of the adjacent C0ŌĆō1 segment has rarely been reported in patients after receiving C1ŌĆō2 vertical distraction procedure with respect to the segmental motion and adjacent segmental degeneration. Yoshida et al. [53] reported that C1ŌĆō2 rigid fixation could provide prophylactic benefits against vertical subluxation and the subaxial subluxation in RA patients. Werle et al. [54] reported that C0ŌĆō1 degeneration is very low after C1ŌĆō2 fusion in RA patients. Therefore, the C0ŌĆō1 segment sparing technique should be the preferred treatment option of CVJ fusion, especially for RA patients.
Several studies have investigated the relationship between postoperative C1ŌĆō2 angle and C2ŌĆō7 sagittal alignment change after C1ŌĆō2 fixation [55-58], and most have reported a reciprocal relationship between changes in the upper cervical spine and subaxial cervical spine [57,58]. If the CVJ is fixed in the hyperlordotic posture, subaxial cervical kyphosis is inevitable, and upper cervical kyphosis causes lordotic compensation in the subaxial cervical spine. The most excellent care is necessary to determine the C1ŌĆō2 fixation angle during surgery to reduce the risk of postoperative malalignment and subluxation of the subaxial cervical spine. Subaxial alignment change is not uncommon after CVJ fixation. Muscle detachment at the C2 spinous process was not a risk factor of kyphotic change. Our findings suggest small range of motion (ROM) of the C0ŌĆō1 segment with or without occipital fixation and combined subaxial laminoplasty are risk factors of subaxial kyphotic change. Although the little study has sought to identify the risk factors of postoperative subaxial malalignment following CVJ fixation, we recently reported subaxial cervical spine alignment might change significantly during the first year after CVJ posterior fixation. The subaxial cervical alignment is significantly related to the upper cervical angle after CVJ fixation. Furthermore, the study suggested a small ROM of the C0ŌĆō1 segment with or without occipital fixation and combined subaxial laminoplasty independently predict subaxial kyphotic change after upper cervical fixation. Interestingly, extensor muscle detachment at C2 itself was not identified as a risk factor of kyphotic change, but when combined with subaxial laminoplasty and wider dissection of deep extensor muscle, the risk of subaxial kyphotic change was significantly increased [59].
These results suggest the C0ŌĆō1 segment is the dominant segment for compensating for abnormal angulation of the subaxial cervical spine after upper cervical fixation. These results imply it is important not to include occipital fixation during upper cervical fixation to avoid loss of subaxial cervical lordosis and preserve C0ŌĆō1 segmental motion.
C1ŌĆō2 joint distraction technique has a clinically significant concern regarding the C2 neuropathic pain after C2 root resection. Although C2 root resection is not mandatory for C1ŌĆō2 facet opening, it is useful to make a broad exposure for the C12 facet surface, especially when C1ŌĆō2 interlaminar space is narrow and venous plexus is prominent [44,60]. Thus, C2 root resection is one of the critical parts of the C1ŌĆō2 joint distraction procedure. In the literature, most authors have reported that C2 neuropathic pain incidence is relatively low after C2 root resection [44,60]. However, C2 neuropathic pain could be troublesome for some patients. Therefore, surgeons should be aware of both the benefits and risks associated with C2 root resection. C2 neuropathic pain may cause significant postoperative occipital neuralgia, which could also occur due to the C2 root irritation by C1 screw, excessive manipulation, and traction on the C2 root during surgery. Several reports have advocated the deliberate C2 root resection or C1 screw without threads to minimize C2 root complications [61-63]. However, the relationship between C2 root resection and neuropathic pain in the literature remains controversial. Goel et al. [61] reported no significant concerns regarding C2 neuropathic pain among 160 patients who underwent the C2 root sacrifice. Dewan et al. [64] reported that C2 root transection is associated with increased occipital numbness. However, this finding does not affect patient-reported outcomes and quality of life, and the C2 root preservation can be associated with occipital neuralgia, which harms patient disability and quality of life. Elliott et al. [65] demonstrated that the C2 root transection decreases blood loss and operative time and significantly reduces postoperative neuralgia. However, Yeom et al. [66] reported that C2 root resection could cause a high incidence of postoperative occipital neuralgia. In our study, occipital neuralgia was significantly induced in the C2 root resection group than that of the C2 root-sparing group.
Furthermore, the C1 screw entry point is significantly related to postoperative C2 neuropathy. Although C2 root resection allows for better visualization for venous bleeding control, getting a broader view for screw insertion to prevent screw malposition and a wider bone fusion surface, C2 root resection is an independent risk factor of postoperative C2 root neuropathy. So, excessive C2 root manipulation and resection should be carefully determined because it can cause occipital neuralgia. However, C2 root resection could be inevitable for vertical reduction, especially in cases of rheumatoid BI. Given that C1ŌĆō2 joint mutilation is so severe in rheumatoid BI, the height of the C2 foramen becomes too narrow for the manipulation of the C1ŌĆō2 facet joint and insertion of screw without damaging the C2 nerve root.
Even though several reports of vertical reduction technique showed excellent clinical and radiological results without neurological complication [21,37,43,44], we experienced cases with transient neurological worsening after craniocervical realignment and encountered several cases with a significant change in IONM during the vertical reduction technique [67]. To date, we are not sure whether it is related to spinal cord stretch or spinal cord ischemia due to the VA stretch. However, we found that IONM change is usually recovered after reducing the amount of C1ŌĆō2 joint distraction and the facet graft size. This finding shows the efficacy of IONM during craniocervical realignment or vertical reduction procedure for congenital craniocervical segmentation anomaly.
IONM change is more common in CVJ surgery than at any other level of the cervical spine. Preoperative T2 signal change on MRI and narrow canal diameter are 2 risk factors that are significantly related to IONM change in our CVJ surgery case series [46,67,68]. We suggest that IONM may help in preventing unexpected neurological deterioration during posterior craniocervical distraction and realignment.
A ŌĆ£military tuckŌĆØ posture (neutral head posture, the extension of the lower cervical spine, posterior translation of the occiput-C1 complex) usually reduces C1ŌĆō2 subluxation, while optimizing the surgical exposure and allowing for a favorable screw trajectory [69]. It is a commonly used posture for posterior instrumentation in the upper cervical spine.
However, previous studies have shown that a decrease in the OC2 angle in the retraction position causes a reduction in the oropharyngeal airway space and postoperative dysphagia [70,71].
Dysphagia is not an uncommon complication after posterior cervical surgeries, as well as anterior cervical spine surgeries [72-74]. Moreover, dysphagia after posterior OC fusion has been recognized as a severe postoperative complication. Miyata et al. [70] reported that dysphagia after OC fusion is caused by glossoptosis, which is the downward displacement of the tongue root, resulting in the narrowing of the oropharynx and the impairment of the ability of the epiglottis to move sufficiently. They emphasized that a decrease in the OC2 angle may likely induce a reduction in the pharyngeal space and can be a predictor of postoperative dysphagia, which is not compensated by the middle or lower cervical spine. We reported that the upper cervical segmentsŌĆÖ movement could be an essential compensation mechanism for swallowing, and the fixation of the upper cervical spine could be one of the risk factors for postoperative dysphagia.
Moreover, C0ŌĆō1 angle change and movement during swallowing are significantly greater in the retraction posture than in the neutral posture, and the C0ŌĆō1 segment is the dominant level of compensatory motion that occurs when subjects experience dysphagia in the retraction position [75]. Therefore, if we fix up to the cranium in the retraction position, the compensatory movementŌĆÖs major segment cannot function properly to allow swallowing, leading to postoperative dysphagia [76]. Therefore, avoiding OC fixation in retraction posture and sparing C0ŌĆō1 segment are critical technical tips for preventing postoperative dysphagia.
Each type of CVJ deformity has its characteristics and treatment challenges. Regardless of the underlying cause, this complicated disease entity has 2 typesŌĆöreducible deformity and irreducible deformityŌĆöaccording to whether the dislocation is reduced after dynamic x-ray and traction. Simple posterior fixation and fusion are adequate for reducible CVJ deformity, and any type of reduction procedure is necessary for irreducible CVJ deformity. A reduction of irreducible deformity can be achieved using anterior transoral surgery or posterior C1ŌĆō2 facet joint manipulation procedure, depending on the pathologyŌĆÖs primary focus. With recent advancements in surgical techniques and internal fixation devices, posterior only surgery can achieve ventral decompression and craniocervical realignment. The posterior reduction procedure principle is releasing the C1ŌĆō2 facet joint, pulls down the odontoid process away from the cervicomedullary junction, and craniocervical realignment without neurological injury. C1ŌĆō2 vertical reduction technique can be the optimal surgery for BI with or without abnormal CVJ kyphosis. Additional procedure for craniocervical realignment is necessary to treat congenital BI with severe CVJ kyphosis. Head extension maneuver through the occipital screws and rod system may be a safe and efficacious surgical technique for the craniocervical realignment.
Avoiding neurological complications could be the most crucial issue in this complicated junctional area, and preoperative radiological assessment is critical to detect the structural anomaly and decide an optimal surgical plan. IONM may help in preventing any unexpected neurological deterioration during posterior craniocervical distraction and realignment.
By understanding the nature of CVJ alignment, recent advancements in posterior cervical instrumentation, all available treatment options, and the decision-making process, surgeons can improve patientsŌĆÖ life quality.
SUPPLEMENTARY MATERIALS
Supplementary video clip 1 can be found via https://doi.org/10.14245/ns.2040510.255.v1.
Fig.┬Ā1.
Lateral x-ray shows a normal alignment of the cervical spine. On most occasions, cervical lordosis occurs at the C1ŌĆō2 segment. (A) C0ŌĆō1 segment is kyphotic, and the C1 slope is posteriorly slanted. (B) C0ŌĆō2 segment is a hyperlordotic, and the C1 slope is reversely slanted to maintain the patientŌĆÖs horizontal gaze with sagittal malalignment and subaxial cervical kyphotic deformity patient. (C) Craniovertebral junction kyphotic deformity patient shows a hyperlordotic compensation in the subaxial cervical spine and negative sagittal imbalance to maintain gaze.
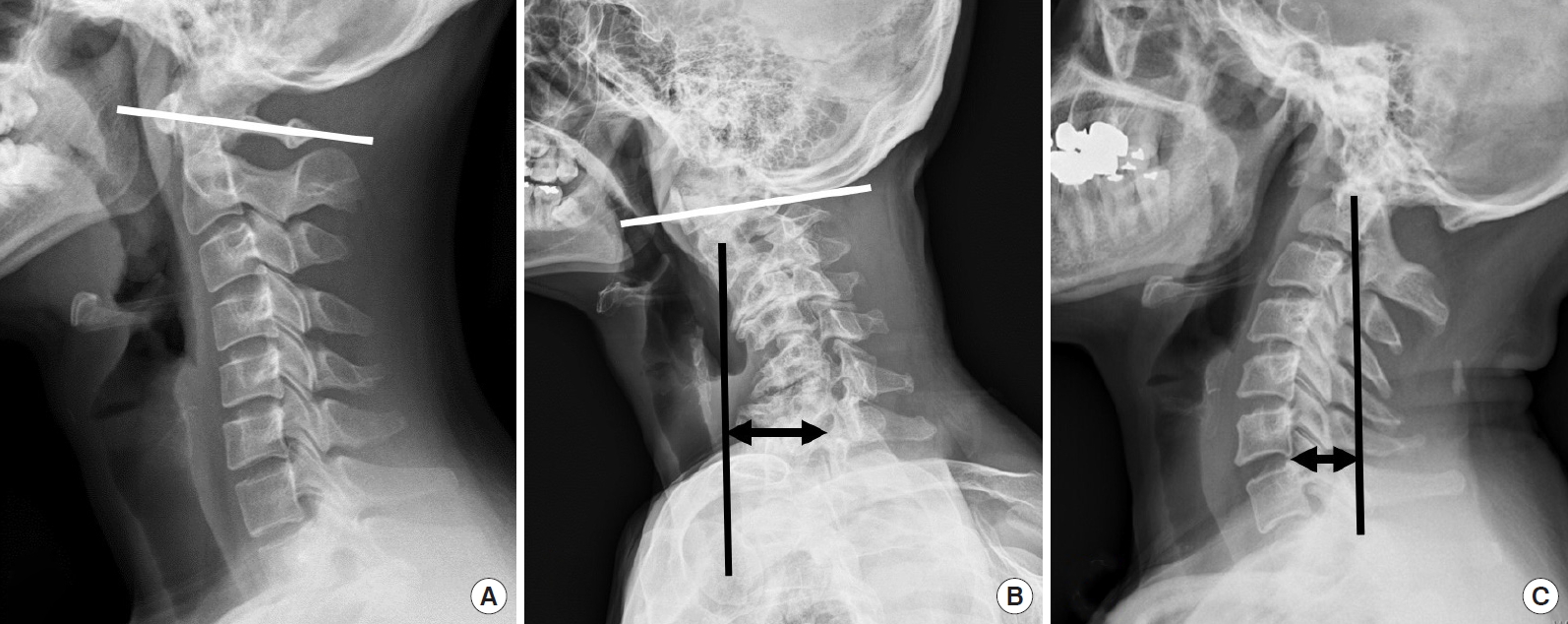
Fig.┬Ā2.
The algorithm shows decision-making pathways for the treatment of the craniovertebral junction (CVJ) deformity. OC, occipitocervical.
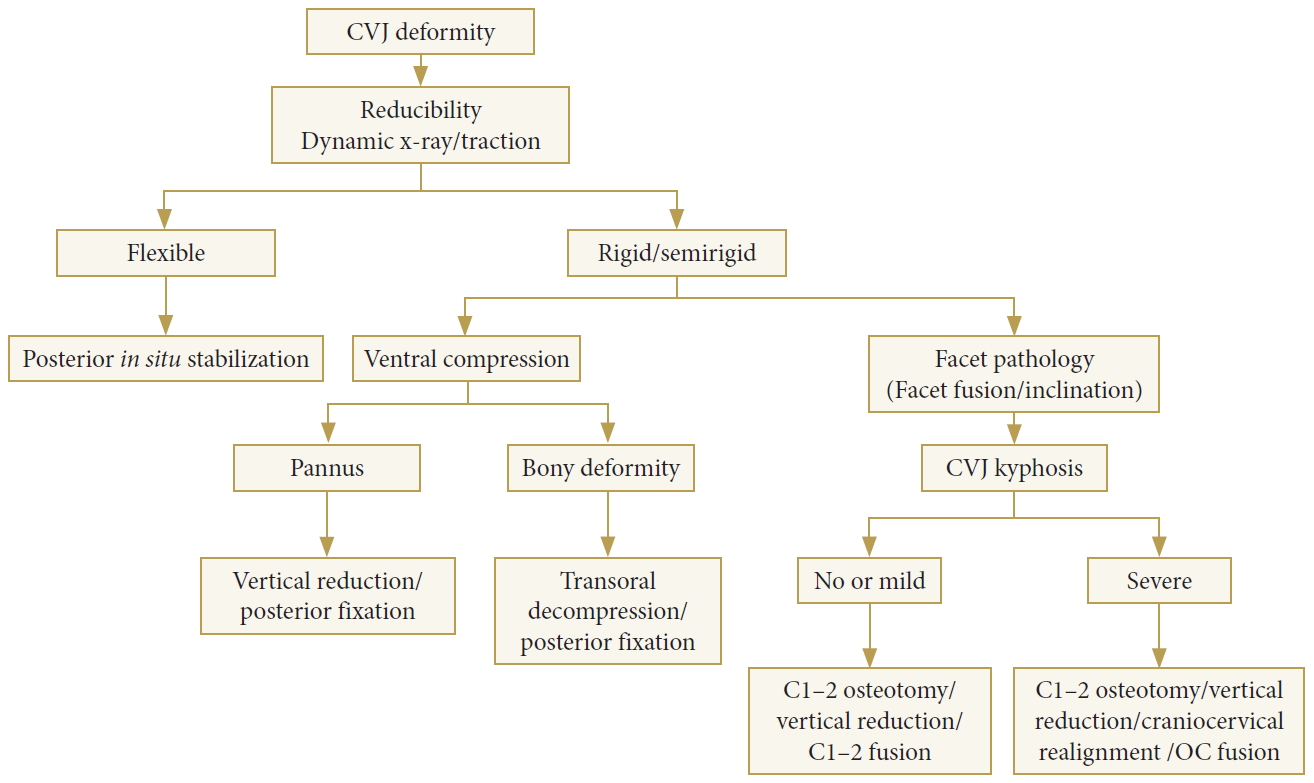
Fig.┬Ā3.
Illustrative case. Fixed C12 subluxation. (A) Coronal reconstructed computed tomography (CT) scan showing narrow C1ŌĆō2 facet joint space and joint mutilation on the right side. (B) Three-dimensional reconstructed CT scan showing lateral tilt and C1ŌĆō2 joint rotation. (C) Intraoperative photograph demonstrates small osteotomes inside the C1ŌĆō2 joint space (arrow) on the right side for joint release. C2 root resection provides panoramic view of C1ŌĆō2 facet joint. (D) Coronal reconstructed CT scan showing remodeling and reduction of the C1ŌĆō2 facet tilt on the right side.
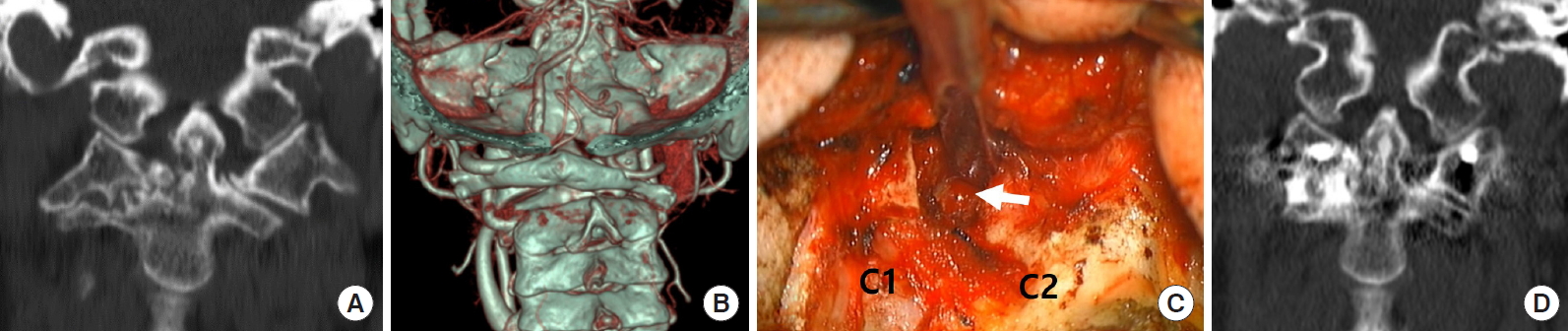
Fig.┬Ā4.
(A, B) Sagittal T2 magnetic resonance imaging (MRI) and sagittal reconstruction computed tomography (CT) image showing odontoid compression over the brain stem and upper cervical cord. Odontoid tip (white arrow) is located above the foramen magnum. (C) Parasagittal reconstruction CT scan showing C1ŌĆō2 facet subluxation and severe joint mutilation of the C0ŌĆō1 facet joint (arrow). (D) Postoperative x-ray showing the final construct of C0ŌĆō2 instrumentation. (E, F) Postoperative sagittal T2 MRI and sagittal reconstruction CT image showing early reduction of retroodontoid rheumatoid pannus (white arrow) and odontoid tip (black arrow) is pulled down below the foramen magnum. (G) Parasagittal reconstruction CT scan shows vertical distraction and intra-articular fusion (white arrow) inside the C1ŌĆō2 facet joints.
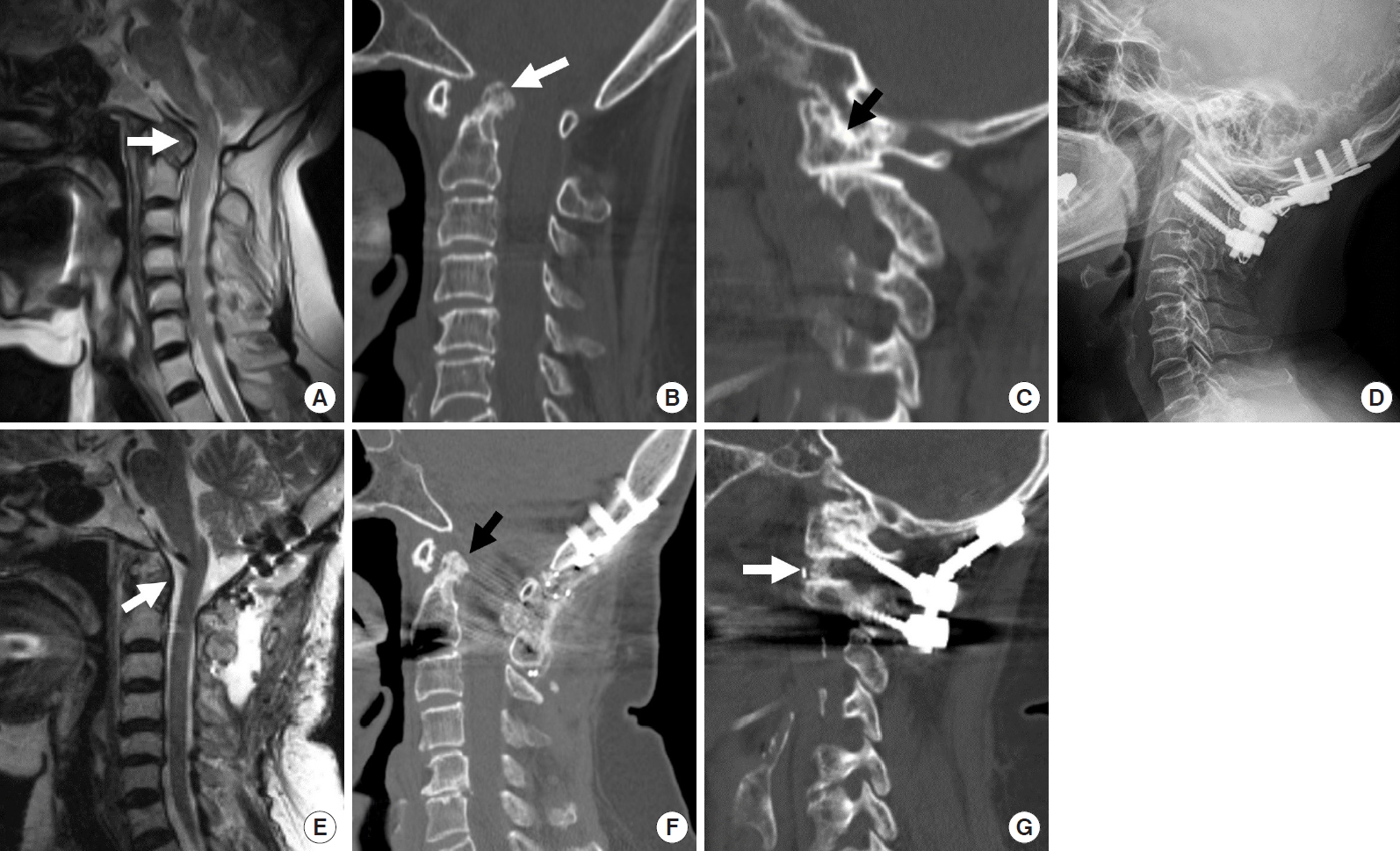
Fig.┬Ā5.
Illustrative case. Congenital craniovertebral junction kyphosis. (A) Preoperative x-ray shows the increased atlanto-dental interval. (B) Sagittal reconstructed computed tomography (CT) scan reveals the abnormal inclination of C1ŌĆō2 facet joint (arrow). (C) Sagittal T2-weighed image magnetic resonance imaging (MRI) showing os odontoideum and cord compression by the C2 dens over the upper cervical cord. (D) Postoperative x-ray shows the kyphosis reduction and facet grafts inside of C1ŌĆō2 joint (arrow). (E) Sagittal reconstructed CT scan showing remodeling of the C1ŌĆō2 facet joint and facet graft inside (arrow). (F) Postoperative MRI showing the decompression of the spinal cord and C2 realignment.
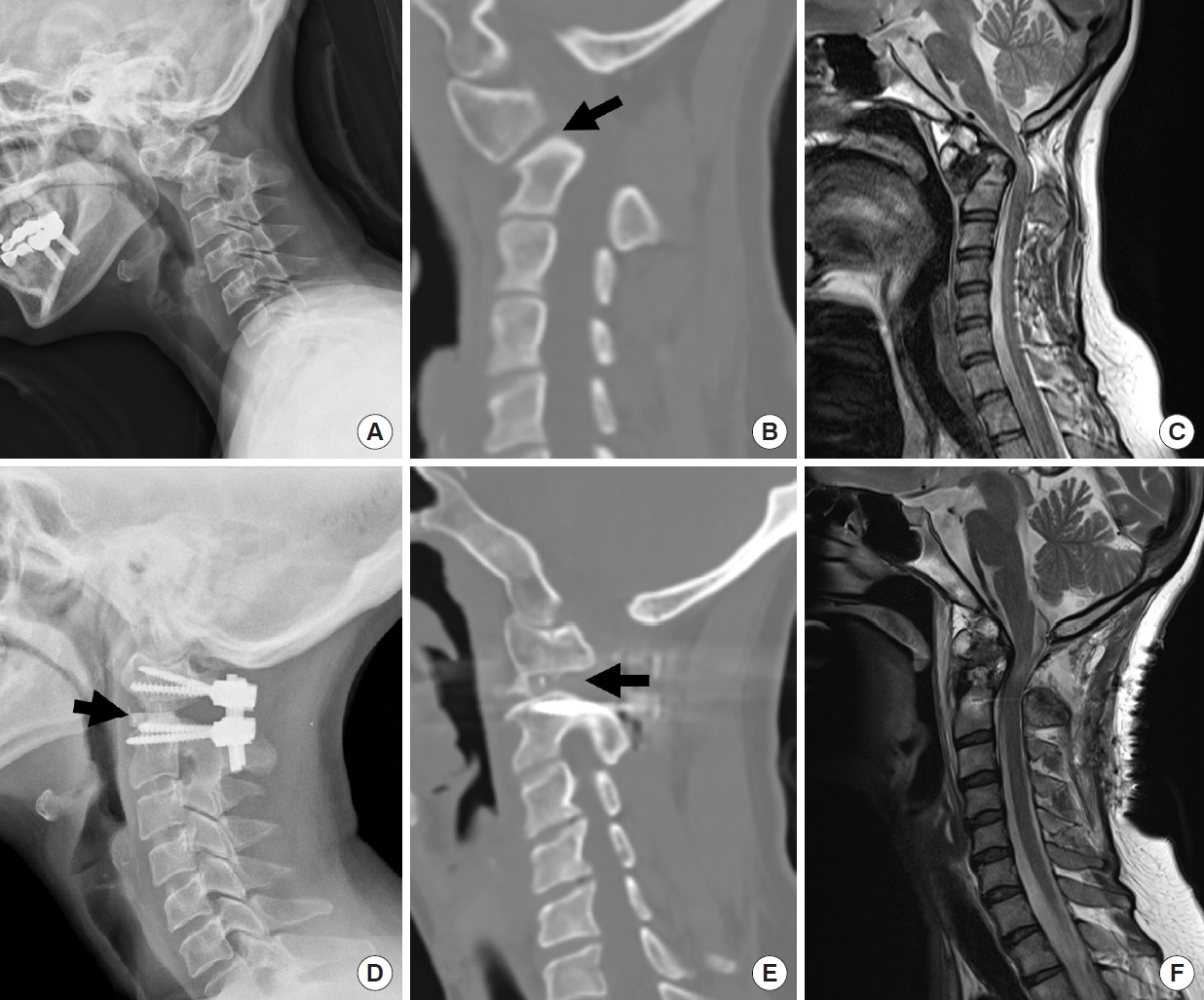
Fig.┬Ā6.
Illustrative case. Congenital basilar invagination and craniovertebral junction (CVJ) kyphosis. (A) Preoperative x-ray shows the atlantoaxial dislocation and severe kyphotic CVJ deformity caused by the os odontoideum. (B) Sagittal reconstructed computed tomography (CT) scan reveals the abnormal inclination and radiographical stiffness of C1ŌĆō2 facet joint (arrow). (C) Postoperative x-ray shows a reduction of C1ŌĆō2 dislocation. CVJ kyphosis is also reduced and clivus-canal angle is increased. (D) Sagittal reconstructed CT scan reveals postoperative remodeling and flattening of the C1ŌĆō2 facet joint and PEEK cage inside the joint (arrow).

REFERENCES
1. Visocchi M, Mattogno PP, Signorelli F, et al. Complications in craniovertebral junction instrumentation: hardware removal can be associated with long-lasting stability. Personal Experience. Acta Neurochir Suppl 2017 124:187-94.


3. Goel A, Sathe P, Shah A. Atlantoaxial fixation for basilar invagination without obvious atlantoaxial instability (group b basilar invagination): outcome analysis of 63 surgically treated cases. World Neurosurg 2017 99:164-70.


4. Lee DH, Hong JT, Sung JH, et al. Morphologic analysis of occipital sinuses for occipital screw fixation using digital subtraction angiography. World Neurosurg 2016 91:279-84.


5. Dlouhy BJ, Policeni BA, Menezes AH. Reduction of atlantoaxial dislocation prevented by pathological position of the transverse ligament in fixed, irreducible os odontoideum: operative illustrations and radiographic correlates in 41 patients. J Neurosurg Spine 2017 27:20-8.


6. Deepak AN, Salunke P, Sahoo SK, et al. Revisiting the differences between irreducible and reducible atlantoaxial dislocation in the era of direct posterior approach and C1-2 joint manipulation. J Neurosurg Spine 2017 26:331-40.


7. Yin YH, Tong HY, Qiao GY, et al. Posterior reduction of fixed atlantoaxial dislocation and basilar invagination by atlantoaxial facet joint release and fixation: a modified technique with 174 cases. Neurosurgery 2016 78:391. -400. discussion 400.



8. Yi HJ, Hong JT, Lee JB, et al. Analysis of risk factors for posterior C1 screw-related complication: a retrospective study of 358 posterior C1 screws. Oper Neurosurg (Hagerstown) 2019 17:509-17.



9. Hong JT, Kim IS, Kim JY, et al. Risk factor analysis and decision-making of surgical strategy for V3 segment anomaly: significance of preoperative CT angiography for posterior C1 instrumentation. Spine J 2016 16:1055-61.


10. Takigawa T, Simon P, Espinoza Orias AA, et al. Biomechanical comparison of occiput-C1-C2 fixation techniques: C0-C1 transarticular screw and direct occiput condyle screw. Spine (Phila Pa 1976) 2012 37:E696-701.


11. Hong JT, Takigawa T, Udayakunmar R, et al. Biomechanical effect of the C2 laminar decortication on the stability of C2 intralaminar screw construct and biomechanical comparison of C2 intralaminar screw and C2 pars screw. Neurosurgery 2011 69(1 Suppl Operative):ons1. -6. discussion ons6-7.



12. Hong JT, Takigawa T, Sugisaki K, et al. Biomechanical and morphometric evaluation of occipital condyle for occipitocervical segmental fixation. Neurol Med Chir (Tokyo) 2011 51:701-6.


13. Hong JT, Kim TH, Kim IS, et al. The effect of patient age on the internal carotid artery location around the atlas. J Neurosurg Spine 2010 12:613-8.


14. Hong JT, Lee SW, Son BC, et al. Analysis of anatomical variations of bone and vascular structures around the posterior atlantal arch using three-dimensional computed tomography angiography. J Neurosurg Spine 2008 8:230-6.


15. Kukreja S, Ambekar S, Sin AH, et al. Occipitocervical fusion surgery: review of operative techniques and results. J Neurol Surg B Skull Base 2015 76:331-9.




16. Salunke P, Sahoo S, Deepak AN. Different facets in management of congenital atlantoaxial dislocation and basilar invagination. Neurosurgery 2015 77:E985-7.



17. Salunke P, Sahoo SK, Deepak AN, et al. Comprehensive drilling of the C1-2 facets to achieve direct posterior reduction in irreducible atlantoaxial dislocation. J Neurosurg Spine 2015 23:294-302.


18. Goel A, Nadkarni T, Shah A, et al. Bifid anterior and posterior arches of atlas: surgical implication and analysis of 70 cases. Neurosurgery 2015 77:296. -305. discussion 305-6.



19. Debernardi A, D'Aliberti G, Talamonti G, et al. The craniovertebral junction area and the role of the ligaments and membranes. Neurosurgery 2015 76 Suppl 1:S22-32.



20. Krauss WE, Bledsoe JM, Clarke MJ, et al. Rheumatoid arthritis of the craniovertebral junction. Neurosurgery 2010 66:83-95.


21. Goel A, Pareikh S, Sharma P. Atlantoaxial joint distraction for treatment of basilar invagination secondary to rheumatoid arthritis. Neurol India 2005 53:238-40.


22. Hong JT. Human craniovertebral alignment as a ŌĆ£tertiary curvatureŌĆØ. Neurospine 2019 16:251-4.




23. Mizutani J, Verma K, Endo K, et al. Global spinal alignment in cervical kyphotic deformity: the importance of head position and thoracolumbar alignment in the compensatory mechanism. Neurosurgery 2018 82:686-94.



24. Karabag H, Iplikcioglu AC. The assessment of upright cervical spinal alignment using supine MRI studies. Clin Spine Surg 2017 30:E892-5.


25. Sharan AD, Krystal JD, Singla A, et al. Advances in the understanding of cervical spine deformity. Instr Course Lect 2015 64:417-26.

26. Joaquim AF, Riew KD. Management of cervical spine deformity after intradural tumor resection. Neurosurg Focus 2015 39:E13.

27. Kim HJ, Lenke LG, Oshima Y, et al. Cervical lordosis actually increases with aging and progressive degeneration in spinal deformity patients. Spine Deform 2014 2:410-4.


28. Scheer JK, Tang JA, Smith JS, et al. Cervical spine alignment, sagittal deformity, and clinical implications: a review. J Neurosurg Spine 2013 19:141-59.


29. Miyamoto H, Hashimoto K, Ikeda T, et al. Effect of correction surgery for cervical kyphosis on compensatory mechanisms in overall spinopelvic sagittal alignment. Eur Spine J 2017 26:2380-5.



30. Botelho RV, Ferreira ED. Angular craniometry in craniocervical junction malformation. Neurosurg Rev 2013 36:603. -10. discussion 10.




31. Ferreira JA, Botelho RV. The odontoid process invagination in normal subjects, Chiari malformation and Basilar invagination patients: Pathophysiologic correlations with angular craniometry. Surg Neurol Int 2015 6:118.



32. Xu S, Gong R. Clivodens angle: a new diagnostic method for basilar invagination at computed tomography. Spine (Phila Pa 1976) 2016 41:1365-71.


33. Chandra PS, Goyal N, Chauhan A, et al. The severity of basilar invagination and atlantoaxial dislocation correlates with sagittal joint inclination, coronal joint inclination, and craniocervical tilt: a description of new indexes for the craniovertebral junction. Neurosurgery 2014 10 Suppl 4:621. -9. discussion 9-30.



34. Anderst WJ, Donaldson WF 3rd, Lee JY, et al. Cervical motion segment contributions to head motion during flexion\ extension, lateral bending, and axial rotation. Spine J 2015 15:2538-43.


35. Song MS, Lee HJ, Kim JT, et al. Ponticulus posticus: morphometric analysis and its anatomical implications for occipito-cervical fusion. Clin Neurol Neurosurg 2017 157:76-81.


36. Hong JT, Jang WY, Kim IS, et al. Posterior C1 stabilization using superior lateral mass as an entry point in a case with vertebral artery anomaly: technical case report. Neurosurgery 2011 68:246. -9. discussion 9.



37. Liao Y, Pu L, Guo H, et al. Selection of surgical procedures for basilar invagination with atlantoaxial dislocation. Spine J 2016 16:1184-93.


38. Menezes AH, Traynelis VC, Gantz BJ. Surgical approaches to the craniovertebral junction. Clin Neurosurg 1994 41:187-203.

39. Menezes AH. Surgical approaches: postoperative care and complications ŌĆ£transoral-transpalatopharyngeal approach to the craniocervical junctionŌĆØ. Childs Nerv Syst 2008 24:1187-93.



40. Perrini P, Benedetto N, Di Lorenzo N. Transoral approach to extradural non-neoplastic lesions of the craniovertebral junction. Acta Neurochir (Wien) 2014 156:1231-6.



41. Jain VK, Behari S, Banerji D, et al. Transoral decompression for craniovertebral osseous anomalies: perioperative management dilemmas. Neurol India 1999 47:188-95.

42. Goel A, Phalke U, Cacciola F, et al. Surgical management of high cervical disc prolapse associated with basilar invagination--two case reports. Neurol Med Chir (Tokyo) 2004 44:142-5.


43. Goel A, Shah A. Atlantoaxial joint distraction as a treatment for basilar invagination: a report of an experience with 11 cases. Neurol India 2008 56:144-50.


44. Chandra PS, Prabhu M, Goyal N, et al. Distraction, compression, extension, and reduction combined with joint remodeling and extra-articular distraction: description of 2 new modifications for its application in basilar invagination and atlantoaxial dislocation: prospective study in 79 cases. Neurosurgery 2015 77:67. -80. discussion 80.



45. Kim IS, Hong JT, Sung JH, et al. Vertical reduction using atlantoaxial facet spacer in basilar invagination with atlantoaxial instability. J Korean Neurosurg Soc 2011 50:528-31.




46. Kim CH, Hong JT, Chung CK, et al. Intraoperative electrophysiological monitoring during posterior craniocervical distraction and realignment for congenital craniocervical anomaly. Eur Spine J 2015 24:671-8.



47. Bydon M, Macki M, Qadi M, et al. Regression of an atlantoaxial rheumatoid pannus following posterior instrumented fusion. Clin Neurol Neurosurg 2015 137:28-33.


48. Yonezawa I, Okuda T, Won J, et al. Retrodental mass in rheumatoid arthritis. J Spinal Disord Tech 2013 26:E65-9.


49. Landi A, Marotta N, Morselli C, et al. Pannus regression after posterior decompression and occipito-cervical fixation in occipito-atlanto-axial instability due to rheumatoid arthritis: case report and literature review. Clin Neurol Neurosurg 2013 115:111-6.


50. Goel A, Dange N. Immediate postoperative regression of retroodontoid pannus after lateral mass reconstruction in a patient with rheumatoid disease of the craniovertebral junction. Case report. J Neurosurg Spine 2008 9:273-6.


51. Lagares A, Arrese I, Pascual B, et al. Pannus resolution after occipitocervical fusion in a non-rheumatoid atlanto-axial instability. Eur Spine J 2006 15:366-9.



52. Park JH, Hong JT, Lee JB, et al. Clinical analysis of radiologic measurements in patients with basilar invagination. World Neurosurg 2019 131:e108-15.


53. Yoshida G, Kamiya M, Yukawa Y, et al. Rheumatoid vertical and subaxial subluxation can be prevented by atlantoaxial posterior screw fixation. Eur Spine J 2012 21:2498-505.




54. Werle S, Ezzati A, ElSaghir H, et al. Is inclusion of the occiput necessary in fusion for C1-2 instability in rheumatoid arthritis? J Neurosurg Spine 2013 18:50-6.


55. Kennedy BC, DŌĆÖAmico RS, Youngerman BE, et al. Long-term growth and alignment after occipitocervical and atlantoaxial fusion with rigid internal fixation in young children. J Neurosurg Pediatr 2016 17:94-102.


56. Matsunaga S, Onishi T, Sakou T. Significance of occipitoaxial angle in subaxial lesion after occipitocervical fusion. Spine (Phila Pa 1976) 2001 26:161-5.


57. Yoshida G, Kamiya M, Yoshihara H, et al. Subaxial sagittal alignment and adjacent-segment degeneration after atlantoaxial fixation performed using C-1 lateral mass and C-2 pedicle screws or transarticular screws. J Neurosurg Spine 2010 13:443-50.


58. Yoshimoto H, Ito M, Abumi K, et al. A retrospective radiographic analysis of subaxial sagittal alignment after posterior C1-C2 fusion. Spine (Phila Pa 1976) 2004 29:175-81.


59. Kim HS, Lee JB, Park JH, et al. Risk factor analysis of postoperative kyphotic change in subaxial cervical alignment after upper cervical fixation. J Neurosurg Spine 2019 Apr 26 1. -6. https://doi.org/10.3171/2019.2.SPINE18982. [Epub].

60. Goel A, Bhatjiwale M, Desai K. Basilar invagination: a study based on 190 surgically treated patients. J Neurosurg 1998 88:962-8.


61. Goel A, Desai KI, Muzumdar DP. Atlantoaxial fixation using plate and screw method: a report of 160 treated patients. Neurosurgery 2002 51:1351. -6. discussion 6-7.


62. Rocha R, Safavi-Abbasi S, Reis C, et al. Working area, safety zones, and angles of approach for posterior C-1 lateral mass screw placement: a quantitative anatomical and morphometric evaluation. J Neurosurg Spine 2007 6:247-54.


63. Squires J, Molinari RW. C1 lateral mass screw placement with intentional sacrifice of the C2 ganglion: functional outcomes and morbidity in elderly patients. Eur Spine J 2010 19:1318-24.




64. Dewan MC, Godil SS, Mendenhall SK, et al. C2 nerve root transection during C1 lateral mass screw fixation: does it affect functionality and quality of life? Neurosurgery 2014 74:475. -80. discussion 480-1.



65. Elliott RE, Kang MM, Smith ML, et al. C2 nerve root sectioning in posterior atlantoaxial instrumented fusions: a structured review of literature. World Neurosurg 2012 78:697-708.


66. Yeom JS, Buchowski JM, Kim HJ, et al. Postoperative occipital neuralgia with and without C2 nerve root transection during atlantoaxial screw fixation: a post-hoc comparative outcome study of prospectively collected data. Spine J 2013 13:786-95.


67. Lee JJ, Hong JT, Kim IS, et al. Significance of multimodal intraoperative monitoring during surgery in patients with craniovertebral junction pathology. World Neurosurg 2018 118:e887-94.


68. Lee HJ, Kim IS, Sung JH, et al. Significance of multimodal intraoperative monitoring for the posterior cervical spine surgery. Clin Neurol Neurosurg 2016 143:9-14.


70. Miyata M, Neo M, Fujibayashi S, et al. O-C2 angle as a predictor of dyspnea and/or dysphagia after occipitocervical fusion. Spine (Phila Pa 1976) 2009 34:184-8.


71. Maulucci CM, Ghobrial GM, Sharan AD, et al. Correlation of posterior occipitocervical angle and surgical outcomes for occipitocervical fusion. Evid Based Spine Care J 2014 5:163-5.




72. Hong JT, Lee SW, Son BC, et al. Hypoglossal nerve palsy after posterior screw placement on the C-1 lateral mass. Case report. J Neurosurg Spine 2006 5:83-5.


74. Park SJ, Kim AR, Hong JT, et al. Crystallization kinetics of lead halide perovskite film monitored by in situ terahertz spectroscopy. J Phys Chem Lett 2017 8:401-6.



- TOOLS



























