Pediatric Cervical Spine Injuries and SCIWORA: WFNS Spine Committee Recommendations
Article information
Abstract
Cervical trauma in children have variations from the adults mainly due to anatomic differences. An optimal diagnostic and treatment strategy is critical, particularly when there is a lack of standardized protocols for the management of such cases. This review paper examines the diagnostic and treatment options of pediatric cervical trauma and Spinal Cord Injury Without Radiographic Abnormality (SCIWORA). A literature search for the last 10 years were conducted using key words. Case reports, experimental studies, papers other than English language were excluded. Up-to-date information on pediatric cervical trauma and SCIWORA were reviewed and statements were produced to reach a consensus in 2 separate consensus meeting of WFNS Spine Committee. The statements were voted and reached a consensus using Delphi method. This review reflects different aspects of contemporary pediatric cervical trauma decision-making and treatment, and SCIWORA. The mainstay of SCIWORA treatment is nonsurgical with immobilization, avoidance of risky activities. Prognosis generally depends on the initial neurological status and magnetic resonance imaging. Due to a significant discrepancy in the literature on diagnostic and management, future randomized controlled trials are needed to aid in generating standardized protocols.
INTRODUCTION
Despite the rare occurrence of pediatric cervical spine trauma (1.7%), the potential neurological complications can seriously reduce the quality of life for the child and family [1].
Although reliable results are achieved by using decision-making protocols in analysing cervical spine injuries (CSIs) in adults, there is a dearth of such guidelines for the management of CSI in the pediatric cohort. The main differences of pediatric cervical trauma from adults are variation in anatomy, mechanism of trauma, and utility of imaging modalities in children [2,3]. The literature on relationship between risk factors (age, sex, type of trauma) and cervical spinal injury is conflicting [4]. Anatomical variations (e.g., intrinsic elastic characteristics) in children are critical when implementing management strategies for CSIs [5].
The purpose of this review is to analyse the risk factors, accident site management strategies, injury clearing protocols, imaging dilemmas, and treatment strategies in paediatric CSIs in the hope of yielding formal recommendations for good clinical practice.
METHODS
This systematic review was carried out according to guidelines of PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-analysis) guidelines. Our search syntax is demonstrated in Fig. 1. This search was conducted in PubMed, ScienceDirect, and Cochrane Library databases. All studies published in the last 10 years (2009–2019) were considered for inclusion. Search terms comprised relevant key words on pediatric cervical spine trauma and Spinal Cord Injury Without Radiographic Abnormality (SCIWORA). All related clinical studies/original articles, review articles, and meta-analyses were included. All studies evaluating risk factors, primary management strategies at the accident site, controversies in imaging in children, and SCIWORA were included. Exclusion criteria included any experimental studies, case reports, irrelevant studies, or studies not published in the English language. Subsequently, fulltext articles were screened for eligibility (Fig. 1).
The authors created statements for each topic for the consensus meeting of World Federation of Neurosurgical Societies (WFNS) Spine Committee. First meeting was in June, 2019 in Moscow and a re-evaluation meeting was held in November 2019 in Peshawar, Pakistan. Those prepared statements were subjected to discussions, followed by voting process by the members of the WFNS Spine Committee using Delphi method. Answering to the questionnaire each expert voted for all of the statements grading every item on a 5-point scale according to Delphi method. 1=total disagreement, 2=disagreement, 3=agreement, 4=more than agreement, 5= total agreement. consensus is reached when the sum of items “1”+“2” or “3”+“4”+“5”exceeds 66%. We called a negative consensus if 1-2> 66%, positive consensus= 3-4-5> 66%, no consensus=1-2 or 3-4-5< 66%.
The following data were extracted from the included articles: year of publication, study design, number of patients, anatomical differences in the paediatric and adult cervical spine, risk factors for cervical spine trauma, use of imaging modality, and management strategies.
REVIEW
1. Paediatric CSI
The search with the selected keywords yielded 144 studies on the searched database. Of these reports, 29 studies complying with the inclusion criteria were included (Inclusion flowchart, Fig. 1). From the studies included, 10 studies evaluating risk factors for CSIs; 9 studies analysing imaging modalities; 4 studies evaluating initial management at the accident site were included.
1) Paediatric anatomy
The specifics of the pediatric anatomy with often significant variations from the adult patient's anatomy is essential to be aware of and consider in the management of the pediatric cervical spine trauma [5-7]. Some of them are summarized below:
(1) Synchondroses separate ossification centers in a developing spine and can lead to an erroneous diagnosis of injury in a setting of a traumatic event [8-10].
(2) Atlas, for example, has 3 ossification centers at birth. They fuse at about 7 years of age.
(3) Axis has 4 ossification centers at the birth and they fuse at age 3–6 years. So, no synchondrosis should be visible on open-mouth odontoid view in a child older than 6 years.
(4) The subaxial cervical spine vertebrae also have 3 ossification centers at birth (the body and 2 neurocentral arches) which fuse at age 3–6 years. Ossified vertebral bodies are initially wedge-shaped until square at about the age of 7 years [11].
(5) Intrinsic elastic characteristics are also a pertinent variation between the spinal column of children and adults, which could lead to abrupt self-reduction after dislocation trauma, which in turn could lead to the absence of radiological evidence of traumatic abnormality (SCIWORA) [12]. The superficial and horizontal facing facet joints, and the immaturity of the uncinate processes in children also contribute to higher degree of motion [1].
(6) A larger head to body ratio in kids allows a greater force through the upper cervical spine region and makes children more vulnerable to injury, the so-called “fulcrum effect.” The fulcrum (point of maximal mobility) of cervical motion is at C2–3 leading to high levels of cervical injury. With age, the fulcrum goes down the cervical spine levels [13].
2) Risk factors for CSI in children
The cervical spine region is involved quite often between the ages 0–8 years, more specifically, up to 39% of cases involve the first 2 cervical levels [1,14].
(1) Age
The literature on the correlation between age and rate of cervical spinal injury is varied. Mallory et al. [15], analysing motor vehicle accidents in different age groups revealed that younger patients were more likely to sustain upper spinal trauma than older age groups.
In regards to the cervical region, a lower rate of upper CSIs and an increase in the lower cervical spinal injury rate in children aged more than 9 were reported [5,16]. Also, Mallory et al. [15], revealed that children < 7 years were more likely to sustain an atlanto-occipital dislocation in comparison to other age groups.
(2) Sex
Males are reported to be at higher risk (male:female ratio of 2:1) for CSI in comparison to female counterparts [17]. Ugalde et al. [18], found a male dominance, every 2 in 3 patients, in those with cervical injury after blunt trauma. Chaudhry et al. [19], also revealed similar results, in which CSI patients were more likely to be male.
3) Aetiology and epidemiology
Blunt trauma is one of the most common reasons (95%) for cervical spine trauma, frequently due to motor vehicle incidents [1,13,17,20-23]. CSIs due to falls were 9 percent higher in children aged less than 8 years in comparison to children aged over 8 years old. On the other hand, CSIs due to sports were more frequent in children over 8-year-old [24-26].
Spinal injury is relatively rare in children (overall 1%–2% incidence). Pediatric spinal trauma is not unusual, with 60%–80% of the spinal trauma occurring in the cervical region versus 30%–40% of the cases in the adult spinal trauma affecting the cervical spine. Motor vehicle accidents are the leading cause of pediatric SCI (60%) with falls and sports injuries after that.
Evidence for risk factors is summarized in (Table 1).
4) Early management strategies
Thorough assessment at the accident site in trauma cases is considered of great significance, as errors in the initial management are known to be resulting in avoidable mortality approximately 40% of patients [27,28]. Despite “Advanced Trauma Life Support protocols” being in place, various deviations from these protocols have been identified, of which 4/10 could be categorized as erroneous practices, which could lead to significant harm to the patients [29-31].
5) Immobilization
The management strategy at the trauma site involves an optimum immobilization of the cervical spine until the patient has undergone full examination at the hospital, as it avoids further harm in the presence of instability and increase the neurological symptoms during the transfer period [32,33]. Kim et al. [34], analysed immobilization practices during transfer for pediatric cohorts with CSI. Results revealed that out of 84 patients who were younger than 24 months, immobilization through the collar and hard spinal board was only appropriately done in 41% of patients [34].
Some studies suggest that hard collars are not practical for very young children, and fitting can be complicated due to patients being agitated and in pain, leading to severe complications. Furthermore, Chan et al. [35], revealed severe collar-related complications in 10% of CSI patients, mainly in the form of erythema and ulcers.
Some authors report practices of sandbags supplemented with tape for immobilization in CSIs [36]. Due to a larger head to body ratio in children, the lay-flat strategy can lead the head to increased flexion. Hence, an occipital recess or elevation of the thoracic region is considered pertinent in minimizing the effect [37,38].
6) Clinical examination
The diagnostic process of a pediatric patient following a traumatic event follows the usual algorithm with history, physical examination and imaging to establishing the diagnosis.
Predisposing conditions should also be learned. Approximately 15% of Down syndrome patients would have atlantoaxial instability, which is a predisposing moment for C0-C1-C2 segment injury. Achondroplasia could be associated with cervicomedullary junction stenosis and an individual predisposition for high cervical spinal cord injuries.
7) Imaging techniques
Unfortunately, there is still no consensus on diagnosing of the pediatric CSI patients. There is considerable debate on the most suitable imaging method for CSIs, as all modalities yield varying degrees of sensitivity. It is pertinent to prevent unnecessary imaging in children due to effects of radiation [8].
CNS guidelines [39] indicate that in children younger than 3 years old, no imaging should be conducted if Glasgow Coma Scale (GCS) point score is higher than 13, no evidence of neurological deficit, no posterior midline cervical fragility, no related cause of hypotension, no intoxication and have not been in any of the 3 scenarios (motor vehicle accident, fall from a height greater than 10 feet, nonaccidental trauma). In the absence of even one criterion, imaging of the cervical spine is advisable.
(1) Direct radiograms
There is considerable debate on the usefulness of the plain radiograph in paediatric CSI patients. A lateral view can identify up to 80%–90% of fractures dislocations and subluxations [40,41].
(2) CT scan
Hale et al. [20], evaluated the sensitivity of x-rays and computed tomography (CT) in CSIs in the paediatric cohort (n = 1,296). Results revealed that x-ray missed 32% of CSIs identified via National Emergency X-radiography Utilisation (NEXUS) [20]. Furthermore, CT was linked to an equally high (94% and 97%) sensitivity in children, however, radiation risk in young children is a significant limitation [42,43]. National Institute for Health and Care Excellence (NICE) guidelines suggest that indication for CT scan within the first 60 minutes is reserved for patients with GCS point score less than 13 on admittance. Other indications are patients with intubation, or a focal neurological deficit, upper/lower limb paresthesia, and substantial doubt of trauma despite the absence of abnormal radiograph, if the radiograph is technically challenging/unclear or if radiographs suggest a critical bone trauma [44].
Furthermore, NICE guidelines suggest that CT should be advised if a precise diagnosis of CSI is required in new cases or before surgical intervention [44]. In the absence of such variables, radiographs should be indicated within the first 60 minutes, if a harmless examination of neck movement cannot be conducted or a dangerous mechanism of trauma is present [45]. There is a significant discrepancy in the average radiation dose for paediatric cervical spine imaging. Hence, there is a need for standardization in terms of radiation dosage for CT exams.
(3) Magnetic resonance imaging
Magnetic resonance imaging (MRI) is considered one of the most informative imaging modalities and enables examination of ligamentous trauma that could be overlooked on CT imaging [45,46]. Gargas et al. [47] (n = 173) analysed the findings on MRI after a normal CT scan in paediatric CSIs. He found significant injuries on MRI in 17% of patients, of which 2.9% needed stabilization [47]. There are significant limitations of MRI, including time; instability of patient during the initial resuscitation period, expense, radiologist’s availability, the need for anesthesia for younger children. Furthermore, in severe cervical spinal injuries, MRI does not significantly affect the clinical outcomes [48]. SCIWORA frequently occurs in younger children and upper cervical spinal region [32]. Furthermore, pediatric patients who had blunt trauma from sports or recreational activities were 19% more likely to have SCIWORA in comparison to other reasons [49].
8) Treatment
There are various characteristic patterns in CSIs in children (Table 2). In regards to treatment, conservative strategies such as immobilization via collars, halo vest, and Minerva jackets, and surgical treatment (internal fixation of the cervical spine) have all important implications in the paediatric population.
Immobilization of the cervical spine is complicated due to the intrinsic elastic characteristics. Besides, natural limitations of the collar (allowing some degree of rotation leading to incomplete stability), halo vest (infection, loosening of the pin, as well as psychological effects) are all crucial points to consider [50].
(1) Medical management
Spinal cord hypoperfusion is now known to be one of the biggest reasons for secondary injuries in spinal cord trauma. Methylprednisolone usage is not recommended, although some authors claim that its usage should be made on an individual basis, particularly in paediatric spinal cord injuries [38,51,52].
(2) Surgery
The indications for surgical decompression include incomplete spinal cord injury with spinal cord compression. Some authors believe that surgical decompression within 24 hours or even within 8 hours is most efficient in terms of function preservation and recovery [7]. However, due to lack of level I evidence, any concrete conclusions should be reserved until further randomized controlled trials are conducted. Internal fixation is indicated in cases of unstable injuries, progressive deformity, and extensive decompression [53].
Statement 1: Children with neurological spinal cord signs and without x-ray/CT-scan abnormalities need MRI. (100% yes)
Statement 2: Surgery is indicated for irreducible rotatory atlanto-occipital dislocation. (100% yes)
Statement 3: Minerva cast may be used instead of Halo in children < 5 years with cervical spine fracture or dislocation without surgical indication. (100% yes)
2. Spinal Cord Injury Without Radiographic Abnormality
1) Introduction
SCIWORA continues to be a controversial diagnosis in patients presenting with signs of spinal cord injury and clinicalradiographic mismatch. This is in part due to the rarity of the condition, especially in the settings of growing availability of CT/MRI scans and lack of high-grade research data.
Unfortunately, there are no uniform guidelines on diagnosis and treatment of SCIWORA patients.
This manuscript aims to perform a thorough review of the data on the topics of SCIWORA, medical management of spinal cord injury and regenerative therapy based on the current clinical evidence and results of WFNS consensus meeting.
The first case of SCIWORA was reported by Burke in 1974, the acronym “SCIWORA” was defined in 1982 by Pang and Wilbarger in a series of 24 pediatric patients who suffered traumatic myelopathy with no radiographic evidence of fractures, dislocations, or malalignment of the spinal column and later the concept was translated to adults by Hirsh et al. [12,39,54,55].
SCIWORA is commonly seen in the pediatric age group involving cervical spine more frequently than the thoracic and lumbar spine. Adult SCIWORA is a rare phenomenon and there is limited data concerning its exact pathophysiology. The incidence has been reported between 13 to 19% of spinal injuries in children and 10%–12% in adults. It is far more common in males. The NEXUS Study reported a 0.08% frequency of SCIWORA among the enrolled adult population [56]. Some authors state that SCIWORA might be underreported in adults, although this might be explained by the growing availability of CT/MRI scans [39,55,57-59].
The term “adult SCIWORA”, although rarely seen in literature, creates considerable controversy. In view of a profound discrepancy with the emergence of routine MRI protocols for patients with clinical signs of myelopathy, a series of more precise terms like “SCIWORET” (SCI without radiographic evidence of trauma), “SCIWOCTET” (SCI without CT evidence of trauma), “SCIWONA” (SCI without neuroimaging abnormality) and a few others have subsequently come into use (Table 3) [58,60].
2) Etiology and epidemiology
Carroll et al. [13] performed a systematic review of patients with SCIWORA. From the 368 documented cases, approximately 68.5% were male, and 31.5% were female. Cervical spine was involved in 87% of the patients; thoracic spine was involved in 9.5%; lumbar spine was involved in 1.5%; and in 2% the SCI spanned the cervical and thoracic levels [13].
In a series of 297 children patients with SCIWORA, Knox demonstrated that overall, the most common cause of injury was sports injuries which were responsible for 41% of injuries, followed by motor vehicle collisions of 26%, then by falls of 14%, assault of 4%, and being struck by a falling object of 3% [61]. In adult patients, falls appear to be the most common mechanism of injury. Como et al. reported that, of the 24 adult patients with SCIWORA, 67% had a mechanism of fall [62].
3) Pathophysiology and mechanisms of injury
Based on recent literature, several pathologic mechanisms causing SCIWORA have been described. These include spinal cord traction and injury due to hyperflexion or hyperextension and parenchymal damage from edema or vascular injury [57,63,64].
It is conceivable that SCIWORA is seen less frequently in adults as a result of age-related changes in bone morphology and a decrease in ligamentous laxity. Preexisting degenerative changes to the spine, such as posterior vertebral spurs, ligamentum flavum bulging, and decreased vertebral body height are frequently found in adults presenting with a clinicoradiological mismatch. Hyperextension injury in spinal stenosis, even if trivial, can result in a central cord syndrome [60,65,66].
4) Diagnostic evaluation
Published reports indicate that patients with SCIWORA may present with a wide range of symptoms, including different level of lower and upper extremity weakness, sensory loss, paresthesia, changes in tendon reflexes, loss of bladder and bowel function, signs of anterior/central/posterior cord or Brown-Sequard syndrome [57,67]. Boese and Lechler [55] showed that adults most commonly presented with American Spinal Injury Association (ASIA) grade C (39.7%) and grade D (22.8%) [13].
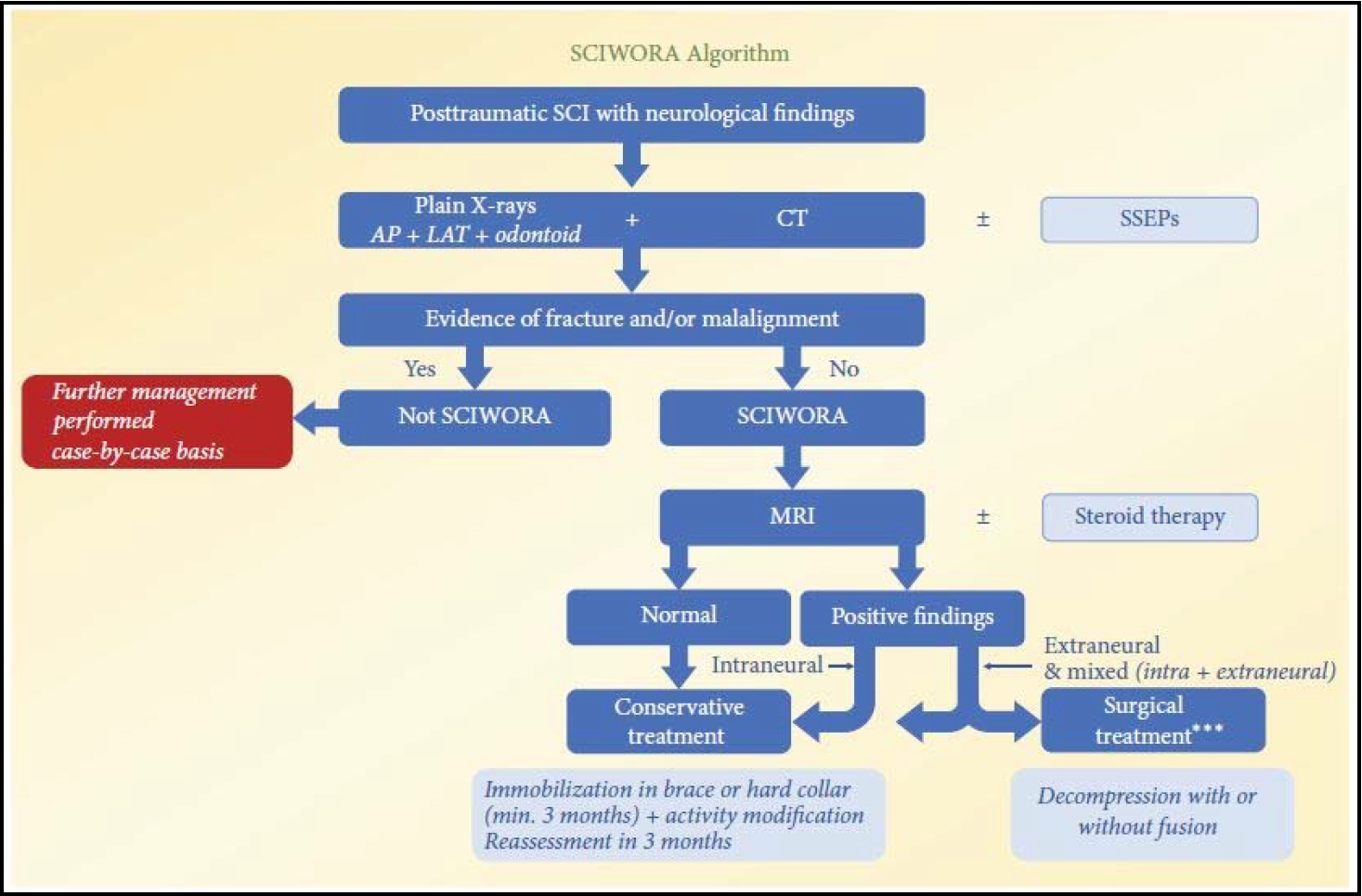
Patients with suspected SCI should first be checked with direct radiograms and CT scans [57]. CT scans should better be performed in thin-sections with coronal and sagittal 3-dimensional reconstructions. This is currently the standard screening method for diagnosing patients with acute spinal trauma [57,68].
Neither x-ray nor CT will show any signs of vertebral column injury in patients with SCIWORA despite the positive neurological findings, although new generation 64-slice CT scanners can demonstrate better diagnostic results [57,68].
MRI studies have become the gold standard for diagnostic imaging in patients with suspected SCI, able to detect characteristic pathomorphological soft tissue changes in SCIWORA patients. They are typically divided into 5 common patterns reflecting the patient’s present state: The first is complete spinal cord disruption. The next are major intramedullary hemorrhage (more than 50% of cord on axial MRI) and minor intramedullary hemorrhage (less than 50%). The last 2 patterns are spinal cord edema and patients with neurologic symptoms and no signs of spinal cord injury [57,60,63].
MRI within first 24 hours has been recommended in patients displaying a clinic-radiologic mismatch. However, if no pathology is found on early MRI, a follow-up scan may show intramedullary changes days later [39].
Boese and Lechler [55] classified MRI imaging patterns into 4 types: type I, patients with no detectable pathology; type II, patients with abnormalities on MRI scans; which in turn was divided into type IIa, extraneural abnormalities; type IIb, intraneural abnormalities; and type IIc, intra- and extraneural anomalies.
According to the study, 7.1% had no MRI abnormalities (type I) and 92.9% exhibited abnormal scan results (type II). Of the latter, 11.7% revealed extraneural (type IIa), 36.9% revealed intraneural (type IIb), and 44.3% revealed combined abnormalities (type IIc). The authors also proved a strong correlation between the type of damage seen on MRI and patient outcome. This supports the conclusion by Machino et al. [66], that severity of intramedullary MRI changes is directly related to symptom severity and patient outcome [39,57].
Diffusion-weighted MRI is promising but has not added any useful predictive information to conventional sagittal fluid-sensitive sequences [58,69].
5) Treatment
The universally accepted approach is to rule out “red flag” signs, followed by a conservative treatment course and restriction of physical activities up to 6 months regardless of immobilization used. The patient and their attendants are educated about log rolling precautions, prevention of bedsores, and care of bowel and bladder [39,57,59,67]. Physiotherapy can be instituted as soon as the general condition of the patient permitted and continued throughout the course of treatment.
In most SCIWORA cases, intravenous (IV) steroid therapy is started before an MRI scan can be completed and any detailed information with regard to pathological findings is available. Sharma et al. in their SCIWORA series used methylprednisolone therapy only when it was applicable as per North American Spinal Cord Injury Study III criteria, although there is a lack of evidence supporting routine use of high-dose IV steroids in SCIWORA patients [57,59].
In a series including 48 adult SCIWORA patients, Martinez-Perez et al. [68] treated 14 patients operatively. There was an improvement of at least one point on the ASIA Scale in 86% of the patients who received operative treatment compared with 76% of the patients who were treated conservatively [57,68].
Based on current evidence, surgical treatment is not recommended in SCIWORA patients with normal or pure intraneural MRI findings (i.e., cord edema or contusion without compression) regardless of patient’s neurological status. Although clear MRI evidence of ligamentous injury, instability, spinal cord compression along with worsening, or not-improving neurological findings should be indications for surgical treatment. However, no controlled study to date has compared the outcomes of surgical and nonsurgical treatment (Fig. 2) [57].
6) Prognosis
Although SCIWORA often results from serious trauma, mortality is rare. In a 2016 study of 297 patients with SCIWORA, the authors reported a mortality rate of 2% [61]. In general, most SCIWORA patients treated conservatively show improvement in neurological status after injury, and surgical treatment is rarely justifiable. It is worth noting, that the injury itself should not be considered mild in nature, and for some patients, the prognosis can be dreadful such as permanent neurological impairments and death [57].
Long-term outcomes predictors of prognosis are the initial neurological status and patient MRI findings. Patients with major cord hemorrhage typically have a poor neurologic prognosis, whereas patients with minor hemorrhage have the possibility of partial recovery. Patients with cord edema alone on MRI typically have a good prognosis. Patients with neurologic findings consistent with SCIWORA on exam and normal MRI typically have an excellent prognosis [63].
Martinez-Perez et al. [68] reported that, at 1-year post injury follow-up, complete recovery in neurological status (ASIA grade E) was achieved only in patients with incomplete neurological injury (ASIA grades C and D) at admission.
CONCLUSION
The discrepancies in current practices on the diagnostic and treatment strategies of paediatric CSIs can largely be attributed to a lack of standardized protocols (level I evidence). Future randomized controlled trials are needed to draw reliable conclusions on the optimum management of cervical spine trauma in children.
In patients with SCIWORA MRI is necessary. MRI findings are in accord with the symptoms and predict neurologic outcome. A conservative treatment is suitable in most of those cases.
After consensus meetings and discussions, the WFNS Spine Committee has issued the following recommendations.
WFNS SPINE COMMITTEE RECOMMENDATIONS
Recommendations for Pediatric CSI
• Children with neurological spinal cord signs and without x-ray/CT-scan abnormalities need MRI
• Surgery is indicated for irreducible rotatory atlanto-occipital dislocation
• Minerva cast may be used instead of Halo in children < 5 years with cervical spine fracture or dislocation without surgical indication.
Recommendations for SCIWORA
• SCIWORA is a clinical-radiological condition of spinal cord injury without radiographic or CT evidence of fracture, dislocation, disc and ligaments damage or signs of instability. This statement reached a full (100%) consensus.
• We should always perform an MRI if the patient, after cervical trauma, has neurologic symptoms, but his x-ray/CT findings are nonconclusive. This statement reached a full (100%) consensus.
• MRI findings in patients with SCIWORA correlate with symptoms and predict neurologic outcome. This statement got a strong consensus (91% yes).
• In patients with SCIWORA, conservative treatment should be preferred instead of surgical treatment. This statement got a strong consensus (82% yes).
Notes
The authors have nothing to disclose.