 |
 |
- Search
|
|
||
See commentary "Making a Good Surgery Even Better" in Volume 18 on page 197.
Abstract
Objective
Anterior cervical discectomy and fusion (ACDF) is a common procedure for the treatment of cervical disease. Circumferential procedures are options for multilevel pathology. Potential complications of multilevel anterior procedures are dysphagia and pseudarthrosis, whereas potential complications of posterior surgery include development of cervical kyphosis and postoperative chronic neck pain. The addition of posterior cervical cages (PCCs) to multilevel ACDF is a minimally invasive option to perform circumferential fusion. This study evaluated the biomechanical performance of 3-level circumferential fusion with PCCs as supplemental fixation to anteriorly placed allografts, with and without anterior plate fixation.
Methods
Nondestructive flexibility tests (1.5 Nm) performed on 6 cervical C2–7 cadaveric specimens intact and after discectomy (C3–6) in 3 instrumented conditions: allograft with anterior plate (G+P), PCC with allograft and plate (PCC+G+P), and PCC with allograft alone (PCC+G). Range of motion (ROM) data were analyzed using 1-way repeated-measures analysis of variance.
Results
All instrumented conditions resulted in significantly reduced ROM at the 3 instrumented levels (C3–6) compared to intact spinal segments in flexion, extension, lateral bending, and axial rotation (p < 0.001). No significant difference in ROM was found between G+P and PCC+G+P conditions or between G+P and PCC+G conditions, indicating similar stability between these conditions in all directions of motion.
Conclusion
All instrumented conditions resulted in considerable reduction in ROM. The added reduction in ROM through the addition of PCCs did not reach statistical significance. Circumferential fusion with anterior allograft, without plate and with PCCs, has comparable stability to ACDF with allograft and plate.
Anterior cervical discectomy and fusion (ACDF) is a common procedure for the surgical management of cervical pathology that produces excellent clinical results [1]. ACDF is used most commonly in the treatment of younger patients with single or 2-level disease. Traditional open posterior surgery is used more commonly for older patients with multilevel disease. Multilevel ACDF or combined anterior and posterior procedures are options for patients with multilevel pathology, including those with kyphosis. A common criticism of multilevel anterior procedures is that they may increase the likelihood of dysphagia, postoperative hoarseness, and pseudarthrosis [2,3]. The severity of the dysphagia is correlated with the operative time, which is usually longer as more levels are treated, and with use of an anterior plate [4]. Multilevel anterior procedures, particularly in patients with risk factors for pseudarthrosis (e.g., nicotine use, diabetes, and chronic corticosteroid use), lead to higher rates of nonunion [5].
A posterior cervical approach can be used either as a primary or a complementary choice to treat central or foraminal stenosis. Compared with anterior approaches, traditional posterior approaches such as laminectomy and lateral mass fusion and laminoplasty demand wide paraspinal muscle detachment, have an increased risk of complications such as chronic neck pain, and have a higher rate of infection due to fluid accumulation [6]. A common criticism of posterior surgery is that it may increase the likelihood of cervical kyphosis and lead to more blood loss, longer hospitalization, and greater neck pain.
Paraspinal muscles play an essential role in spinal stabilization; hence, keeping them intact is desirable. The use of a posterior cervical cage (PCC) is an alternative approach to treat spondylosis associated with radicular pain [7-9]. The PCC can be implanted via a tubular retractor system in a percutaneous or mini-open approach without the necessity of a large exposure, which keeps paraspinal muscles intact. Other advantages of the PCC include that it is a technically easier device to implant with relatively fast operative time and low estimated blood loss. One main disadvantage is the potential for loss of cervical lordosis, because a small distraction is performed on the posterior column with potential displacement when a stand-alone procedure is performed. In a clinical study, Tan et al. [10] reported no significant difference in the preoperative and postoperative cervical lordotic angles using a similar PCC device. Goel and Shah [7] reported excellent 6-month outcomes with intraarticular facet joint spacers without significant impairment of cervical lordosis.
A circumferential surgical approach to fusion using PCCs in tandem with anterior cervical discectomy and allograft may help alleviate risks associated with both anterior and posterior surgical approaches. Few in vitro biomechanics studies have investigated PCC placement either alone or as an adjunct to traditional instrumentation [11-13]. This study evaluated the biomechanical stability of bilaterally placed PCCs as supplemental fixation to anteriorly placed allografts, with and without anterior plating, in a 3-level ACDF human cadaveric model. We hypothesized that the addition of a PCC to an ACDF with allograft alone would result in a reduction in range of motion (ROM) similar to that of an ACDF with allograft and anterior plate. If anterior plating can be avoided, it may be possible to decrease operative time, cost, and complications related to retraction, including dysphagia and hoarseness. Reducing these complications has a potential socioeconomic impact, because a patient who experiences such complications can require feeding tubes and long-term follow-up with a speech therapist. Elderly patients with multilevel surgery are especially at risk. The addition of circumferential fusion with PCCs will likely translate clinically into higher fusion rates, without the morbidity of open posterior cervical approaches.
Six fresh-frozen cervical cadaveric specimens (C2–7) were selected for this study (N = 6); 4 were male and 2 were female (age: range, 25–65 years; mean ± standard deviation [SD], 44.83 ± 5.54 years). All specimens underwent direct visual inspection, palpation, plain film radiography, and a review of donor medical history to exclude any abnormalities or flaws that could adversely affect joint health and joint mobility (e.g., metastatic disease, osteophytes, disc narrowing, and joint arthrosis). Dual-energy x-ray absorptiometry scans were performed on C4 of each specimen to assess bone mineral density (mean ± SD, 0.57 ± 0.11 g/cm2) because osteoporosis was an exclusion criterion.
Specimens were cleaned of muscle tissue while keeping intact all ligaments, joint capsules, and intervertebral discs. The caudal endplate of C7 was reinforced with household wood screws, placed in a square metal mold, and embedded using fast-curing resin (Smooth-Cast, Smooth-On, Inc., Easton, PA, USA) for rigid attachment to the base of the testing apparatus. Similarly, the cranial portion of the C2 vertebra, including the dens, was reinforced with household screws and embedded in resin using a cylindrical cast for load application. Specimens were wrapped with double bags and stored at -20°C until tested. Immediately prior to testing, specimens were thawed in a bath of normal saline at 21°C. The specimens were subsequently tested intact and in 3 instrumented conditions: (1) discectomy with allograft plus anterior cervical plate (G+P), (2) discectomy with PCC plus allograft plus anterior plate (PCC+G+P), and (3) discectomy with PCC plus allograft after anterior plate removal (PCC+G) (Fig. 1).
A 3-level fusion construct was chosen to simulate a potentially challenging clinical scenario because higher rates of pseudarthrosis and failure have been reported with increasing levels of fusion [14-16]. After intact testing, 3-level anterior discectomies (C3–4, C4–5, C5–6), followed by posterior longitudinal ligament release, were performed using standard surgical tools such as rongeurs and curettes. Allografts (VertiGraft VG1 Cervical Bio-implant, LifeNet Health, Inc., Virginia Beach, VA, USA) were implanted in each disc space. The allografts were sized using fluoroscopic imaging, aiming for moderately tight contact with both endplates, with enough contact to offer resistance when light pull-out traction was performed. Anterior cervical plates (Atlantis, Medtronic, Minneapolis, MN, USA) were appropriately sized for each specimen and secured with 2 screws (4.5 × 13 mm or 4.5 × 15 mm) for each vertebral level. Screws used for plating were also appropriately sized in length to avoid breach of the posterior wall of the vertebral body. Appropriate size and position of all devices were confirmed via fluoroscopy.
After the ACDF condition with G+P was tested, bilateral incisions were made posteriorly at each facet joint capsule at C3–4, C4–5, and C5–6, and a PCC device (CAVUX Cervical Cage and ALLY Bone Screw, Providence Medical Technology, Pleasanton, CA, USA) was placed in each facet joint using the CORUS Spinal System (Providence Medical Technology) for instrumentation. The PCC device had a parallel shape 10 mm in length×5.5 mm in width× 4 mm in height (2.5-mm body with 0.75-mm teeth on either side). Gentle traction was performed to ensure proper purchase between the joint surfaces; the fixation with ALLY screws ensured good surface contact with the cage. For the final spinal condition, the anterior plate and screws were removed from each specimen, leaving PCC cages and allografts (PCC+G) in place.
Nondestructive flexibility tests (1.5 Nm) were performed using a 6-degree-of-freedom robotic testing system [17] (Fig. 2). Test specimens were secured caudally to the robot frame and cranially to a 6-axis load sensor (Model 45E15A4, JR3, Inc., Woodland, CA, USA), which was, in turn, rigidly affixed to the robot end effector. Dynamic pure moment loads were continuously applied at an average global rotation rate of 1.7° per second in flexion, extension, right and left lateral bending, and right and left axial rotation over 3 cycles of loading to precondition specimens for comparison, with data analyzed on the last cycle. Loading using pure moments has the advantage that the same load is distributed to each level of the spine, which ensures an equivalent comparison among all spinal levels [18,19]. All off-axis loads were actively controlled to zero value.
During all tests, 3-dimensional motion measurements were made with the Optotrak 3020 active marker system (Northern Digital, Inc., Waterloo, ON, Canada). This system measures 3-dimensional positions of infrared-emitting markers rigidly attached in a noncollinear arrangement to each vertebra at the tip of three 4-cm surgical-guide Kirschner wires drilled into each vertebral body. Custom software was used to convert the marker coordinates to an appropriate angular ROM about each of the anatomical axes [20]. A 1-way repeated-measures analysis of variance was used to compare angular ROM among all 4 conditions tested. Post hoc Holm-Šidák comparisons were performed as needed. All statistical comparisons had a significance level set at p < 0.05.
All 3 instrumented conditions significantly reduced ROM compared to the intact condition at all instrumented spinal levels (C3–6) and directions of testing (p < 0.001) (Tables 1 and 2, Fig. 3). In flexion and extension over the instrumented spinal levels, PCC+G+P exhibited the smallest instrumented ROM (< 0.2°). Mean instrumented ROM for the G+P and PCC+G conditions were 0.4° and 0.6° in flexion and 0.3° and 1.0° in extension, respectively. The greatest numerical difference in ROM between G+P and PCC+G occurred in extension; ROM in PCC+G demonstrated a mean difference of 0.5° to 0.8° over the 3 instrumented levels compared to G+P. In lateral bending, all instrumented-level ROMs were small, with combined left and right ROMs averaging 0.3° for the G+P condition and 0.2° for both the PCC+G+P and PCC+G conditions. Similarly, in axial rotation, the G+P condition exhibited a combined left and right mean instrumented ROM of 0.6°, which was slightly larger than mean values of 0.2° and 0.3° for the PCC+G+P and PCC+G conditions, respectively.
Overall, the PCC+G+P condition demonstrated the smallest numerical ROM overall for instrumented levels and all test directions; however, these values were not significantly different than those for the G+P condition (p ≥ 0.28 for all). The only statistically significant difference between instrumented conditions occurred between PCC+G and PCC+G+P at C4–5 in extension (0.8° and 0.1°, respectively, p = 0.02). No difference in ROM was found between G+P and PCC+G conditions (p≥ 0.11 for all) (Tables 1 and 2, Fig. 3), indicating comparable stability between these 2 instrumented conditions at all instrumented levels in all directions of motion.
Traditionally, ACDF and open posterior techniques have been used to treat cervical myelopathy, radiculopathy, and degenerative disc disease. Studies that have examined long-term results of ACDF show an excellent clinical result, evidenced by improvement of Neck Disability Index, visual analogue scale, and medullary complaints compared to preoperative levels, as well as arrest of disease progression in patients with myelopathy and radiculopathy [21-24]. The use of an anterior plate in ACDF with cortical bone allograft significantly enhances arthrodesis when compared to those without a plate [25,26]. However, multilevel ACDF with an anterior plate has been associated with complications such as dysphagia, dysphonia, and hoarseness due to laryngeal nerve palsy, as well as vascular and esophageal injury [4]. Furthermore, prolonged operative duration is a significant predictor of postoperative airway complication [27]. Multilevel ACDF is also associated with higher rates of pseudarthrosis.
Traditional open posterior approaches allow surgeons to perform neural decompression (i.e., laminectomy and foraminotomy) as well as fusion but can be associated with prolonged operative duration, increased blood loss, postoperative neck pain, longer hospital stays, and kyphosis [28]. Posterior instrumented fusions have low rates of pseudarthrosis. Circumferential fusion, including multilevel ACDF with or without a plate and posterior instrumented fusion, has the benefit of anterior central and foraminal decompression, improvement in alignment, and the lowest associated rates of pseudarthrosis [29]. Unfortunately, this approach is associated with morbidity for both the anterior and the traditional posterior procedures. It is therefore of interest to examine the feasibility of an alternate approach to decompression and fusion, that is, the coupling of ACDF with or without plate fixation with bilateral, tissue-sparing, multilevel PCC fixation to achieve anterior decompression and circumferential fusion.
The current study evaluated the biomechanical stability of bilaterally placed PCCs as supplemental fixation to anteriorly placed allografts, with and without anterior plating, in a 3-level ACDF human cadaveric model. When used as an adjunct to an anterior allograft with plate, the addition of a PCC uniformly produced the smallest ROM over all instrumented spinal levels and directions of testing; however, no statistically significant differences were noted among the instrumented constructs with one exception (PCC+G and PCC+G+P at C4–5 in extension had ROM of 0.8° and 0.1°, respectively; p = 0.02). When a PCC was used as the primary stabilizing instrumentation with allografts only (PCC+G) in a 3-level fusion model, spinal stability was statistically comparable at all instrumented spinal levels and in all directions of motion to traditional anterior plate fixation with allograft (G+P).
Few biomechanical studies have evaluated the stability associated with PCC devices alone or as an adjunct, and no study has evaluated PCC implants over 3 spinal levels. Previous investigations of in vitro PCC devices have used pure moment end loads that result in a uniformly distributed bending moment (magnitude) across the entire specimen length. The nature of this loading thus makes cross-study comparisons valid. Leasure and Buckley [30] biomechanically evaluated an earlier design of the PCC used in our current study. In their study, 7 C2–T2 cadaveric specimens were multidirectionally tested to 1.5 Nm with a single-level PCC at C4–5, and with an added anterior plate at the same level. No graft or device was used at the intervertebral space; however, a nucleotomy was performed. PCC alone reduced ROM compared to intact spinal segments in all directions except for extension. When an anterior plate was added, extension ROM was significantly less than that for intact; however, the authors did not report significant differences between the PCC alone and the PCC and plate conditions. The earlier PCC design used in their study was not fixated with bone screws at the superior facet surface.
In a similar study, Voronov et al. [11] investigated the effectiveness of a bilateral stand-alone PCC device (C5–6, with intact disc) in limiting motion compared to single-level (C6–7) ACDF in the same specimens. This device was the same design of PCC used in the current study but without superior facet screw fixation. The authors reported comparable reductions for both levels with no statistically significant differences between instrumented conditions in all directions of motion. Although ROM for both levels was similar in flexion-extension (approximately 2.5°), the authors noted that ROM reductions in lateral bending and axial rotation were larger for the PCC level than for the ACDF level (mean values of 0.4° vs. 1.6° in lateral bending, and 0.6° vs. 1.7° in axial rotation). Their study also compared supplementing ACDF with PCCs over 2 levels (C3–5) in the same test specimens. Overall, the addition of PCCs significantly reduced ROM in all directions compared to single-level (C6–7) and 2-level (C3–5) ACDF. In contrast, our current study did not find statistically significant ROM differences when 3-level plated ACDF with allograft was supplemented with posterior PCCs. Possible confounding factors that may be involved in these study outcomes include different spinal levels tested and variability in tissue and in the type, size, and placement of interbody devices. In a separate study, Voronov et al. [12] also compared 2-level (C4–6) stability with a stand-alone PCC (without superior facet screw fixation) to lateral mass screw and rod fixation (LMS) across the same levels in the same specimens. They reported no statistically significant difference in axial rotation or flexion-extension but noted that LMS ROM was smaller in flexion-extension (3.1° with LMS vs. 5.0° for PCC) and that ROM was significantly reduced with PCC compared to that with LMS fixation in lateral bending.
The results from these earlier studies and our current study contrast with those of Maulucci et al. [13], who investigated the effects of 2-mm-thick allograft facet spacers alone and with LMS instrumentation in cadaveric cervical specimens tested to a 1.5-Nm load limit. They observed that the introduction of facet spacers produced a mild, statistically insignificant reduction in motion. After the subsequent addition of LMS instrumentation, ROM was significantly reduced. Numerous potential factors could account for these differences in study results, particularly cage design, size, and fixation. For example, the facet spacer studied by Maulucci et al. [13] was flat and thin, with no teeth to engage the facet surfaces and no containment or screw fixation mechanism. The PCC design investigated in our study was made of implant-grade titanium alloy, was 10 mm long and 5.5 mm wide, and had a base height of 2.5 mm with teeth protruding 0.75 mm on either side of the implant, for a total height of 4 mm. Additionally, our PCC implants were secured with a small 1-cm-long and 1.5-mm diameter screw that entered through the posterior end of the PCC, angled upward, and secured into the superior facet surface.
The subaxial cervical spine comprises highly mobile joints. Posteriorly located facet joints in tandem with the intervertebral discs provide for a 3-column support structure. Cervical lordotic curvature and the orientation of the facets favors posterior column load sharing of compressive forces in the subaxial cervical spine [31,32]. The facet joints also resist anterior shear, prevent adjacent vertebra from engaging in motions that could cause neural damage, and serve to guide vertebral motion, particularly strongly coupled lateral bending and axial rotation movement via relative translations between the joint surfaces [32]. The current and previous biomechanical studies provide evidence that a PCC aimed at restricting relative facet joint motion can also provide significant spinal stability by subsequently arresting relative vertebral rotation. Complemented with ACDF and a well-fitted allograft, the PCC+G condition demonstrated comparable stability in all tested directions of motion to that of an ACDF with an anterior cervical plate, with particular effectiveness in lateral bending and axial rotation.
As with any cadaver study that seeks to mimic an in vivo environment, our study has certain limitations that must be considered. Our results represent the acute, immediately postoperative, kinematic changes that a patient might experience after placement of a PCC in a circumferential cervical construct. Cyclic experiments were not performed because of the limited time frame of mechanical viability of cadaveric specimens in vitro. It is our belief that the low, nondestructive load magnitudes used in standard biomechanical tests, coupled with tissue degeneration outside of the body, preclude any meaningful measure or simulation of prolonged in vivo service. In addition, the evaluations in this study used pure moment loading to produce bending in all 3 anatomical planes. Although this method is a well-accepted one that reproduces in vivo cervical spine kinematics [33], it represents a simplification of the in vivo loading environment. Nevertheless, the method was robust enough to provide a direct comparison of different fixation-implantation scenarios. Lateral radiographs of the instrumented constructs were also not standardized well enough to allow accurate repeatable assessments of lordosis measures because of the high level of mobility of the cervical cadaveric specimens. Given the clinical significance of sagittal balance principles, future biomechanical and clinical studies related to PCC constructs should endeavor to strictly control and quantify this parameter. Furthermore, careful long-term clinical observation and analysis are required to ensure no displacements and no pseudarthrosis. Lastly, the absence of paraspinal and neck muscle activity, which plays a role in stabilizing the spine, as well as the absence of a disease mechanism that represents an indication for the surgery, present further limitations.
All instrumented conditions resulted in greatly reduced ROM compared with the intact condition. The addition of PCCs to an ACDF using an allograft and anterior plate with screws did not significantly affect spinal stability. The use of PCCs to supplement ACDF with only an allograft spacer demonstrated acute stability over 3 instrumented spinal levels similar to that of an ACDF with allograft and anterior plate and screw fixation. Further studies are needed to assess the role of PCC as a supplement or alternative to anterior plating.
CONFLICT OF INTEREST
J.E. Heller is a consultant in education for NuVasive, Inc. (San Diego, CA), Providence Medical Technology, Inc. (Pleasanton, CA), RTI Surgical Holdings, Inc. (Alachua, FL), SI-BONE, Inc. (Santa Clara, CA), Stryker Corp. (Kalamazoo, MI), and Zimmer Biomet Holdings. Inc. (Warsaw, IN); receives grant support from Ethicon (Somerville, NJ); and has investments in ATEC Spine (Carlsbad, CA), Portola Pharmaceuticals, Inc. (South San Francisco, CA), and Spine BioPharma, LLC (Dover, DE). Other authors have no relevant disclosures.
ACKNOWLEDGEMENTS
The study received financial support from Providence Medical Technology, Inc. The sponsor had no involvement in the study design; collection, analysis, or interpretation of data; writing of the report; or decision to submit the article for publication. The authors thank the staff of Neuroscience Publications at Barrow Neurological Institute for assistance with manuscript preparation.
Fig. 1.
Radiographic images of the 4 spinal conditions studied: (A) intact spine; (B) spine after C3–6 anterior cervical discectomy with allograft spacers and 3-level anterior cervical plate (G+P); (C) lateral view of the spine after C3–6 anterior cervical discectomy with posterior cervical cages, allograft spacers, and plate (PCC+G+P); and (D) spine with posterior cervical cages and allograft spacers after anterior plate removal (PCC+G).
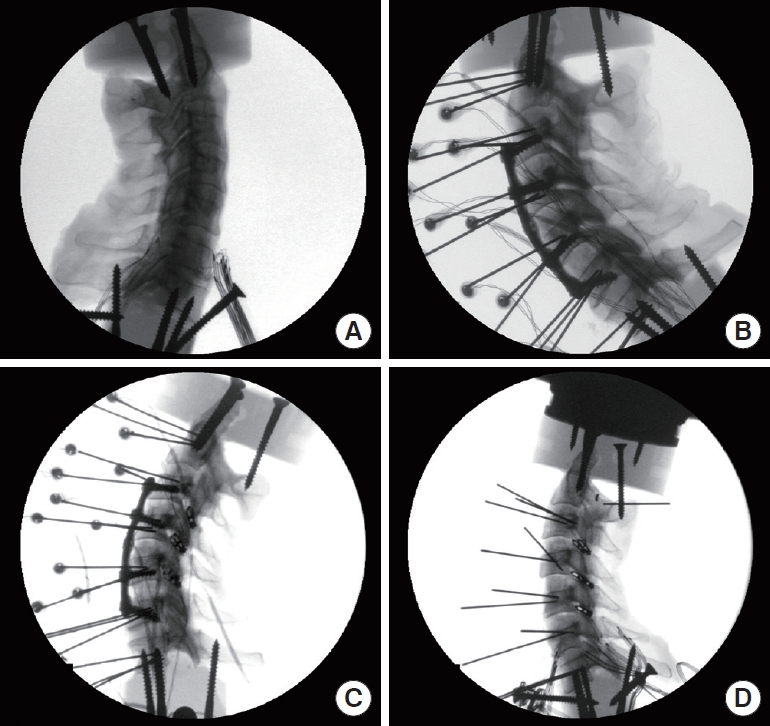
Fig. 2.
Test setup. Photograph of a cervical test specimen mounted in a gantry-style 6-degree-of-freedom robotically controlled test system with real-time load control. Specimens were tested multidirectionally under continuously applied pure moment loads at a global rotation rate of 1.7° per second. Optical LED markers were attached to Kirschner wires secured into each vertebral body to record vertebral body movement.

Fig. 3.
Mean cervical range of motion at each cervical level for each of the 4 spinal conditions tested: (A) flexion, (B) extension, (C) pooled left-right lateral bending, and (D) pooled left-right axial rotation. Error bars indicate one standard deviation. *Significant difference compared to the intact condition (p < 0.001). **Significant difference at C4–5 in extension between posterior cervical cages with allograft (PCC+G) and posterior cervical cages, allograft spacers, and plate (PCC+G+P) (p = 0.02). ROM, range of motion; PCC, posterior cervical cage.
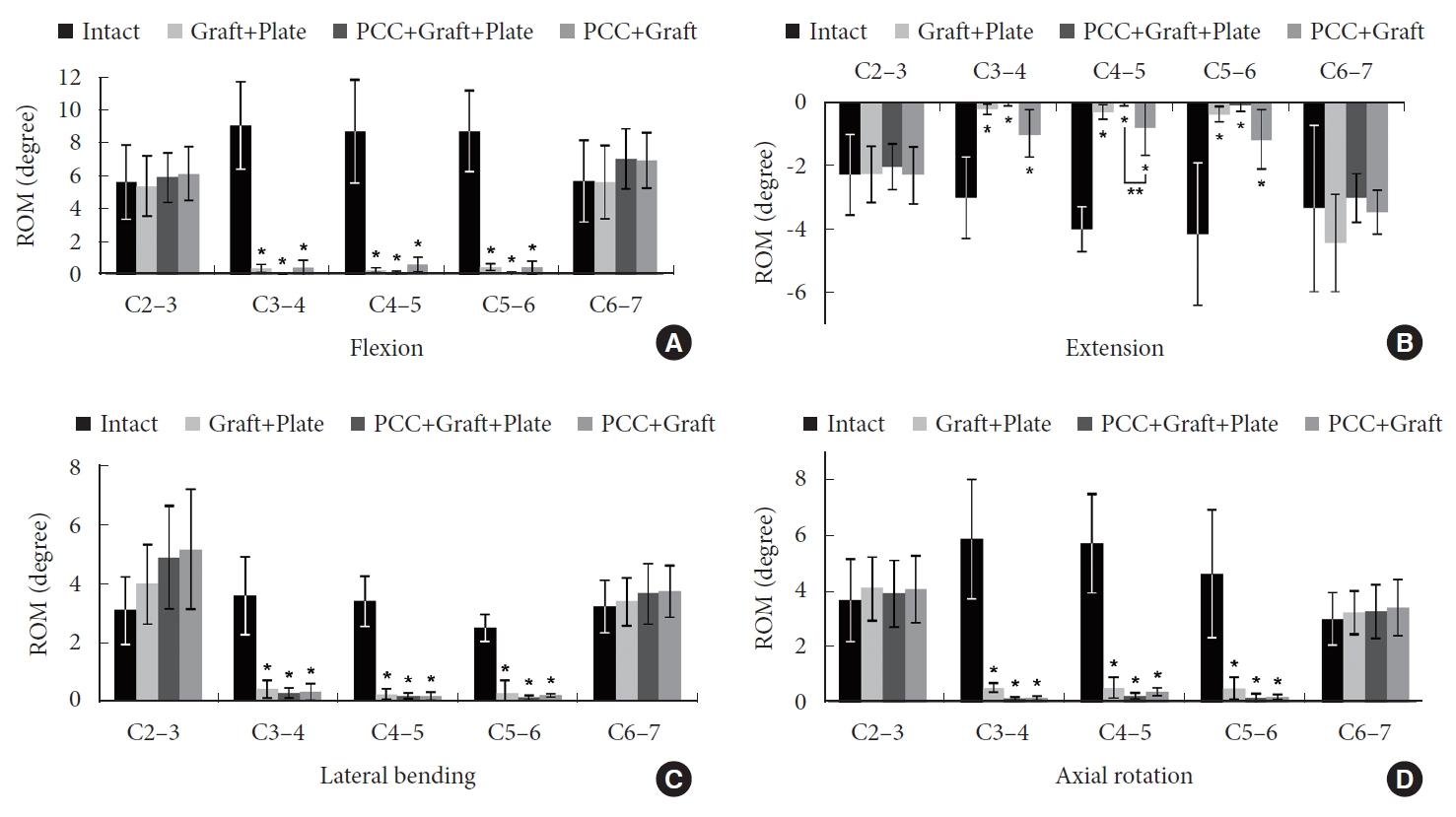
Table 1.
Segmental range of motion for each direction of loading and spinal condition tested
| Level | Intact | Graft+Plate | PCC+Graft+Plate | PCC+Graft |
|---|---|---|---|---|
| Flexion | ||||
| C2–3 | 5.6 ± 2.2 | 5.4 ± 1.8 | 5.9 ± 1.5 | 6.1 ± 1.6 |
| C3–4 | 9.1 ± 2.6 | 0.4 ± 0.2‡ | 0.1 ± 0.1‡ | 0.4 ± 0.4‡ |
| C4–5 | 8.7 ± 3.1 | 0.3 ± 0.1‡ | 0.1 ± 0.1‡ | 0.6 ± 0.4‡ |
| C5–6 | 8.7 ± 2.4 | 0.5 ± 0.2‡ | 0.1 ± 0.1‡ | 0.4 ± 0.3‡ |
| C6–7 | 5.7 ± 2.4 | 5.6 ± 2.2 | 7.0 ± 1.8 | 6.9 ± 1.6 |
| Extension | ||||
| C2–3 | 2.2 ± 1.2 | 2.2 ± 0.8 | 2.0 ± 0.7 | 2.2 ± 0.9 |
| C3–4 | 3.0 ± 1.2 | 0.2 ± 0.1‡ | 0.1 ± 0.1‡ | 0.9 ± 0.7‡ |
| C4–5 | 3.9 ± 0.7 | 0.3 ± 0.2‡ | 0.1 ± 0.1‡ | 0.8 ± 0.8‡,§ |
| C5–6 | 4.1 ± 2.2 | 0.4 ± 0.2‡ | 0.1 ± 0.1‡ | 1.1 ± 0.9‡ |
| C6–7 | 3.3 ± 2.5 | 4.4 ± 1.5 | 3.0 ± 0.7 | 3.4 ± 0.6 |
| Lateral bending | ||||
| C2–3 | 3.1 ± 1.1 | 4.0 ± 1.3 | 4.9 ± 1.7 | 5.1 ± 2.0 |
| C3–4 | 3.6 ± 1.3 | 0.4 ± 0.2‡ | 0.3 ± 0.1‡ | 0.3 ± 0.2‡ |
| C4–5 | 3.4 ± 0.8 | 0.2 ± 0.1‡ | 0.2 ± 0.1‡ | 0.2 ± 0.1‡ |
| C5–6 | 2.5 ± 0.4 | 0.3 ± 0.4‡ | 0.1 ± 0.1‡ | 0.2 ± 0.1‡ |
| C6–7 | 3.2 ± 0.9 | 3.4 ± 0.8 | 3.6 ± 1.0 | 3.7 ± 0.9 |
| Axial rotation | ||||
| C2–3 | 3.6 ± 1.4 | 4.1 ± 1.1 | 3.9 ± 1.2 | 4.0 ± 1.2 |
| C3–4 | 5.8 ± 2.1 | 0.5 ± 0.1‡ | 0.1 ± 0.1‡ | 0.1 ± 0.1‡ |
| C4–5 | 5.7 ± 1.7 | 0.5 ± 0.3‡ | 0.2 ± 0.1‡ | 0.4 ± 0.1‡ |
| C5–6 | 4.6 ± 2.3 | 0.5 ± 0.4‡ | 0.1 ± 0.1‡ | 0.2 ± 0.1‡ |
| C6–7 | 3.0 ± 0.9 | 3.2 ± 0.7 | 3.2 ± 0.9 | 3.4 ± 1.0 |
Table 2.
p-values from 1-way repeated-measures analysis of variance for each direction of range of motion
| Level |
Relative to intact |
Relative to Graft+Plate |
Relative to PCC+Graft+Plate |
|||
|---|---|---|---|---|---|---|
| Graft+Plate | PCC+Graft+Plate | PCC+Graft | PCC+Graft+Plate | PCC+Graft | PCC+Graft | |
| Flexion | ||||||
| C2–3 | 0.455 | 0.455 | 0.455 | 0.455 | 0.455 | 0.455 |
| C3–4 | < 0.001* | < 0.001* | < 0.001* | 0.858 | 0.949 | 0.926 |
| C4–5 | < 0.001* | < 0.001* | < 0.001* | 0.865 | 0.923 | 0.936 |
| C5–6 | < 0.001* | < 0.001* | < 0.001* | 0.926 | 0.981 | 0.837 |
| C6–7 | 0.868 | 0.095 | 0.082 | 0.081 | 0.078 | 0.981 |
| Extension | ||||||
| C2–3 | 0.739 | 0.739 | 0.739 | 0.739 | 0.739 | 0.739 |
| C3–4 | < 0.001* | < 0.001* | < 0.001* | 0.653 | 0.208 | 0.136 |
| C4–5 | < 0.001* | < 0.001* | < 0.001* | 0.278 | 0.108 | 0.018 |
| C5–6 | < 0.001* | < 0.001* | < 0.001* | 0.719 | 0.491 | 0.411 |
| C6–7 | 0.481 | 0.481 | 0.481 | 0.481 | 0.481 | 0.481 |
| Lateral bending | ||||||
| C2–3 | 0.110 | 0.110 | 0.110 | 0.110 | 0.110 | 0.110 |
| C3–4 | < 0.001* | < 0.001* | < 0.001* | 0.985 | 0.968 | 0.927 |
| C4–5 | < 0.001* | < 0.001* | < 0.001* | 0.974 | 0.991 | 0.955 |
| C5–6 | < 0.001* | < 0.001* | < 0.001* | 0.738 | 0.840 | 0.689 |
| C6–7 | 0.293 | 0.293 | 0.293 | 0.293 | 0.293 | 0.293 |
| Axial rotation | ||||||
| C2–3 | 0.703 | 0.703 | 0.703 | 0.703 | 0.703 | 0.703 |
| C3–4 | < 0.001* | < 0.001* | < 0.001* | 0.901 | 0.806 | 0.974 |
| C4–5 | < 0.001* | < 0.001* | < 0.001* | 0.928 | 0.791 | 0.950 |
| C5–6 | < 0.001* | < 0.001* | < 0.001* | 0.947 | 0.871 | 0.981 |
| C6–7 | 0.796 | 0.796 | 0.796 | 0.796 | 0.796 | 0.796 |
REFERENCES
1. Fraser JF, Hartl R. Anterior approaches to fusion of the cervical spine: a metaanalysis of fusion rates. J Neurosurg Spine 2007 6:298-303.


2. Cheung JP, Luk KD. Complications of anterior and posterior cervical spine surgery. Asian Spine J 2016 10:385-400.



3. Anderson KK, Arnold PM. Oropharyngeal Dysphagia after anterior cervical spine surgery: a review. Global Spine J 2013 3:273-86.



4. Rihn JA, Kane J, Albert TJ, et al. What is the incidence and severity of dysphagia after anterior cervical surgery? Clin Orthop Relat Res 2011 469:658-65.


5. Leven D, Cho SK. Pseudarthrosis of the cervical spine: risk factors, diagnosis and management. Asian Spine J 2016 10:776-86.



6. Wang SJ, Jiang SD, Jiang LS, et al. Axial pain after posterior cervical spine surgery: a systematic review. Eur Spine J 2011 20:185-94.


7. Goel A, Shah A. Facetal distraction as treatment for single- and multilevel cervical spondylotic radiculopathy and myelopathy: a preliminary report. J Neurosurg Spine 2011 14:689-96.


8. McCormack BM, Bundoc RC, Ver MR, et al. Percutaneous posterior cervical fusion with the DTRAX Facet System for single-level radiculopathy: results in 60 patients. J Neurosurg Spine 2013 18:245-54.


9. Lenzi J, Nardone A, Passacantilli E, et al. Posterior cervical transfacet fusion with facetal spacer for the treatment of single-level cervical radiculopathy: a randomized, controlled prospective study. World Neurosurg 2017 100:7-14.


10. Tan LA, Straus DC, Traynelis VC. Cervical interfacet spacers and maintenance of cervical lordosis. J Neurosurg Spine 2015 22:466-9.


11. Voronov LI, Siemionow KB, Havey RM, et al. Bilateral posterior cervical cages provide biomechanical stability: assessment of stand-alone and supplemental fixation for anterior cervical discectomy and fusion. Med Devices (Auckl) 2016 9:223-30.



12. Voronov LI, Siemionow KB, Havey RM, et al. Biomechanical evaluation of DTRAX((R)) posterior cervical cage stabilization with and without lateral mass fixation. Med Devices (Auckl) 2016 9:285-90.


13. Maulucci CM, Sansur CA, Singh V, et al. Cortical bone facet spacers for cervical spine decompression: effects on intervertebral kinetics and foraminal area. J Neurosurg Spine 2016 24:69-76.


14. Bohlman HH, Emery SE, Goodfellow DB, et al. Robinson anterior cervical discectomy and arthrodesis for cervical radiculopathy. Long-term follow-up of one hundred and twenty-two patients. J Bone Joint Surg Am 1993 75:1298-307.


15. Wright IP, Eisenstein SM. Anterior cervical discectomy and fusion without instrumentation. Spine 2007 32:772. -4. discussion 5.


16. Bolesta MJ, Rechtine GR 2nd, Chrin AM. Three- and four-level anterior cervical discectomy and fusion with plate fixation: a prospective study. Spine 2000 25:2040. -4. discussion5-6.


17. Kelly BP, Bennett CR. Design and validation of a novel Cartesian biomechanical testing system with coordinated 6DOF real-time load control: application to the lumbar spine (L1-S, L4-L5). J Biomech 2013 46:1948-54.


18. Panjabi MM. Biomechanical evaluation of spinal fixation devices: I. A conceptual framework. Spine 1988 13:1129-34.


19. Panjabi MM, Abumi K, Duranceau J, et al. Biomechanical evaluation of spinal fixation devices: II. Stability provided by eight internal fixation devices. Spine 1988 13:1135-40.


20. Crawford NR, Dickman CA. Construction of local vertebral coordinate systems using a digitizing probe. Technical note. Spine 1997 22:559-63.


22. Bertalanffy H, Eggert HR. Clinical long-term results of anterior discectomy without fusion for treatment of cervical radiculopathy and myelopathy. A follow-up of 164 cases. Acta Neurochir (Wien) 1988 90:127-35.

23. Buttermann GR. Anterior cervical discectomy and fusion outcomes over 10 years: a prospective study. Spine 2018 43:207-14.

24. Vleggeert-Lankamp CLA, Janssen TMH, van Zwet E, et al. The NECK trial: effectiveness of anterior cervical discectomy with or without interbody fusion and arthroplasty in the treatment of cervical disc herniation; a double-blinded randomized controlled trial. Spine J 2019 19:965-75.


25. Kaiser MG, Haid RW Jr, Subach BR, et al. Anterior cervical plating enhances arthrodesis after discectomy and fusion with cortical allograft. Neurosurgery 2002 50:229. -36. discussion 36-8.


26. Wang JC, McDonough PW, Endow KK, et al. Increased fusion rates with cervical plating for two-level anterior cervical discectomy and fusion. Spine 2000 25:41-5.


27. Lim S, Kesavabhotla K, Cybulski GR, et al. Predictors for airway complications following single- and multilevel anterior cervical discectomy and fusion. Spine 2017 42:379-84.


28. Thompson SE, Smith ZA, Hsu WK, et al. C5 palsy after cervical spine surgery: a multicenter retrospective review of 59 cases. Global Spine J 2017 7:64S-70S.



29. Sembrano JN, Mehbod AA, Garvey TA, et al. A concomitant posterior approach improves fusion rates but not overall reoperation rates in multilevel cervical fusion for spondylosis. J Spinal Disord Tech 2009 22:162-9.


30. Leasure JM, Buckley J. Biomechanical evaluation of an interfacet joint decompression and stabilization system. J Biomech Eng 2014 136:0710101-8.



31. Pal GP, Routal RV. A study of weight transmission through the cervical and upper thoracic regions of the vertebral column in man. J Anat 1986 148:245-61.






























