INTRODUCTION
Relatively rare compared to its intracranial counterpart, spinal meningioma accounts for 1.2% of all meningiomas of the central nervous system and 25% of all spinal cord tumors6). Spontaneous intraspinal bleeding associated with spinal meningioma has rarely been reported, whereas spinal subdural hematoma has occasionally been reported in patients with bleeding diathesis and spinal injury, even after lumbar puncture5,9).
We report a rare case of a 58-year-old woman with thoracolumbar spinal meningioma accompanied by intraspinal subdural hematoma that developed after a spinal nerve block.
CASE REPORT
A 58-year-old woman was admitted to the hospital with severe back pain accompanied with nausea and vomiting. Because of the back pain, she underwent a selective nerve root block in the lumbar spine at another hospital. After the spinal procedure, the patient felt more severe back pain, newly developed pain, and weakness in her right leg. She had intact voiding sensation and no difficulty voiding at the time of admission. The paraparesis was checked during a neurologic examination. In the right lower extremity, ankle dorsiflexion (Gr III/IV+) and toe flexion and extension (IV-/IV+) weaknesses were prominent compared with those of the left lower extremity. Deep tendon reflex was absent on the right knee, and the "extensor toe sign" was negative bilaterally, without clonus. Sensory impairment to the pain was noted on the L5 dermatome of the right calf.
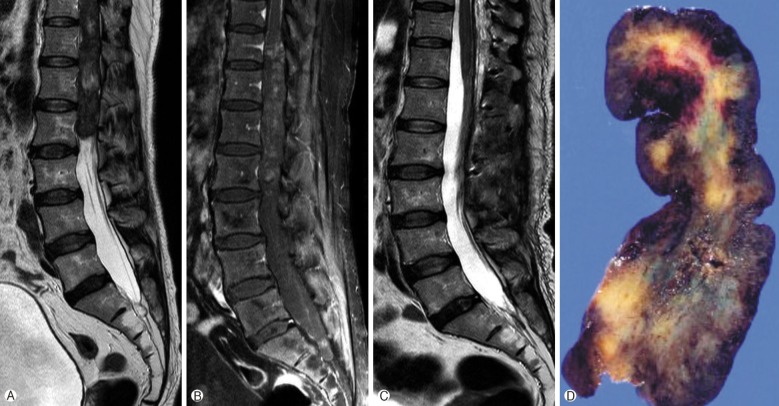
An MRI revealed a ventrally located intradural mass extending from T10 to L2; the mass was iso- to hypointense on T1-, and the hyperintense portion on the T2-weighted images was compatible to the bleeding inside the tumor. The lesion was heterogeneously enhanced with contrast administration (Fig. 1).
One day after her admission, the patient experienced worsening of her neurological status with weakness of the lower extremities and the loss of anal tone and voiding sensation. She underwent an emergency operation with high-dose methylprednisolone therapy.
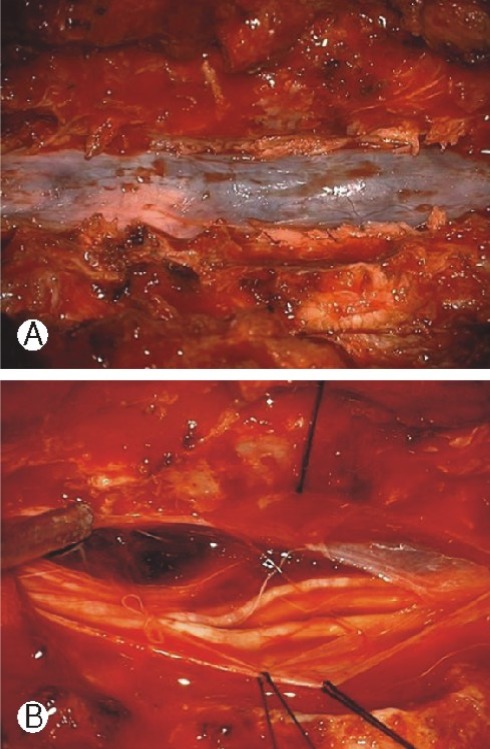
A laminectomy from the T10 to L2 levels was performed using a posterior midline approach. After the dura was opened, the mass was exposed to be dark and oil-colored, and a chocolate-colored subdural hematoma was observed above and below the mass (Fig. 2). The mass was not connected to the nerve roots. However the mass was adhered to the ventral dura. Therefore, the mass was removed en bloc under an operating microscope, with coagulation of the dural attachment. A frozen section biopsy of the mass was performed, and under microscopic examination, it was compatible with a meningioma. The dura was closed in a watertight fashion with bleeding control. Although no wave was observed on intraoperative monitoring at the beginning of the surgery, some wave was observed at the end of surgery. The pathologic report confirmed the meningioma was a fibrous type WHO grade I/III tumor.
During the postoperative course, the status of motor and sensory was improved. The patient felt bladder fullness and anal contraction after 2 postoperative days. She was transferred to the department of rehabilitation for an intense rehabilitation program. Two months post-surgery, the patient was freed from the Foley catheter, and her urinary symptoms recovered without urinary incontinence, frequency, residual urine sensation, and urgency.
DISCUSSION
Intraduralextramedullary spinal cord tumors are predominantly meningiomas or peripheral nerve sheath tumors, whereas less common occurrences include paragangliomas, metastases, lipomas, spinal nerve sheath myxomas, sarcomas, and vascular tumors1). Meningiomas are dural-based tumors that arise from arachnoid cap cells and, consequently, can be found in any location that dura is present. Approximately 25% of all primary spinal cord tumors are meningiomas. More than 80% of spinal cord meningioma patients are women, and 80% of these tumors occur in the thoracic region. In men, spinal cord meningiomas are equally distributed between the cervical and thoracic cords. Overall, 15% of spinal cord meningiomas occur in the cervical spine, 81% in the thoracic spine, and 4% in the lumbar spine. Most meningiomas are slow-growing low-grade tumors (WHO grade I)1,4).
The clinical symptoms of spinal meningioma depend on the tumor location, with respect to the spinal cord and nerve roots. Common presenting symptoms include back pain (70%), motor weakness (60%), sensory change (40%), and incontinence (40%)1,7).
Acute subdural spinal hematoma has been reported to occur rarely; however, after it occurs, neurological deficits caused by cord compression from a hematoma may occur. The typical clinical presentations of acute subdural spinal hematoma are the sudden onset of spinal or radicular pain and myelopathy, including paraplegia and sensory and autonomic dysfunction. An MRI scan is quite useful for the early detection of the hemorrhage and for establishing the correct diagnosis. Nontraumatic acute spinal subdural hematomas have been reported in cases of coagulation abnormalities and iatrogenic causes, such as spinal punctures. Swann reported that spinal tumors, particularly in the region of the caudaequina and conus medullaris, can cause spontaneous spinal subarachnoid hemorrhage, although this occurrence is uncommon. The mechanism of hematoma formation is believed to be related to the vulnerability to trauma of the blood vessels surrounding these tumors8). Most cases of hemorrhage associated with meningiomas are extratumoral and subarachnoid, whereas subdural, intracerebral and intratumoral hemorrhage cases are reported less frequently2,5,10). In our case, the patient was considered to have subdural and intratumoral hemorrhages after an epidural block that most likely caused the subdural space injury. The injected particle into tissue including vascular structures around tumor could induced the hemodynamic changes and the hemodynamic changes may cause the intratumoral hemorrhage.
In cases in which an acute subdural hematoma is formed either from iatrogenic or coagulation abnormalities, early surgical treatment is always important for the prognosis when the patient's neurological status progressively deteriorates2). The treatment of spinal meningiomas is predominantly surgical. A recent study that analyzed 131 patients who underwent surgical resection of a spinal meningioma found that the spinal meningioma complete resection rate was 97%, and the procedure was generally considered to be effective following bipolar coagulation6).
When the neurological status of a patient is not complicated, favorable outcomes are achieved in most cases. However, progressive spinal cord compression by spinal meningioma with neurologic deterioration may result in permanent neurologic deficit even after successful surgery7). The long-term functional outcome of spinal meningioma treatment has been reported to be correlated with the neurological deficit severity at the time of presentation and the symptomduration3).
CONCLUSION
Although spinal meningioma with bleeding has been reported, reports of spinal meningioma associated with iatrogenic causes, such as spinal nerve block, are rare to the best of our best knowledge. If a severe neurological deterioration occurs after a spinal procedure, an intra-spinal mass with hemorrhage could be considered.