Cement Augmentation of Vertebral Compression Fractures May Be Safely Considered in the Very Elderly
Article information
Abstract
Objective
The objective of the current study was to perform a retrospective review of a national database to assess the safety of cement augmentation for vertebral compression fractures in geriatric populations in varying age categories.
Methods
The 2005–2016 National Surgical Quality Improvement Program databases were queried to identify patients undergoing kyphoplasty or vertebroplasty in the following age categories: 60–69, 70–79, 80–89, and 90+ years old. Demographic variables, comorbidity status, procedure type, provider specialty, inpatient/outpatient status, number of procedure levels, and periprocedure complications were compared between age categories using chi-square analysis. Multivariate logistic regressions controlling for patient and procedural variables were then performed to assess the relative periprocedure risks of adverse outcomes of patients in the different age categories relative to those who were 60–69 years old.
Results
For the 60–69, 70–79, 80–89, and 90+ years old cohorts, 486, 822, 937, and 215 patients were identified, respectively. After controlling for patient and procedural variables, 30-day any adverse events, serious adverse events, reoperation, readmission, and mortality were not different for the respective age categories. Cases in the 80- to 89-year-old cohort were at increased risk of minor adverse events compared to cases in the 60- to 69-year-old cohort.
Conclusion
As the population ages, cement augmentation is being considered as a treatment for vertebral compression fractures in increasingly older patients. These results suggest that even the very elderly may be appropriately considered for these procedures (level of evidence: 3).
INTRODUCTION
Vertebral compression fractures are the most common complication of osteoporosis [1]. Population studies have estimated that 8% of adults over 50 years old have osteoporosis of the lumbar spine1 and the age-adjusted incidence of compression fractures is 117 per 100,000 person-years [2]. The US Census Bureau projects that the population aged 65 years and older will double between 2012 and 2050 [3]. The very elderly are particularly vulnerable to compression fractures, as the risk of this condition increases with advancing age [4]. As the population ages, the safety of cement augmentation procedures, such as kyphoplasty and vertebroplasty, in geriatric patients with compression fractures is an important but unanswered question.
Vertebral compression fractures are not all clinically significant, but the nearly 30% of patients who are symptomatic may suffer from debilitating pain and progressive kyphosis, causing a decrease in quality of life that has been shown to be more severe than geriatric hip, forearm, or humerus fractures [5,6]. Although compression fractures are typically managed conservatively, cement augmentation of vertebral compression fractures is a viable treatment modality for patients with severe and intractable pain [7]. Both kyphoplasty and vertebroplasty have been shown to have beneficial effects in selected patient populations [8]. Perioperative complications following these procedures include medical/anesthetic complications, cement leakage, new vertebral fracture, and infection [9,10].
Studies evaluating cement augmentation of compression fractures have shown that older geriatric patients do benefit from cement augmentation procedures [11,12]. DePalma et al. [11] followed 123 vertebroplasty patients and found similar rates of recurrent fracture between nonagenarian and younger patients. Kamei et al. [12] retrospectively reviewed the postoperative courses of 130 vertebroplasty patients and found that nonagenarian patients had similar 1-year survival rates to younger patients. Both of these studies were limited by relatively small patient numbers, single-center design, and failure to assess short-term postprocedure adverse outcomes.
As the population ages, cement augmentation is being considered for increasingly older patients. The very elderly are at increased risk for compression fractures and are perhaps the population with the most to gain from an expedited recovery after a fracture. The aim of the current study was to utilize the large, multicenter National Surgery Quality Improvement Program (NSQIP) database to assess the risk of postprocedural complications and adverse events in vertebroplasty and kyphoplasty cases performed in very elderly patients.
MATERIALS AND METHODS
1. Data Source
The NSQIP database aggregates more than 250 variables from representative surgical procedures performed at more than 600 participating institutions. Interrater discordance has been shown to be less than 1.5%, suggesting that this data is highly reliable. The current study utilized data from the 2005–2016 NSQIP databases and was found to be exempt from Human Investigational Committee review based on the fact that all data were deidentified.
2. Study Cohort
Cases of vertebroplasty and kyphoplasty were included in the study population. Cases of vertebroplasty were identified using Current Procedural Terminology (CPT) codes 22510, 22511, 22520, and 22521. Cases of kyphoplasty were identified using CPT codes 22513, 22514, 22523, and 22524. The number of operative levels was determined for each case by counting instances of additional level CPT codes 22512, 22515, 22522, and 22525.
Cases involving concomitant decompression and/or fusion procedures were excluded. Additionally, cases of concomitant vertebroplasty and kyphoplasty were excluded from the study population for simplicity of analysis.
Subcohorts were defined based on age categories of 60–69, 70–79, 80–89, and 90 years old and above were included. For each category, demographics were defined including: age, sex, height, and weight, which were directly abstracted from the NSQIP database. Height and weight data were used to calculate body mass index (BMI) (kg/m2). Preoperative functional status and American Society of Anesthesiologists (ASA) physical status classification are tracked in the NSQIP database, both of which were used to approximate each case’s comorbidity burden prior to operation.
In order to determine provider specialty, cases were also identified as being performed by a neurosurgeon versus an orthopaedic surgeon and compared based on demographic statistics and outcomes. Cases performed by interventional radiologists were excluded from the study cohort due to low case volumes in this dataset resulting in inadequate statistical power for comparison.
Finally, lengths of hospital stay (LOS) in days were also extracted from the dataset. Cases were classified as outpatient if they were discharged less than 24 hours after the procedure, where as those associated with a hospital LOS of greater than 24 hours were classified as inpatient.
3. Postoperative Adverse Outcomes
NSQIP records the occurrence of adverse outcomes for 30-day postprocedure, regardless of the patient’s discharge status. The occurrences of individual adverse outcomes were combined into 3 complication categories: any adverse events (AAEs), serious adverse events (SAEs), and minor adverse events (MAEs).
The occurrence of any of the following constituted a SAE: deep surgical site infection, sepsis, failure to extubate, unplanned reintubation, postoperative renal failure, deep vein thrombosis, pulmonary embolism, cardiac arrest, myocardial infarction, or stroke. The occurrence of any of the following constituted a MAE: superficial surgical site infection, wound dehiscence, pneumonia, urinary tract infection, or postoperative renal insufficiency. The occurrence of a major adverse event or MAE was considered to represent the occurrence of AAE.
NSQIP began recording the number of 30-day readmissions for each case after the year 2011 and the number of 30-day reoperations for each case after the year 2012. These outcomes were separately tabulated and assessed. The analysis of readmission includes 2,453 of the 2,460 cases (99.7% of total cases) and the analysis of reoperation includes 2,395 of the 2,460 cases (86.7% of total cases). Mortality was evaluated separately as an outcome of interest.
4. Statistical Analysis
Patient demographic characteristics, occurrence of adverse events, reoperation rate, readmission rate, and mortality were first compared between age-wise cohorts. Chi-square tests were used to compare sex, functional status, procedure type, and surgical specialty. Analysis of variance (ANOVA) was used to compare BMI and average LOS across groups. Kruskal-Wallis equality-of-populations tests were used to compare ASA physical status classification and number of operative levels across groups.
Multivariate logistic regression models were then constructed to determine the odds ratios of experiencing different categories of adverse events in the age-wise cohorts, using the 60- to 69-year-old cohort as the referent and controlling for sex, BMI, functional status, ASA physical status classification, procedure type, operative specialty and number of operative levels.
The level of significance for all tests was adjusted by Bonferroni correction to reduce the false discovery rate. All statistical analyses were performed using Stata 13 (StataCorp LP, College Station, TX, USA).
RESULTS
1. Study Population
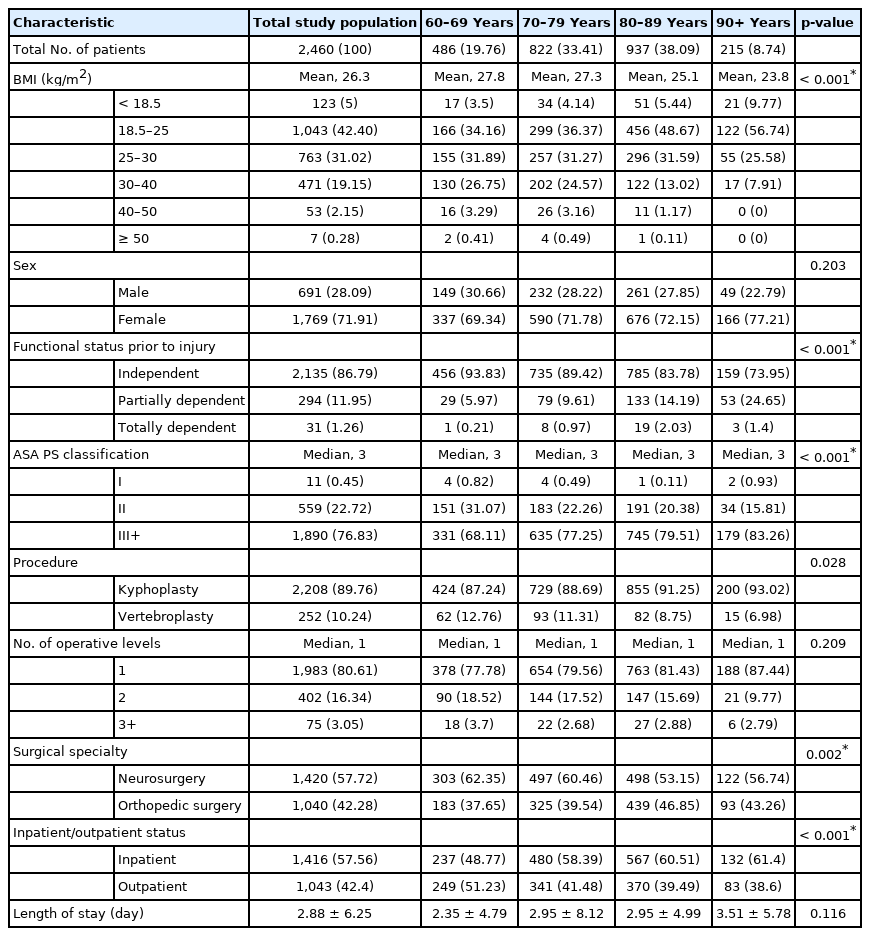
After excluding cases involving concomitant deformity correction procedures, 2,460 cases involving patients over the age of 60 undergoing either vertebroplasty or kyphoplasty were available for analysis. Of these 2,460 patients, 486 were between 60 to 69 years of age; 822 were between 70 and 79; 937 were between 80 and 89; and 215 patients were 90 years of age or older (Fig. 1). Of these procedures, 2,208 of patients (89.8%) underwent kyphoplasty and 252 (10.2%) underwent vertebroplasty.
Patient characteristics are summarized in Table 1. The age groups did not significantly differ in their gender distribution, number of operative levels, or surgical specialty. The age groups did significantly differ in their BMI range, with older cohorts having decreasing mean BMI (p < 0.001 by ANOVA). Further, while all groups had a median ASA physical status of III, the distribution of ASA physical status classification was significantly different between age groups (p < 0.001 by Kruskal-Wallis). The nonagenarian cohort had the highest proportion of cases with ASA physical status classifications III and above (83.3%), while the 60–69 cohort had the lowest proportion of cases with ASA physical status classification III and above (68.1%). The nonagenarian cohort also had the lowest proportion of outpatient cases (38.6%), while the 60–69 cohort had the highest proportion of outpatient cases (51.2%). There was no statistically significant difference in the length of stay between the different age groups. For these tests, the level of significance was set at 0.006 after Bonferroni correction, as 8 potential differences between groups were being tested.
2. Adverse Events
Thirty-day perioperative adverse outcomes are presented by age category in Table 2. For the entire study population, AAE was noted for 6.6%, SAE was noted for 4.3%, and MAE was noted for 3.5%. The rate of reoperation, readmission, and mortality were 3.9%, 10.7%, 1.9%, respectively.
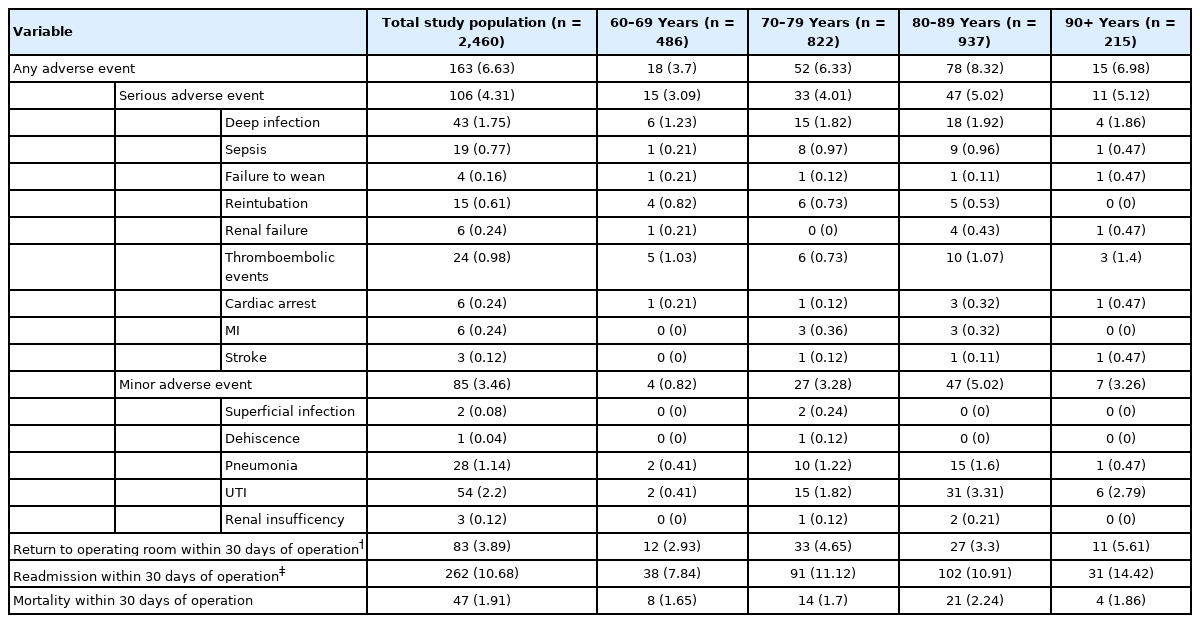
Number of adverse events, return to operating room, readmissions, and mortality for patients of varying age
Multivariate logistic regression models controlling for gender, BMI, functional status, ASA physical status classification, surgical specialty, procedure type, inpatient/outpatient status, and number of operative levels were then fitted on each postoperative adverse outcome (Table 3). The age groups did not significantly differ in odds of experiencing AAE (Fig. 2, shown as an example Forrest plot), SAE, return to operating room, readmission, or mortality within 30 days of index procedure. Cases in the 80–89 years group had an increased odds of experiencing MAE (odds ratio, 5.44; p = 0.002) compared to cases in the 60–69 years group. There were no other significant differences in MAE between the 60–69 years group and other groups. The level of significance for these tests was adjusted to 0.0027 after Bonferroni correction as a total of 18 comparisons were made.
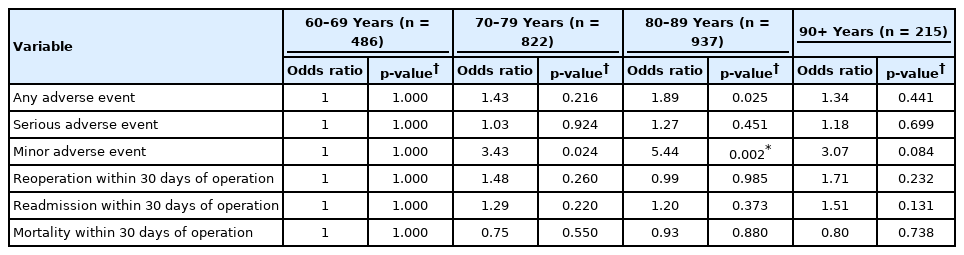
Multivariate odds ratio for adverse events, return to operating room, readmissions, and mortality for patients of varying age (n = 2,460)
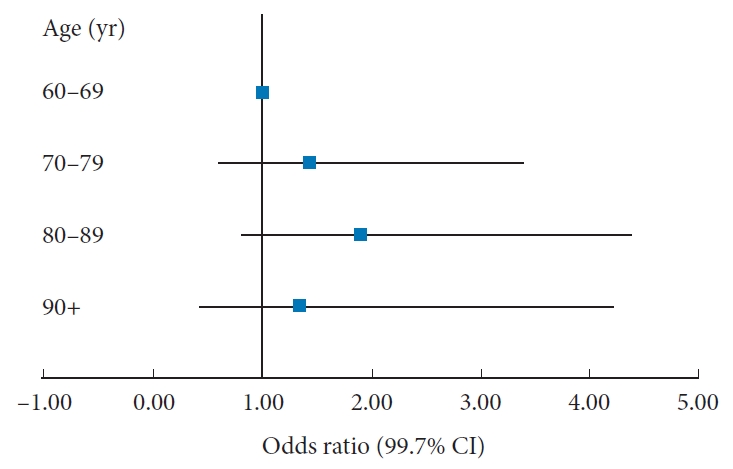
Odds ratios for experiencing a postoperative adverse event by age group. Controlled for sex, body mass index, functional status, American Society of Anesthesiologists physical status classification, surgical specialty, procedure type, inpatient/outpatient status, and number of operative levels. CI, confidence interval.
A subanalysis was also done based on provider specialty (orthopaedic vs. neurosurgery) and no difference in perioperative outcomes was noted when using multivariate analyses.
DISCUSSION
Kyphoplasty and vertebroplasty are procedures that are routinely considered in geriatric patients with symptomatic compression fractures [9,13-17]. A prior study in the United States investigating the time interval for osteopenia progression to osteoporosis in older women found that the interval decreased with age [18]. Given the percutaneous nature of such cement augmentation procedures, they are being offered to increasingly medically complex and older patients who may not tolerate bracing and/or would be deemed poor candidates for other interventions [9,19,20]. Therefore, an analysis of patient characteristics predictive of poor outcomes would certainly be beneficial to practitioners.
The current study evaluates almost 2,500 patients undergoing these procedures. Following cement augmentation procedures, the rates adverse events were as follows: AAE, 6.6%; SAE, 4.3%; and MAE, 3.5%. The rates for reoperation were 3.9%, readmission 10.7%, and mortality 1.9%. Patients undergoing cement augmentation procedures with orthopedic surgeons and neurosurgeons had similar morbidity profiles.
AAE after cement augmentation procedures was noted to occur in 6.6% of the current study population and was within the ranges reported by prior studies. For instance, the meta-analysis of Lee et al. included data from 121 studies and noted overall complication rates of 3.6% after kyphoplasty and 5.2% after vertebroplasty. Goz et al. queried the NIS database and calculated rates of adverse events to be 4.66% after kyphoplasty and 6.05% after vertebroplasty. Toy et al. used the NSQIP database to analyze outcomes in a smaller cohort of 850 patients and found a 30-day AAE of 9.5% [9,13,15-17,21,22]. The reoperation rate (3.9%) was also similar to that previously reported by Choo et al. [23] who studied 2,433 patients in the NSQIP database and noted a rate of 3.6%. Finally, mortality (1.9%) was also within the range reported by prior studies. A meta-analysis by Taylor et al. [9] included data from 43 studies and reported a 3.2% mortality rate and Toy et al. [21] used the NSQIP database to analyze outcomes in a smaller cohort of 850 patients and found a 30-day mortality rate of 1.5%.
The above-noted similarities in the adverse outcomes presented in the current study with previously published studies serve to confirm that the current study population is in line with prior literature. The novel analysis presented here was the assessment of perioperative outcomes according to age category. After controlling for patient and procedural variables, the rates of 30-day AAEs, SAEs, reoperation, readmission, and mortality were no different for any the different age categories. The only notable disparity was that cases in the 80- to 89-year-old cohort were at increased risk of MAEs compared to cases in the 60- to 69-year-old cohort, but this findings was not evident with the other groups, including that 90+-year-old group. The increased odds of MAE in the 80- to 89-year-old cohort seems to be driven by the increased rate of pneumonia and urinary tract infections in this age group compared to that of the 60- to 69-yearold cohort. In a prior epidemiologic study of community-acquired pneumonia in the elderly, the incidence of pneumonia was found to increase with age until the 90+ cohort [24]. Similar epidemiologic data is lacking in nosocomial pneumonia, which is germane to postoperative patients [25]. Likewise, the incidence of urinary tract infections has been shown to increase with age, in prior studies [26].
The safety of lumbar spine surgery in general has previously been evaluated for older patient cohorts. A study of over 2,320 patients over the age of 80 undergoing lumbar spine surgery found that 16% of individuals experienced a complication with a 0.4% death rate and a 6.3% readmission rate within 30 days [27]. Another large study of over 10,000 lumbar decompression and fusion patients over 80 years old reported a 12.6% complication rate and 0.31% death rate 30 days after the index procedure [28].
The current study looked specifically at cement augmentation procedures and in the even more elderly (i.e., 90+ age bracket), as opposed to 80+ years old previously reported by others [29]. Patients over 90 years old had a AAE rate of 7.0%, readmission rate of 14.4%, and mortality rate of 1.9%. These valueswere similar to those observed in the 60- to 69-year-old cement augmentation group.
To our knowledge, there are no previous studies comparing the outcomes of orthopedic surgeons and neurosurgeons performing cement augmentation procedures using a large cohort of patients. We found no difference between the outcomes of the various populations with regards to operator specialty training, consistent with prior studies indicating no differences between the groups [30]. Similar results have been reported with regards to elective lumbar and cervical spine procedures as well [31-33].
There are several limitations to the current study. First of all, the NSQIP database does not provide outcome data beyond the 30-day postoperative period, so morbidity and mortality beyond this window cannot be assessed. Second, NSQIP does not capture the time from diagnosis to operation. Third, NSQIP does not provide radiographic parameters or any patient-reported outcomes. Finally, NSQIP does not track procedure-specific complications like extravasation of cement, neurological injury, or refracture. Despite these weaknesses, the strengths of the current study include the large number of patients analyzed and the high-quality, multicenter data obtained from the NSQIP database.
CONCLUSION
Overall, the current analysis of 2,460 patients identified in the NSQIP database who had undergone cement augmentation procedures suggests that these procedures can be safely performed in appropriately selected very elderly patients. Surgeons may use this information to appropriately counsel individuals with painful vertebral compression fractures who may be considering kyphoplasty and vertebroplasty.
Notes
The authors have nothing to disclose.
Acknowledgements
Research reported in this publication was supported by the National Institute on Aging of the National Institutes of Health under Award Number T35AG049685.