Are Lumbar Fusion Guidelines Followed? A Survey of North American Spine Surgeons
Article information
Abstract
Objective
To evaluate the use of guidelines for lumbar spine fusions among spine surgeons in North America.
Methods
An anonymous survey was electronically sent to all AO Spine North America members. Survey respondents were asked to indicate their opinion surrounding the suitability of instrumented fusion in a variety of clinical scenarios. Fusion indications in accordance with North America Spine Society (NASS) guidelines for lumbar fusion were considered NASS-concordant answers. Respondents were considered to have a NASS-concordant approach if ≥ 70% (13 of 18) of their answers were NASS-concordant answers. Comparisons were performed using bivariable statistics.
Results
A total of 105 responses were entered with complete data available on 70. Sixty percent of the respondents (n = 42) were considered compliant with NASS guidelines. NASS-discordant responses did not differ between surgeons who stated that they include the NASS guidelines in their decision-making algorithm (5.10 ± 1.96) and those that did not (4.68 ± 2.09) (p = 0.395). The greatest number of NASS-discordant answers in the United States. was in the South (5.75 ± 2.09), with the lowest number in the Northeast (3.84 ± 1.70) (p < 0.01). For 5 survey items, rates of NASS-discordant answers were ≥ 40%, with the greatest number of NASS-discordant responses observed in relation to indications for fusion in spinal deformity (80%). Spine surgeons utilizing a NASS-concordant approach had a significant lower number of NASS-discordant answers for synovial cysts (p = 0.03), axial low back pain (p < 0.01), adjacent level disease (p < 0.01), recurrent stenosis (p < 0.01), recurrent disc herniation (p = 0.01), and foraminal stenosis (p < 0.01).
Conclusion
This study serves an important role in clarifying the rates of uptake of clinical practice guidelines in spine surgery as well as to identify barriers to their implementation.
INTRODUCTION
The prevalence of lumbar pain due to spinal disorders is increasing around the world, and instrumented fusion procedures are widely used as an option of treatment [1-3]. Despite the increasing utilization of instrumented fusion for the treatment of lumbar pathology, there is still a lack of medical literature detailing concrete fusion indications and studies validating guidelines as predictors of outcomes [4-6]. This is largely secondary to heterogeneity in clinical decision-making amongst spine surgeons and surgical indications in lumbar spine pathology management.
Improving the quality of care under a patient-centered perspective is an effort that drives medical initiatives like the construction of evidence-based medical (EBM) guidelines. The North American Spine Society (NASS), in an attempt to improve surgical outcomes and patient care, published diagnosis and indications for lumbar fusion as well as qualifying criteria [7]. These guidelines provide a tool to guide clinical decision-making in the treatment of lumbar pathology.
This study is an initiative to evaluate and gain insight into the use of the NASS criteria for indications of lumbar spine fusions among spine surgeons in North America. The results of this survey aim to inform and contribute to future discussions of the applicability of EBM guidelines in assisting surgical decision-making for lumbar spine fusions. The main objective of this study is to evaluate the use of EBM guidelines for lumbar spine fusions among spine surgeons in North America.
MATERIALS AND METHODS
An online electronic survey was generated using Qualtrics software (Provo, UT, USA). The survey questions consisted of 18 clinical vignettes to elucidate participating surgeons’ indications for lumbar spine fusion. Each clinical vignette was framed and discussed by a panel of senior neurosurgeons and spine surgeons at a quaternary university hospital and intended to evaluate the acceptance of the specific indications for arthrodesis published by the NASS as a coverage policy for lumbar fusions after conducting a comprehensive literature review by multidisciplinary experts [7]. All panel members agreed that each clinical vignette had a clear indication for or against lumbar spine fusion based on the NASS guidelines [7].
The survey was available in English, participation was voluntary, without remuneration, anonymized, and was distributed electronically to spine surgeon members of the AO Spine North America (AOSNA). The study was approved by the research committee of the AOSNA and distributed through an electronic invitation that was sent on 4 separate occasions between July and August 2020 to the spine surgeons. The introductory electronic communication with the respondents consisted of an email specifying study objectives, the survey structure, and an online link to the Qualtrics platform (Supplementary material 1). In an effort to eliminate bias, none of the surgeons involved in the study panel filled out the survey.
The first part of the survey consisted of demographic questions about the spine surgeon residency specialty, fellowship training, number of years in practice, and the approach to indicate a lumbar spine fusion (Supplementary material 1). The second part of the survey was based on 18 items with clinical vignettes and radiological images, followed by whether or not the surgeon felt a spine fusion was indicated in the treatment of the patient (Supplementary material 3). The major outcome investigated was the number of answers (fusion indications) in accordance with the NASS guidelines (NASS-concordant answer), assessed with the 18 clinical items of the survey [7]. The participating surgeon was considered to have a NASS-concordant approach if ≥ 70% (13 of 18) of their answers to the survey cases were NASS-concordant. Due to the study design, the study protocol was initially exempted from Institutional Review Board approval.
The survey data was exported from Qualtrics into a tabulated Microsoft Excel file, and data were analyzed with IBM SPSS Statistics ver. 22.0 (IBM Co., Armonk, NY, USA). Continuous variables were reported as means and standard deviation, and categorical variables were reported as frequency and percentage. Differences in frequencies between the groups of responses analyzed were evaluated using a chi-square test and the Fisher exact test based on frequency table cell count. The unpaired 2-tailed Student t-test and the Mann-Whitney U-tests for nonparametric data were used to compare continuous variables as appropriate based on assumptions of normality. A p-value of < 0.05 was set for statistical significance.
RESULTS
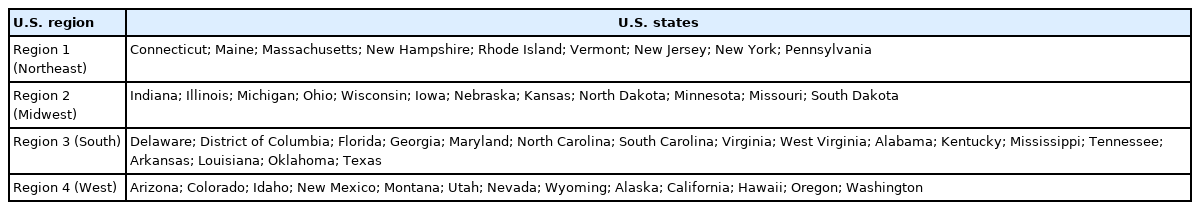
A total of 515 AOSNA members were invited to participate in the survey, 105 responses were received, 35 were excluded due to an incomplete survey, thereby 70 were included in the final analysis. Ninety-one percent (n = 64) of the survey participants practice in the United States (US). Respondents were distributed across 4 provinces in Canada and 27 states in the US, in which the greatest number of responses was in Pennsylvania (n = 7). The 2 regions of the US with the most number of responses were the South (n = 20, 28.6%), followed by the Northeast (n = 19, 27.1%) [8] (Table 1, Fig. 1).

Geographic distribution of survey respondents. The color gradient represents the number of answers per state/province.
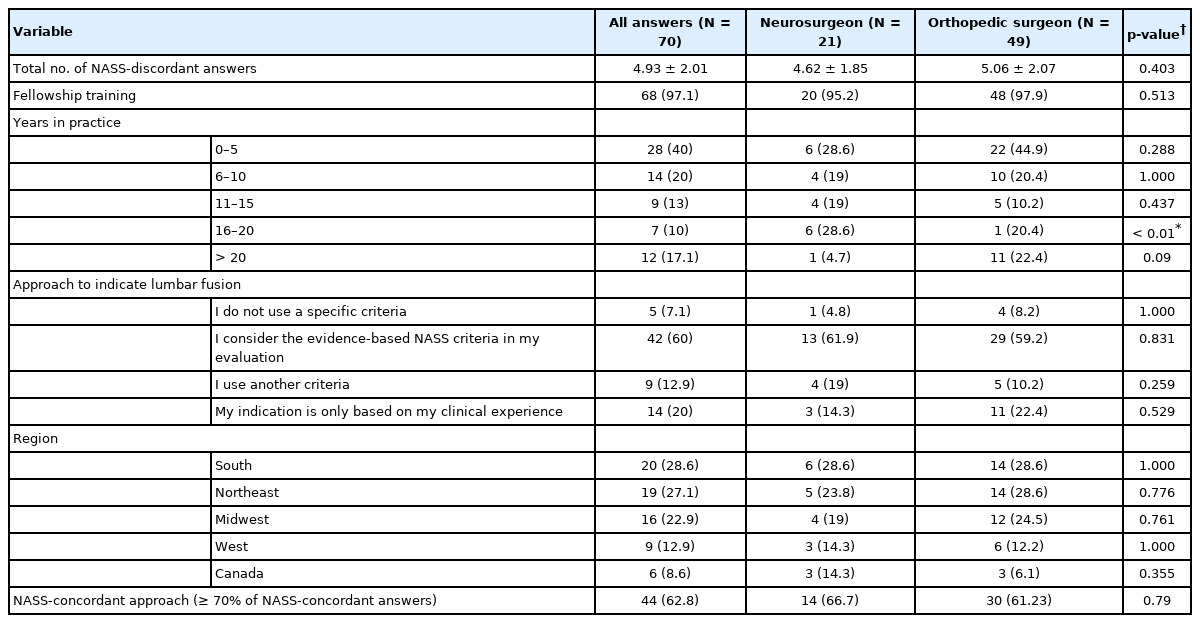
The majority of the responses were from orthopedic surgeons (n = 49, 70 %), 68 participants (97.1%) stated that they have fellowship training in spine surgery, and 41 participants (58.5%) practice in an academic medical center. Out of the 70 participants, 28 (40%) have less than 5 years of clinical practice as a spine surgeon, followed by 14 respondents (20%) that are in practice between 6–10 years. The majority of the spine surgeons (n = 42, 60%) stated that they follow the EBM NASS guidelines in their evaluation of the lumbar fusion indication [7]. Twenty-eight participants reported that the NASS guidelines are not considered in their evaluation of fusion indication; of those 28 responses, 14 (20%) utilize arthrodesis indications based only on their clinical experience, 9 (12.9%) use other criteria, and 5 (7.1%) do not use a specific criteria to indicate a lumbar fusion.
There was no statistical difference in the mean number of NASS-discordant answers between the group of neurosurgeons (4.62 ± 1.85) and the orthopedic surgeons (5.06 ± 2.07) (p = 0.403). The only significant difference between the 2 groups of specialties is the number of respondents who have 16–20 years in practice (p < 0.01), no other variable considered in this survey, was significantly different between the neurosurgery and the orthopedic group (p > 0.05) (Table 2).
The group of participants who answered that they use the NASS criteria in their clinical evaluation was compared with the respondents who answered they do not use the NASS criteria. The mean number of NASS-discordant answers were not significantly different between the group who consider the NASS criteria (5.10 ± 1.96) with the group who do not consider it (4.68 ± 2.09) (p = 0.395). All the other variables compared between both groups were also not significantly different (p > 0.05) (Table 3).

Comparison between the respondents who stated that consider the NASS criteria in their lumbar fusion indication algorithm and the respondents who do not consider
The number of NASS-discordant answers was only significantly different when the regions analyzed were compared (p < 0.01). The region associated with the greatest number of NASS-discordant answers in the US was the South (5.75 ± 2.09), while the region with the lowest number was the Northeast (3.84 ± 1.70) (Table 4, Fig. 2). The comparison between the group of respondents who were considered to have a NASS-concordant approach (≥ 70% of NASS-concordant answers) with the group who have a NASS-discordant approach also confirmed the association of participants from the South with a NASS-discordant approach (p = 0.01) and participants from the Northeast with a NASS-concordant approach (p = 0.02) (Table 5). The mean number of NASS-discordant answers of the spine surgeons who had most of their practice in an academic medical center (4.63 ± 1.75) was also compared with the ones who had in the private practice (5.34 ± 2.30), and they were not statistically different (p = 0.148).
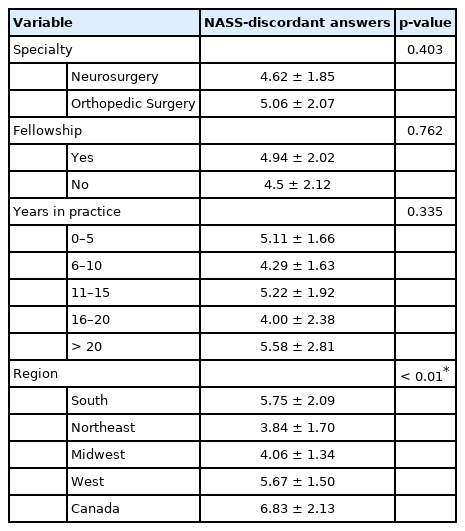
Analysis of the number of NASS-discordant answers stratified by specialty, fellowship training, years in practice, and region
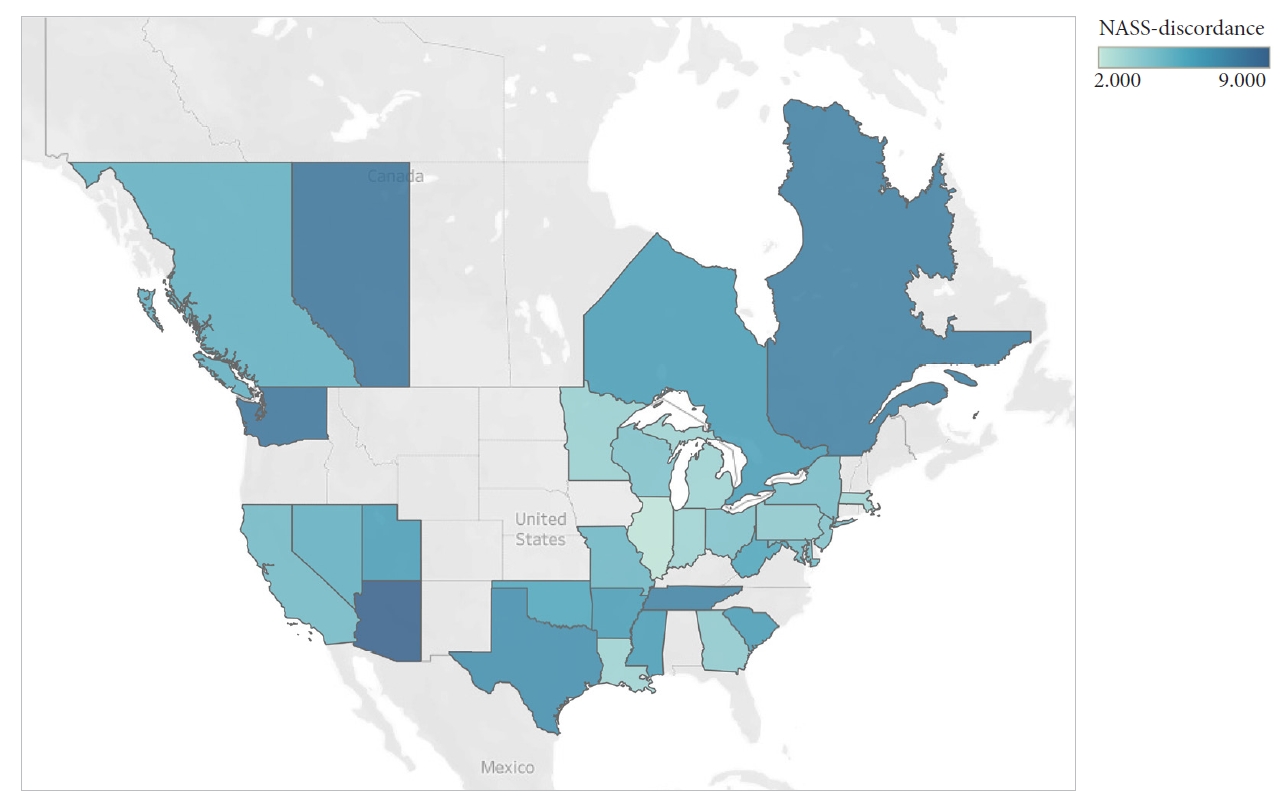
Geographic distribution of NASS-discordant answers. The color gradient represents the number of NASS-discordant answers per state/province. NASS, North America Spine Society.
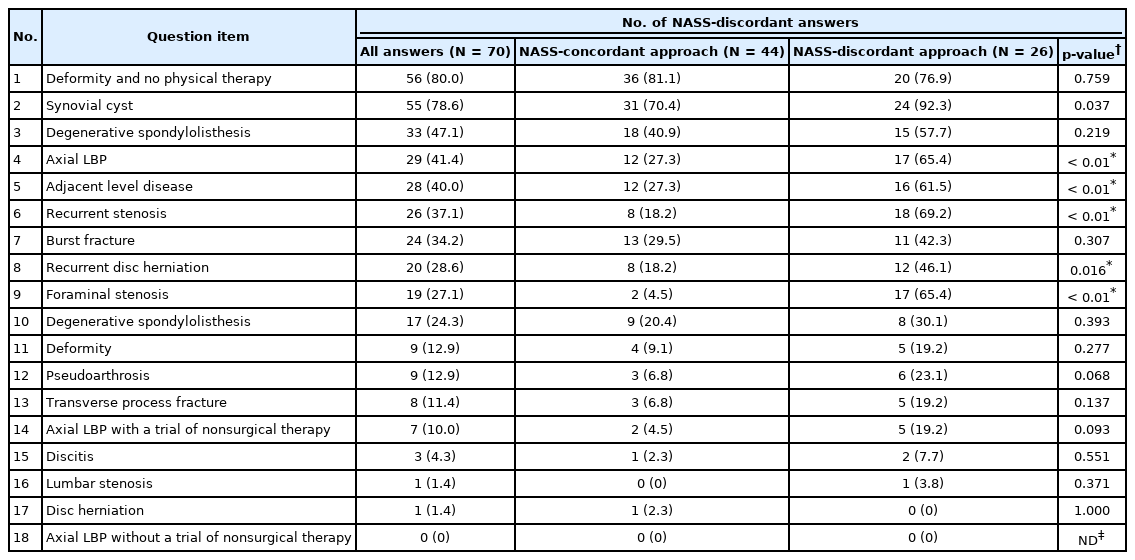
The specific items of the survey that the spine surgeons did not agree were also evaluated. Five items of the survey had an average of NASS-discordant answers ≥ 40% (Table 6). The question item with the greatest number of NASS-discordant responses was the indication of fusion in cases of deformity (80%), followed by synovial cysts (78.6%), degenerative spondylolisthesis (47.1%), axial lumbar pain (41.4%), and adjacent level disease (40%). When the answers were stratified by the respondents who had an overall NASS-concordant approach (≥ 70% NASS-concordant answers in the survey), the items with the greatest number of NASS-discordant answers were the same. Spine surgeons utilizing a NASS-concordant approach had a significantly lower number of NASS-discordant responses in comparison with respondents utilizing a NASS-discordant approach in the following items: synovial cysts (p = 0.03), axial low back pain (LBP) (p < 0.01), adjacent level disease (p < 0.01), recurrent stenosis (p < 0.01), recurrent disc herniation (p = 0.01), and foraminal stenosis (p < 0.01) (Table 6).
DISCUSSION
Spinal fusion utilization, frequency, and hospital charges in the US have been increasing disproportionately compared to other inpatient surgical procedures [3]. Despite this increase in utilization, outcomes in patients undergoing lumbar fusion greatly vary [9-11]. As the armamentarium of lumbar fusion options for LBP grows [3,12], an evidence-based criteria for which spinal pathology to perform fusion on must be established to address the wide variability in treatment and technique. This study attempts to evaluate the role of the NASS criteria in surgical decision-making.
Establishing evidence-based surgical criteria in practice is a necessary part of unifying outcomes and controlling quality in surgical specialties. For instance, even with strong evidence in the literature supporting beneficial outcomes in patients undergoing decompression and fusion for degenerative spondylolisthesis [13-15], there may be non-uniform decision-making by surgeons when addressing these patients [16].
In our experience, it was noted that 60% of surgeons surveyed utilize NASS criteria in surgical decision-making. Despite 40% of surgeons stating they do not consider NASS criteria in surgical decision-making, there was no overall statistically significant difference in the percentage of NASS-concordant answers between surgeons who consider NASS guidelines in decision-making versus those who do not [7]. This was interesting to the authors as it may represent that the NASS criteria serve as a valuable summary or representation of evidenced-based medicine in lumbar spine fusion. Even surgeons who do not consciously use NASS guidelines in decision-making, but indicate surgery based on their understanding of literature, clinical experience, and training, have a similar concordance with the criteria as those surgeons who consider NASS in their surgical indication.
Interestingly, surgeon experience, fellowship training, academic setting, and specialty did not affect the use of NASS guidelines in surgical decision-making, neither the adoption of a NASS-concordant approach [7]. This is contrary to the study Irwin et al. [16], showing that both younger surgeons and orthopedic surgeons exhibited different surgical management strategies, leading to higher fusion rates.
When examining the geographic distribution of NASS-concordant decision-making, this study noted a statistically significant difference in NASS-concordant answers based on region. The Northeast had the lowest mean number of NASS-discordant responses, while the South had the highest mean number of NASS-discordant answers in the US. This regional variability was interesting, given the fact that it seems to correlate with the incidence of surgical treatment of lumbar degenerative disease [3]. As the treatment incidence rises, weaker concordance with EBM criteria such as the NASS criteria may be seen.
Finally, when examining the NASS-concordant approach versus the NASS-discordant approach to surgical management, we noted several pathologies with significant differences in management. NASS concordance was significantly greater in synovial cyst, axial LBP, adjacent level disease, recurrent stenosis, recurrent disc herniation, and foraminal stenosis when comparing surgeons who actively use NASS criteria versus those who do not. Prospectively examining differences in outcome in these groups of patients would be beneficial in assessing NASS criteria as a tool to improve surgeons’ outcomes since these pathologies showed the greatest differences in management decisions between the 2 groups of surgeons.
This study is not without limitations. The current study aimed to compare NASS-concordant versus NASS-discordant responses to spinal indications; however, the indications based upon the NASS guidelines are not solely based on level I evidence. Responses to each clinical vignette may have been biased given the survey’s electronic nature and that a participant can easily compare their responses to NASS guidelines online [7]. We attempted to mitigate this bias by anonymizing each participant. We identified a regional disparity in the study, although this may have been limited by the survey’s response rate of each region. In an attempt to mitigate any regional institutional bias, none of the authors participated in the survey. The demographic information was self-declared by the participants. Lastly, the small sample size and regional distribution may not necessarily correlate with actual regional practices.
CONCLUSION
NASS criteria is a set of EBM guidelines pertaining to lumbar fusion decision-making. When surveying 70 AOSNA members, 60% use the NASS criteria in their decision-making algorithm. Overall, experience, training, specialty did not affect NASS concordance in decision-making. However, geographical differences were seen in survey results. In addition, NASS criteria was met more frequently by surgeons utilizing a NASS-concordant approach for pathology such as synovial cyst, axial LBP, adjacent level disease, recurrent stenosis, recurrent disc herniation, and foraminal stenosis. These pathologies may serve as starting points for further investigation of outcomes associated with NASS criteria and the usefulness of its implementation.
Notes
The authors have nothing to disclose.
SUPPLEMENTARY MATERIALS
Supplementary materials can be found via https://doi.org/10.14245/ns.2142136068.