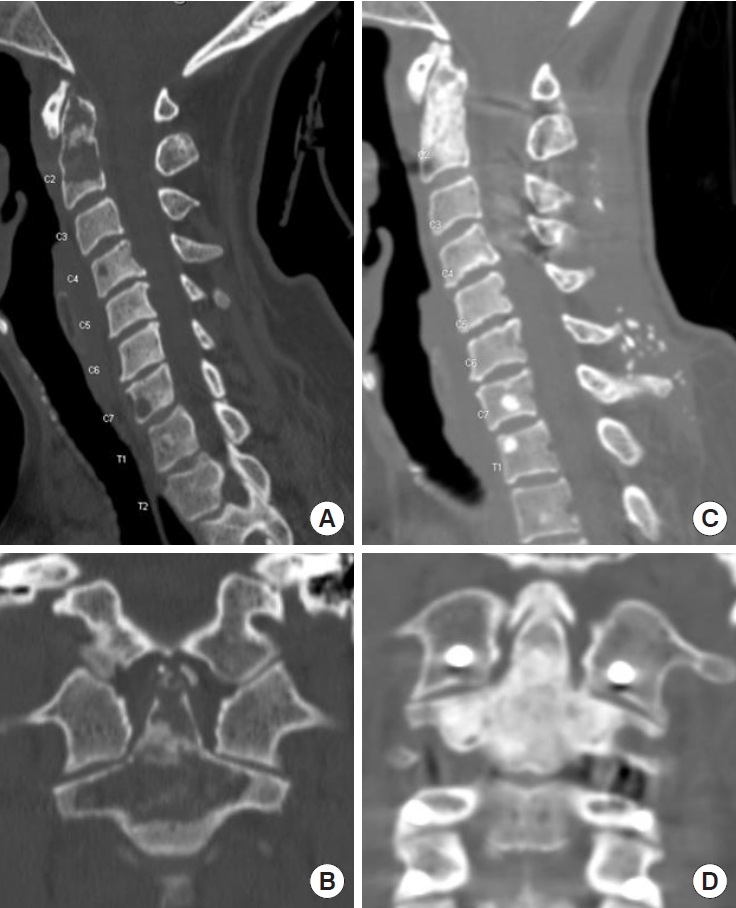
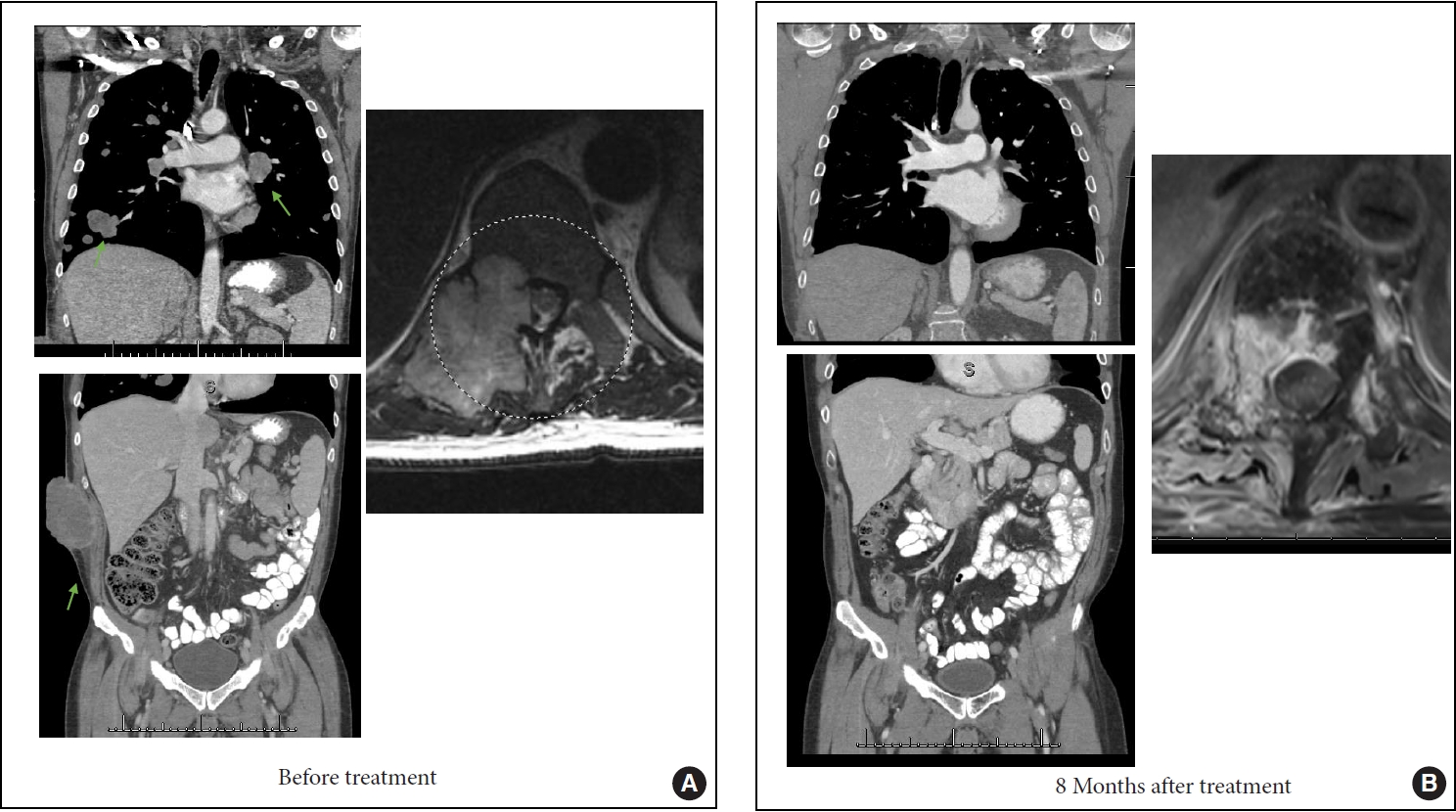
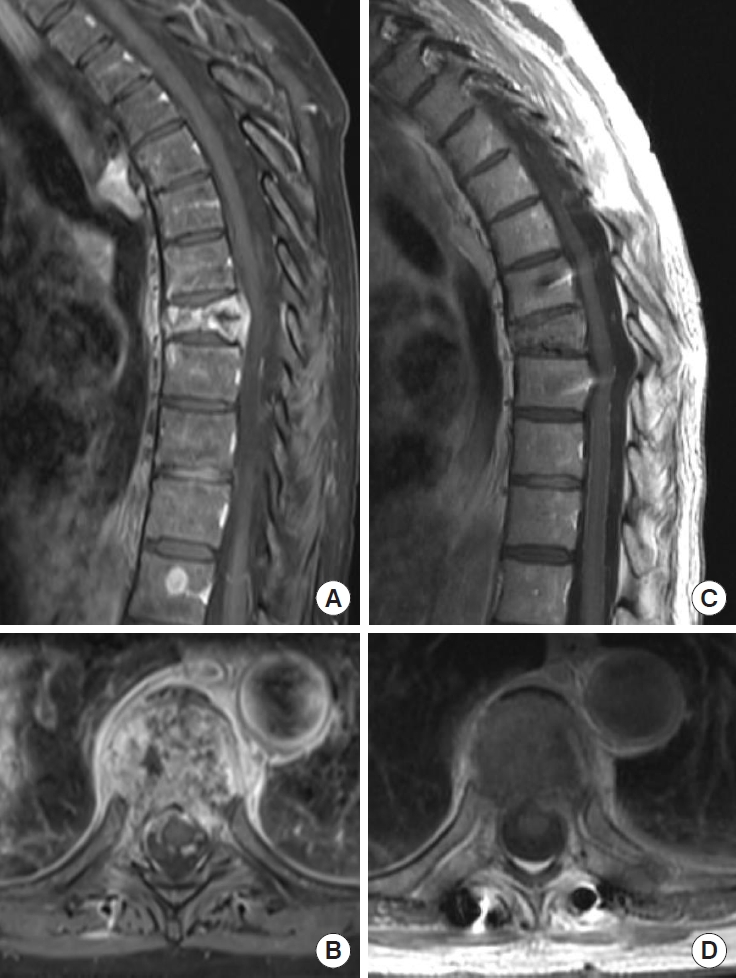
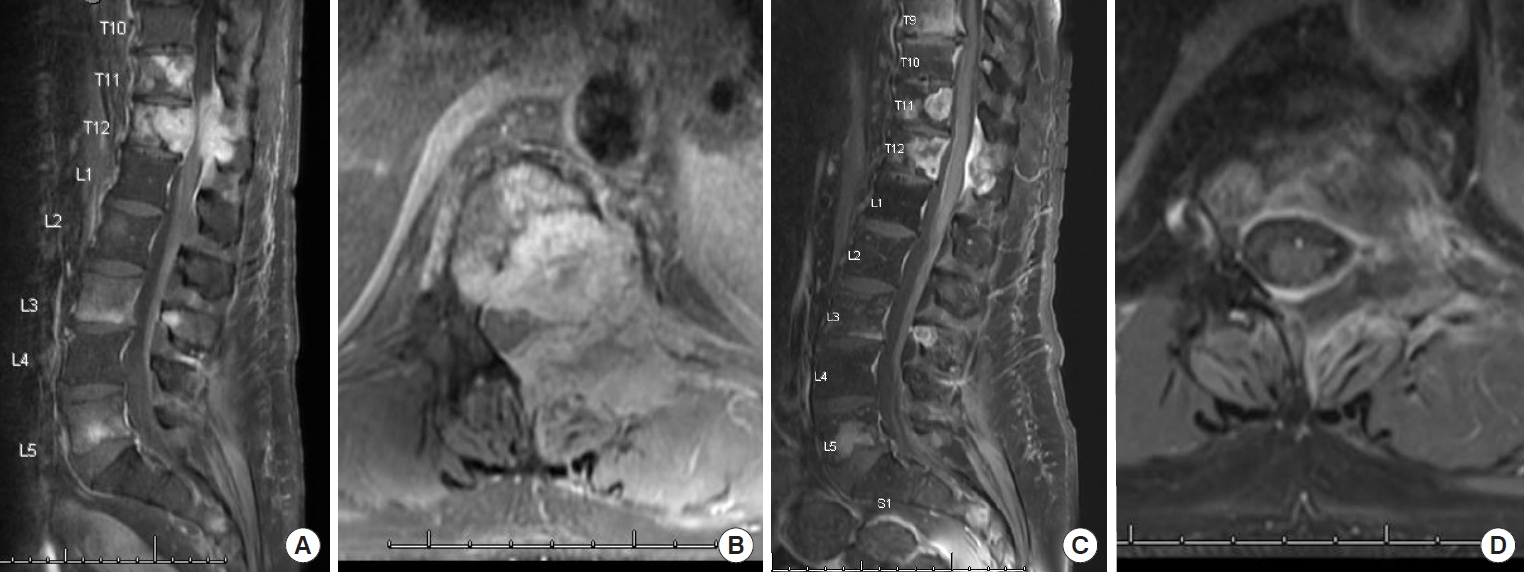
1. Perrin RG, Laxton AW. Metastatic spine disease: epidemiology, pathophysiology, and evaluation of patients. Neurosurg Clin N Am 2004;15:365-73.


4. North RB, LaRocca VR, Schwartz J, et al. Surgical management of spinal metastases: analysis of prognostic factors during a 10-year experience. J Neurosurg Spine 2005;2:564-73.


5. DŌĆÖOronzo S, Coleman R, Brown J, et al. Metastatic bone disease: pathogenesis and therapeutic options: up-date on bone metastasis management. J Bone Oncol 2019;15:004.

6. Wright E, Ricciardi F, Arts M, et al. Metastatic spine tumor epidemiology: comparison of trends in surgery across two decades and three continents. World Neurosurg 2018;114:e809-17.


10. Tomita K, Kawahara N, Kobayashi T, et al. Surgical strategy for spinal metastases. Spine (Phila Pa 1976) 2001;26:298-306.


11. Fourney DR, Frangou EM, Ryken TC, et al. Spinal instability neoplastic score: an analysis of reliability and validity from the spine oncology study group. J Clin Oncol 2011;29:3072-7.


12. Tokuhashi Y, Matsuzaki H, Toriyama S, et al. Scoring system for the preoperative evaluation of metastatic spine tumor prognosis. Spine (Phila Pa 1976) 1990;15:1110-3.


13. Tokuhashi Y, Matsuzaki H, Oda H, et al. A revised scoring system for preoperative evaluation of metastatic spine tumor prognosis. Spine (Phila Pa 1976) 2005;30:2186-91.


14. Surveillance, Epidemiology, and End Results (SEER) Program (www.seer.cancer.gov) SEER*Stat Database: Incidence - SEER Research Data, National Cancer Institute, DCCPS, Surveillance Research Program, released April 2021, based on the November 2020 submission. Accessed February 27, 2022.
15. Chan-Seng E, Charissoux M, Larbi A, et al. Spinal metastases in breast cancer: single center experience. World Neurosurg 2014;82:1344-50.


16. Groenen KHJ, van der Linden YM, Brouwer T, et al. The Dutch national guideline on metastases and hematological malignancies localized within the spine; a multidisciplinary collaboration towards timely and proactive management. Cancer Treat Rev 2018;69:29-38.


17. Pond├® N, Aftimos P, Piccart M. Antibody-drug conjugates in breast cancer: a comprehensive review. Curr Treat Options Oncol 2019;20:37.


18. Yamamoto Y, Nishimura R, Tanigawa T, et al. Efficacy and safety of TS-1 monotherapy for advanced/metastatic breast cancer - an observational study by the Kumamoto Breast Cancer Cooperative Group (KBCCG). Gan To Kagaku Ryoho 2014;41:1221-5.

19. Swain SM, Miles D, Kim SB, et al. Pertuzumab, trastuzumab, and docetaxel for HER2-positive metastatic breast cancer (CLEOPATRA): end-of-study results from a double-blind, randomised, placebo-controlled, phase 3 study. Lancet Oncol 2020;21:519-30.

21. K├╝mler I, Christiansen OG, Nielsen DL. A systematic review of bevacizumab efficacy in breast cancer. Cancer Treat Rev 2014;40:960-73.


26. Goodwin CR, Sankey EW, Liu A, et al. A systematic review of clinical outcomes for patients diagnosed with skin cancer spinal metastases. J Neurosurg Spine 2016;24:837-49.


27. Hauschild A, Grob JJ, Demidov LV, et al. Dabrafenib in BRAF-mutated metastatic melanoma: a multicentre, openlabel, phase 3 randomised controlled trial. Lancet 2012;380:358-65.


28. Dummer R, Ascierto PA, Gogas HJ, et al. Encorafenib plus binimetinib versus vemurafenib or encorafenib in patients with BRAF-mutant melanoma (COLUMBUS): a multicentre, open-label, randomised phase 3 trial. Lancet Oncol 2018;19:603-15.


30. Wolchok JD, Rollin L, Larkin J. Nivolumab and Ipilimumab in Advanced Melanoma. N Engl J Med 2017;377:2503-4.

33. Andtbacka RHI, Kaufman HL, Collichio F, et al. Talimogene Laherparepvec improves durable response rate in patients with advanced melanoma. J Clin Oncol 2015;33:2780-8.


38. Shroff GS, de Groot PM, Papadimitrakopoulou VA, et al. Targeted therapy and immunotherapy in the treatment of non-small cell lung cancer. Radiol Clin North Am 2018;56:485-95.


39. Govindan R, Szczesna A, Ahn MJ, et al. Phase III trial of ipilimumab combined with paclitaxel and carboplatin in advanced squamous non-small-cell lung cancer. J Clin Oncol 2017;35:3449-57.


40. Helissey C, Champiat S, Soria JC. Immune checkpoint inhibitors in advanced nonsmall cell lung cancer. Curr Opin Oncol 2015;27:108-17.


42. Reck M, Taylor F, Penrod JR, et al. Impact of nivolumab versus docetaxel on health-related quality of life and symptoms in patients with advanced squamous non-small cell lung cancer: results from the CheckMate 017 Study. J Thorac Oncol 2018;13:194-204.


43. Savas P, Hughes B, Solomon B. Targeted therapy in lung cancer: IPASS and beyond, keeping abreast of the explosion of targeted therapies for lung cancer. J Thorac Dis 2013;5 Suppl 5(Suppl 5):S579-92.

44. Sequist LV, Yang JCH, Yamamoto N, et al. Phase III study of afatinib or cisplatin plus pemetrexed in patients with metastatic lung adenocarcinoma with EGFR mutations. J Clin Oncol 2013;31:3327-34.


45. Lauro S, Onesti CE, Righini R, et al. The use of bevacizumab in non-small cell lung cancer: an update. Anticancer Res 2014;34:1537-45.

49. Miller JA, Balagamwala EH, Angelov L, et al. Spine stereotactic radiosurgery with concurrent tyrosine kinase inhibitors for metastatic renal cell carcinoma. J Neurosurg Spine 2016;25:766-74.


50. Bergerot P, Burns K, Prajapati D, et al. Advances in the treatment of metastatic renal cell carcinoma. Cancer Treat Res 2018;175:127-37.


51. Choueiri TK, Halabi S, Sanford BL, et al. Cabozantinib versus sunitinib as initial targeted therapy for patients with metastatic renal cell carcinoma of poor or intermediate risk: the alliance A031203 CABOSUN Trial. J Clin Oncol 2017;35:591-7.


54. James ND, Sydes MR, Clarke NW, et al. Addition of docetaxel, zoledronic acid, or both to first-line long-term hormone therapy in prostate cancer (STAMPEDE): survival results from an adaptive, multiarm, multistage, platform randomised controlled trial. Lancet 2016;387:1163-77.


55. Klotz L, Boccon-Gibod L, Shore ND, et al. The efficacy and safety of degarelix: a 12-month, comparative, randomized, open-label, parallel-group phase III study in patients with prostate cancer. BJU Int 2008;102:1531-8.


56. Attard G, Reid AHM, AŌĆÖHern R, et al. Selective inhibition of CYP17 with abiraterone acetate is highly active in the treatment of castration-resistant prostate cancer. J Clin Oncol 2009;27:3742-8.

57. Goodwin CR, Abu-Bonsrah N, Rhines LD, et al. Molecular markers and targeted therapeutics in metastatic tumors of the spine: changing the treatment paradigms. Spine (Phila Pa 1976) 2016;41 Suppl 20:S218-23.


58. Whang YE, Armstrong AJ, Rathmell WK, et al. A phase II study of lapatinib, a dual EGFR and HER-2 tyrosine kinase inhibitor, in patients with castration-resistant prostate cancer. Urol Oncol 2013;31:82-6.


59. Slovin SF, Kelly WK, Wilton A, et al. Anti-epidermal growth factor receptor monoclonal antibody cetuximab plus Doxorubicin in the treatment of metastatic castration-resistant prostate cancer. Clin Genitourin Cancer 2009;7:E77-82.


60. Fleming MT, Sonpavde G, Kolodziej M, et al. Association of rash with outcomes in a randomized phase II trial evaluating cetuximab in combination with mitoxantrone plus prednisone after docetaxel for metastatic castration-resistant prostate cancer. Clin Genitourin Cancer 2012;10:6-14.


62. Mateo J, Carreira S, Sandhu S, et al. DNA-repair defects and olaparib in metastatic prostate cancer. N Engl J Med 2015;373:1697-708.


63. Beer TM, Kwon ED, Drake CG, et al. Randomized, doubleblind, phase III trial of ipilimumab versus placebo in asymptomatic or minimally symptomatic patients with metastatic chemotherapy-naive castration-resistant prostate cancer. J Clin Oncol 2017;35:40-7.


65. Schellhammer PF, Chodak G, Whitmore JB, et al. Lower baseline prostate-specific antigen is associated with a greater overall survival benefit from sipuleucel-T in the Immunotherapy for Prostate Adenocarcinoma Treatment (IMPACT) trial. Urology 2013;81:1297-302.


67. Junghans RP, Ma Q, Rathore R, et al. Phase I trial of anti-PSMA designer CAR-T cells in prostate cancer: possible role for interacting interleukin 2-T cell pharmacodynamics as a determinant of clinical response. Prostate 2016;76:1257-70.


68. Schlumberger M, Tahara M, Wirth LJ, et al. Lenvatinib versus placebo in radioiodine-refractory thyroid cancer. N Engl J Med 2015;372:621-30.


75. Ma M, Lin B, Wang M, et al. Immunotherapy in anaplastic thyroid cancer. Am J Transl Res 2020;12:974-88.