Cervical Spine Chondroma Compressing Spinal Cord: A Case Report and Literature Review
Article information
Abstract
Chondromas are benign tumor of cartilaginous tissue that is rarely found in spine. The authors document a rare case of a 72 year old male patient with a cervical spinal chondroma compressing the spinal cord. The patient had symptoms of motor and sensory deficits, dysphagia and dysarthria. C1 and C2 laminotomy was done and the spinal tumor was removed. The patient gradually recovered from his previous symptom after the surgery.
INTRODUCTION
Chondromas are benign tumor of the cartilaginous tissue719). Spinal chondroma is very rare, especially in the cervical area5). We report a rare case of a 72 year old male patient with cervical spine chondroma compressing spinal cord.
CASE REPORT
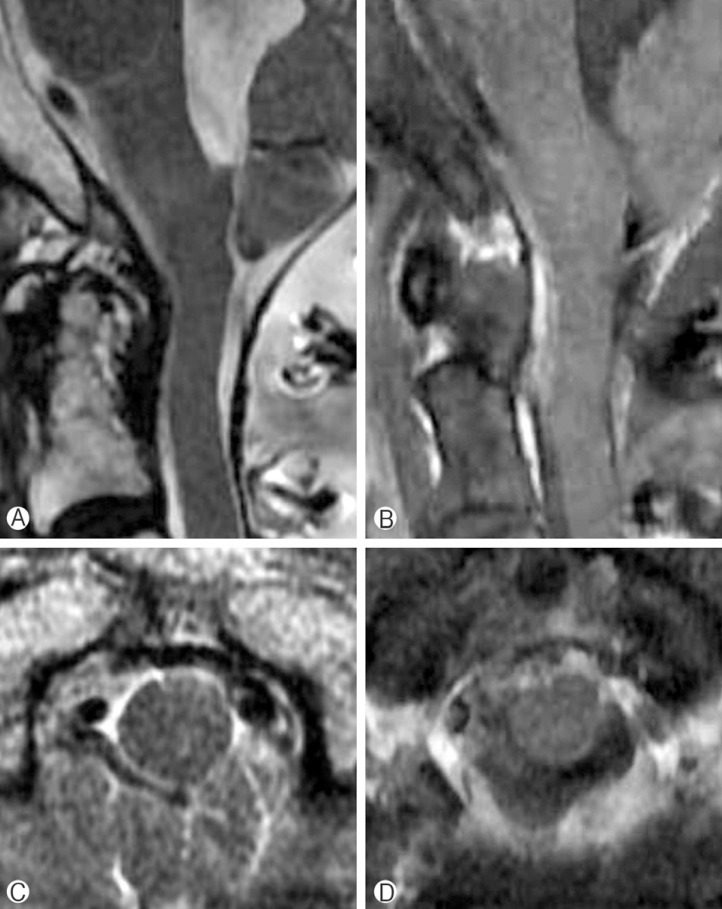
A 72 year old male patient visited the emergency room (ER) with progressive weakness and paresthesia in all extremities that were aggravated several days ago. On physical examination, the patient's motor power was decreased, showing grade III motor weakness on the right side and grade IV- on the left side. He also complained of dysarthria and dysphagia that began 12 hours before the visit to the ER. Cervical spine magnetic resonance (MR) image was taken at the ER and it showed a 3.4×1.3 cm sized heterogeneously enhancing mass with peripheral rim enhancement. The spinal tumor was based on dura and contained a cyst like portion. It was compressing the spinal cord and the exophytic cystic portion of the tumor was compressing the cervicomedullary junction (Fig. 1).
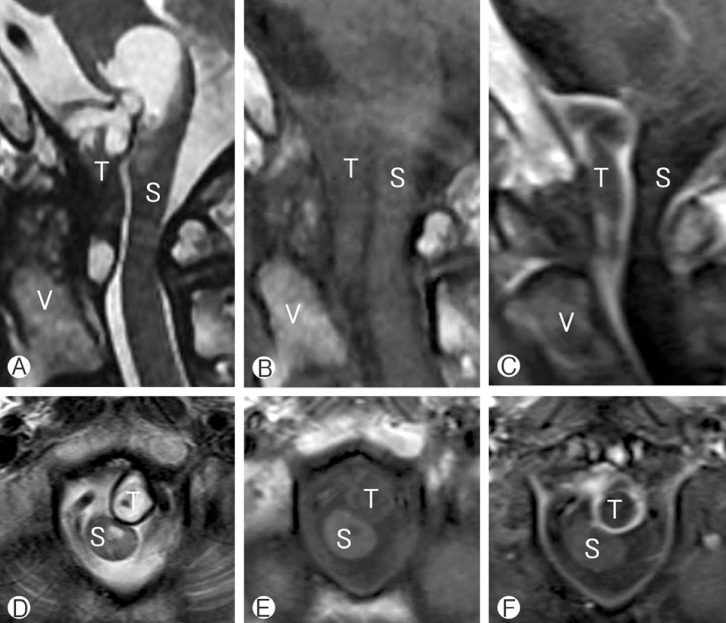
Preoperative magnetic resonance (MR) image showed 3.4×1.3 cm size heterogeneous mass (A, B, D, E). Tumor was based on dura and it contained cyst like portion. Tumor compressed spinal cord and had exophytic cystic portion compressing cervicomedullary junction (C, F). Tumor had peripheral rim enhancement. V, vertebral body; S, spinal cord; T, tumor.
C1, C2 laminotomy was done using a conventional posterior approach. After the dura was opened, a greyish fibrous tumor compressing the spinal cord was seen outside the arachnoid membrane. The tumor was hard and was firmly attached to the ventral dura. There was no adhesion between the tumor and the spinal cord. The spinal cord was mildly deviated to the side due the ventrolaterally compressing tumor (Fig. 2). The tumor was intralesionally excised while saving the nerve roots. Postoperative MRI showed the tumor was removed well and the spinal cord was adequately decompressed (Fig. 3). This conventional posterior approach technique was similar in our previous report for ventral foramen magnum meningioma16).
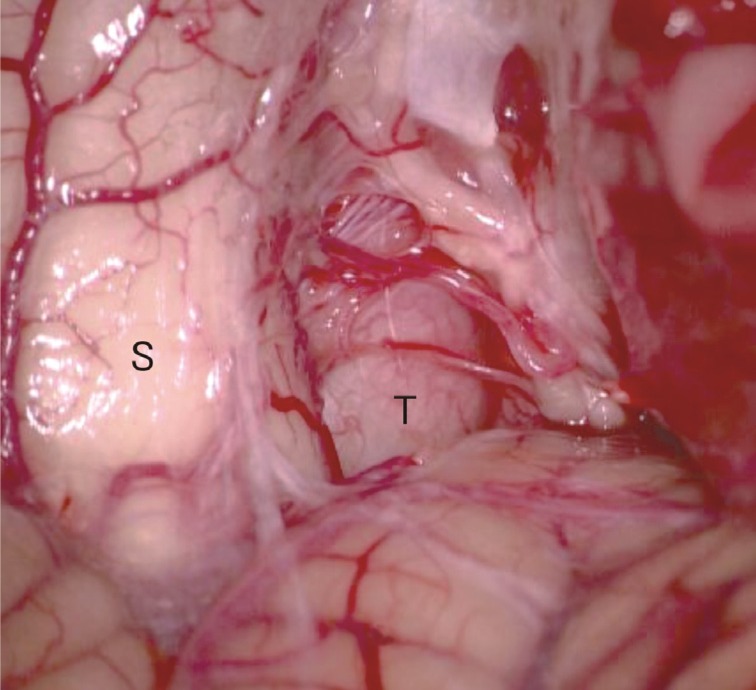
A grayish fibrous tumor compressing the spinal cord was observed after dura was opened. S, spinal cord; T, tumor; C, cerebellum.
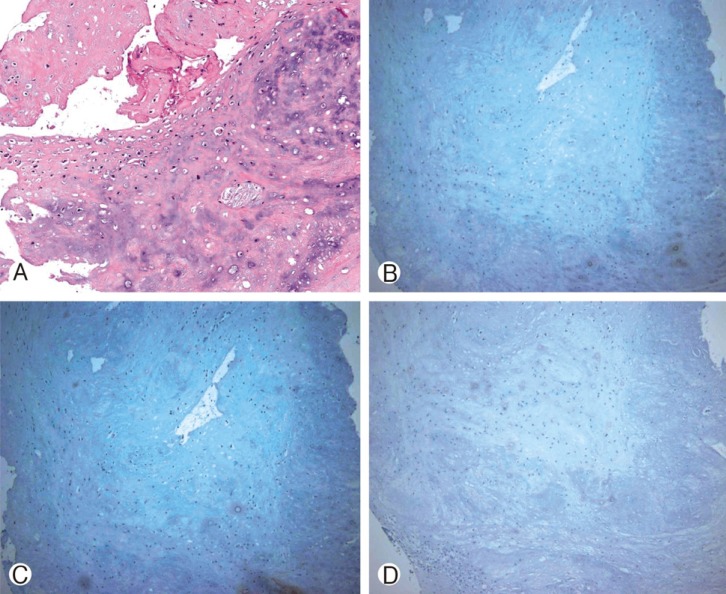
Patient's motor weakness and paresthesia was gradually improved after the surgery. The patient was able to walk using a walker three months after the surgery. Sign of dysphagia also improved over time, and he was able to swallow food without aspiration one year after surgery. The final pathology report confirmed the spinal tumor as a chondroma. Immunohistochemistry staining for cytokeratin, epithelial membrane antigen (EMA), and S-100 protein stain were all negative (Fig. 4).
DISCUSSION
Chondromas are benign tumor of cartilaginous tissue that is found frequently in the tubular bone of hands13). Chondroma within a vertebral body with neural compression has rarely been reported. It is postulated that spinal chondroma is derived from hyperplasia of immature spinal cartilage with migration outside the vertebral axis or from metaplasia of the connective tissue in contact with the spine or the anulusfibrosus8).
There have only been 10 cases of previously reported cervical spine chondroma in adults1236891314151819) (Table 1). Eight among 10 cases occurred in young adults in their 20s-30s and all patients were male except for one case4). Therefore, a cervical spine chondroma in a senile patient over 70 years of age, as in our case, is thought to be very rare. To our knowledge, our case is the first case of cervical spine chondroma with spinal cord compression in Republic of Korea, making it the 3rd case report in Asia.
Chondroma is a slow growing tumor and neurologic symptoms and signs may develop gradually. Radiculopathy or myelopathy can result from direct neural compression11). Most patients in previous studies had signs of spinal cord compression showing weakness and/or sensory deficit of extremities.
A plain X-ray may show a well circumscribed lytic lesion, with widened neural foramen if the tumor is intraforaminal213). A spine CT on bone setting may show a radiolucent and erosive lesion such as cartilage10).
Spinal chondroma usually shows an intermediate signal intensity in T1 weighted MR image. The central part of mass may show a high signal on T2 weighted MR image, which is typically composed of hyaline cartilage with high water content12). When we retrospectively reviewed our case, the cystic portion of the tumor which showed a high signal intensity on T2 weighted MR image, may have been the hyaline cartilaginous portion of the chondroma.
Histologically spinal chondroma is comprised of neoplastic chondrocytes dispersed within an abundant hyaline or myxoid background10). Immunohistochemistry staining showed negative in cytokeratin, epithelial membrane (EMA), and S-100 antibody panels which excluded tumor of a neural crest origin or of a sarcomatous change.
Surgical excision of tumor is the recommended treatment for spinal chondroma although a case of spontaneous remission has been reported13). In our case, we used a conventional posterior approach to resect the tumor instead of using a posterolateral or an anterolateral approach, which are more commonly used when resecting a ventrally located spinal tumor. A recent study has shown conventional posterior approach as a safe and an effective approach to resect tumor of ventral location16). This approach has a few advantages over other approaches, as being easier and more familiar to surgeons and showing faster postoperative recovery rate16).
Chemotherapy is ineffective for spinal chondroma, and radiation therapy is only considered for patients with unresectable tumors17). Recurrence rate is reported as less than 10% following complete resection of a chondroma11).
CONCLUSION
Spinal chondroma is very rare, especially, in the cervical area. Cervical spine chondromas were mostly found in young male adults causing pain or motor/sensory deficit of extremities. We report a rare case of a cervical spine chondroma compressing the spinal cord in an old aged male patient. He showed paresthesia and weakness of all extremities, as well as, signs of dysphasia and dysarthria. Neurologic deficits gradually recovered after surgical resection.
ACKNOWLEDGMENT
This work was supported by the Seoul National University Hospital Research Fund (No. 0420143130).
Notes
CONFLICT OF INTEREST DISCLOSURE: The authors have no disclosure to report.