Preliminary Experiences of the Combined Midline-Splitting French Door Laminoplasty with Polyether Ether Ketone (PEEK) Plate for Cervical Spondylosis and OPLL
Article information
Abstract
Objective
The purpose of this study was to evaluate the safety and efficacy of cervical midline-splitting French-door laminoplasty with a polyether ether ketone (PEEK) plate. The authors retrospectively analyzed the results of patients with cervical laminoplasty miniplate (MAXPACER®) without bone grafts in multilevel cervical stenosis.
Methods
Fifteen patients (13 males and 2 females, mean age 50.0 years (range 35-72)) with multilevel cervical stenosis (ossification of the posterior longitudinal ligament and cervical spondylotic myelopathy) underwent a combined surgery of midline-splitting French-door laminoplasty with or without mini plate. All 15 patients were followed for at least 12 months (mean follow-up 13.3 months) after surgery, and a retrospective review of the clinical, radiological and surgical data was conducted.
Results
The radiographic results showed a significant increase over the postoperative period in anterior-posterior diameter (9.4±2.2 cm to 16.2±1.1 cm), open angles in cervical lamina (46.5±16.0° to 77.2±13.1°), and sectional volume of cervical central canal (100.5±0.7 cm2 to 146.5±4.9 cm2) (p<0.001). The sagittal alignment of the cervical spine was well preserved (31.7±10.0° to 31.2±7.6°, p=0.877) during the follow-up period. The clinical results were successful, and there were no significant intraoperative complications except for screw displacement in two cases. The mini plate constructs did not fail during the 12 month follow-up period, and the decompression was maintained.
Conclusion
Despite the small cohort and short follow-up duration, the present study demonstrated that combined cervical expansive laminoplasty using the mini plate is an effective treatment for multilevel cervical stenosis.
INTRODUCTION
Multilevel cervical canal stenosis causesd by the ossification of posterior longitudinal ligament (OPLL) or cervical spondylotic myelopathy (CSM) is common42326). Most patients are asymptomatic, but those with severe spinal cord compression are predisposed to symptoms of cervical radiculopathy or myelopathy 1626). Surgical treatment is recommended when neurologic symptoms are severe. Cervical laminoplasty represents an effective technique in the treatment of multlevel cervical lesions. The technique enlarges of the canal which indirectly decompresses the spinal cord by allowing the dural sac to drift away from the spondylotic bars223). Several technical variations exist, such as the Z-plasty14) or the open door technique6). The French-door laminoplasty consists of a median lamina split followed by a lateral thinning and opening of both hemilaminae. The technique seems to provide satisfying and reliable long-term results in patients with OPLL and myelopathy due to cervical spondylosis and posterior thickening of the ligamentum flavum225). Cervical expansive laminoplasty was originally carried out as a modified French-door laminoplasty using the spinous processes as spacers15). Since the design of classic open-door laminoplasty with the use of sutures, the procedure has been modified to reduce complications such as restenosis, axial symptoms, and segmental motor paralysis315). With the development of surgical implants, various kinds of lamina spacers, such as the spinous processes, hydroxyapatite spacers, and Centerpiece® have been used to expand the narrowed spinal canal (Fig. 1)15). Each methods had its own advantages and disadvantages, but the recently developed laminoplasty fixation PEEK system with a biocompatible polymer securing the locking hinge of the plate without radiological artifacts has not been studied for its efficacy yet. The authors therefore began to use MAXPACER® (SeohanCare, Gyeonggi-Do, Korea, Fig. 2) as a lamina spacer with the French-door method, and compared the canal expansion rate among these methods.

Medical illustration of a cervical laminoplasty; (A) open-door laminoplasty; (B) French-door laminoplasty with a HA block; and(C) French-door laminoplasty with MAXPACER®.
MATERIALS AND METHODS
Between June and December 2012, combined surgery for multilevel cervical stenosis was performed by one neurosurgeon in a single university hospital. All patients were followed for at least 12 months after their operations. We performed a retrospective study of the 15 patients, analyzing the clinical and radiological results, including the difference in spinal canal areas between pre- and postoperative imaging.
The surgical procedure in the cases discussed here consisted of French-door laminoplasty with and without MAXPACER®-L type (Fig. 2). Horizontal amputation of the spinous processes was performed and bilateral laminar exposure was carried out. The upper half of spinous process was removed and applied to the gutter side after MAXPACER® was used for only bone fusion. Midline laminotomy was then performed with a drill, and lateral outer cortical bone drilling was done to facilitate elevation. During performing lateral outer cortical bone drilling, the surgeon found the lamina-facet junction, which was a landmark for drilling. It was important to drill just medial of the facet joint in order to have enough spinal canal area. Since the narrow drilling space can induce the lamina fracture when the lamina is elevated, adequate space was important to avoid fracturing the lamina. After drilling, the ligament flavum was split centrally and each lamina and ligamentum flavum was opened bilaterally until the lamina stood straight. After proper positioning of the laminae, MAXPACERs® were applied to the space between both laminae and secured by 8mm-sized titanium screws (Fig. 3). The decision of where to apply the MAXPACER® was determined when the canal expansion was no longer sustained by the conventional French-door laminoplasty without the support of medical devices.
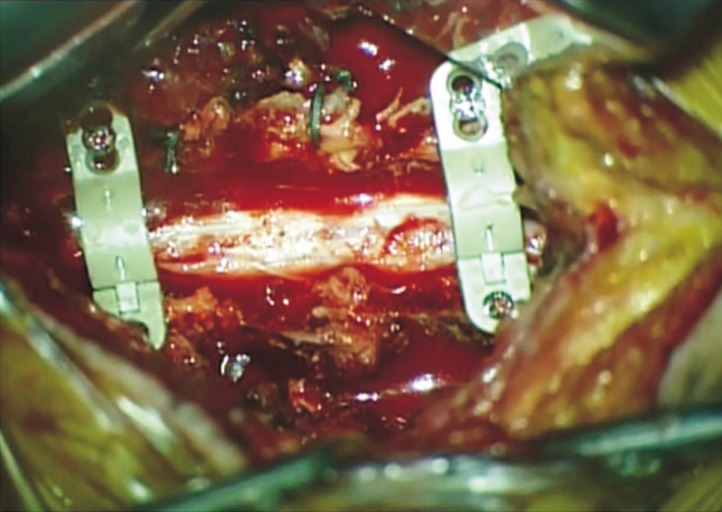
Intraoperative photograph of patient with combined cervical expansive laminoplasty using the MAXPACER® with French-door laminoplasty at the C3 and C6 levels. Horizontal amputation of the spinous processes was performed and bilateral laminar exposure was carried out. The upper half of spinous process was removed and applied to the gutter side after MAXPACER® was used for only bone fusion.
The authors used imaging programs (PACSPLUS PPW, medical standard, Korea) to measure the spinal canal area (Fig. 4). In all vertebrae, anatomical and radiological (magnetic resonance image, MRI) measurements were performed at the levels where MAXPACERs® were applied. Anterior-posterior diameter was the maximum distance between midpoint of the posterior border of vertebral body shadow and shadow of the spinolaminal junction of same vertebra12). The lamina open-door angle was equal to the angle of two lines, one through both sides of the vertebral body crossing the transverse foramen posteriorly and the other connecting with the inside edge of the hinge side of the tangent of the lamina31). The C2-7 angle was measured by formal Cobb methods that verified the angle between the horizontal line of the C2 lower end plate and the horizontal line of the C7 lower end plate30). Anatomical measurements were made by a single observer using an electronic caliper. The area of the preoperative spinal canal was defined as the region surrounded by the posterior border of the vertebral body and the inner border of the lamina, while that of the postoperative spinal canal was defined as the region surrounded by the posterior border of the vertebral body, the inner border of the lamina, and the inner border of a spacer. In cases of OPLL, OPLL was not excluded in the spinal canal area to prevent overestimation of the expansion rate. We adjusted the images to be the same size among the same sections and compared the number of pixels in the spinal canal. The canal expansion rate was calculated by the increase in pixels from the preoperative to the postoperative images.

Cervical measurements: (A) anterior-posterior diameter; (B) open angles; (C) 3D sectional volume; and (D) and (E) dynamic radiographs with Cobb's angle from C2 to C7.
The severity of clinical symptoms was assessed using the various clinical score systems such as the visual analog scale (VAS), neck disability index (NDI), short-form 12 (SF-12), Japanese orthopedic association (JOA) score, and Odom's score. The preoperative and the one-year clinical scores were evaluated to determine whether there were significant differences. Persisting nuchal pain distributed over the posterior neck and shoulder pain in the area of the suspensory muscles were defined as axial symptoms. Preoperative neck and shoulder pain and subjective outcomes regarding axial symptoms were assessed using a VAS questionnaire, on a scale from ten points (extremely severe pain) to one point (almost no pain) at discharge, at 6 months and at the one-year follow-up. Pre- and post-operative VAS scores were evaluated to determine whether there were significant differences between the two groups.
The results are expressed as the mean±standard deviation. Paired Student t-test was used to assess the statistical differences of the demographic, clinical, and radiological data at each time point between the groups using SAS software for Windows (SAS Institute Inc., Cary, NC). A p<0.050 was considered as a significant statistical difference.
RESULTS
Among the 15 patients included in this retrospectively analyzed study, five patients had OPLL, nine had CSM, and one had a combined CSM and OPLL. A patient had a previous operative lesion with anterior cervical discectomy and fusion (ACDF). The mean age of the patients was 50.0 years (range 35-72). The male versus female ratio was 13:2. One patient underwent a laminoplasty C6 with MAXPACER® with a subtotal laminectomy C5 and C7. Another patient had a laminoplasty C2-6 with MAXPACER®. Eight patients underwent laminoplasties of C3-6 with MAXPACER®. Four patients had laminoplasties of C4-6 with MAXPACER® One patient underwent a laminoplasty C5-7 with MAXPACER®. Cervical French-door laminoplasties with and without MAXPACER® were applied in all patients with 53 levels. MAXPACERs® were used in 25 levels, and an average 1.67 levels of MAXPACER® were applied. The most commonly MAXPACER® applied cervical level was C6 (n=13) followed by C3 (n=5), C4 (n=3), C5 (n=3), and C2 (n=1).
Although this data included only a limited number of cases (n=15), the most of radiological outcomes after applying MAXPACER® were significantly increased compared to the preoperative baseline. Anterior-posterior diameter (mm) was grossly increased from 9.4±2.2 in preoperative patients to 16.2±1.1 in the postoperative status (p<0.001, Fig. 5). Accordingly, the application level of MAXPACER®, all mean anterior-posterior diameters (mm) were increased in the postoperative compared to preoperative status (7.6 to 10.8 in C2, 8.3 to 16.1 in C3, 8.9 to 15.4 in C4, 9.0 to 14.8 in C5, and 10.1 to 17.2 in C6). The statistical differences were only observed in C5 and C6 due the small number of cases (a case in C2, 5 in C3, 3 in C4 and C5, and 13 in C6). The open angles before and after laminoplaty using MAXPACER® were significantly increased except at the level of C5, as is presented in Fig. 5; from 46.5° to 77.2° in all cervical levels (n=50, p<0.001), 43.7° to 78.6° in C2 (n=2, p=0.013), 38.7° to 71.1° in C3 (n=10, p<0.001), 51.1° to 84.2° in C4 (n=6, p=0.012), 51.8° to 82.0° in C5 (n=6, p=0.110), and 47.5° to 76.7° in C6 (n=26, p<0.001). The C2-7 angle by Cobb methods were preserved after laminoplaty using MAXPACER® as 31.7±10.0° in preoperative and 31.2±7.6° in final postoperative follow-up (p=0.645, Fig. 5). The expansion area of the spinal canal was significantly increased from 100.5±0.7 cm2 preoperatively to 146.5±4.9 cm2 postoperative 12 months (p<0.001, Fig. 5).
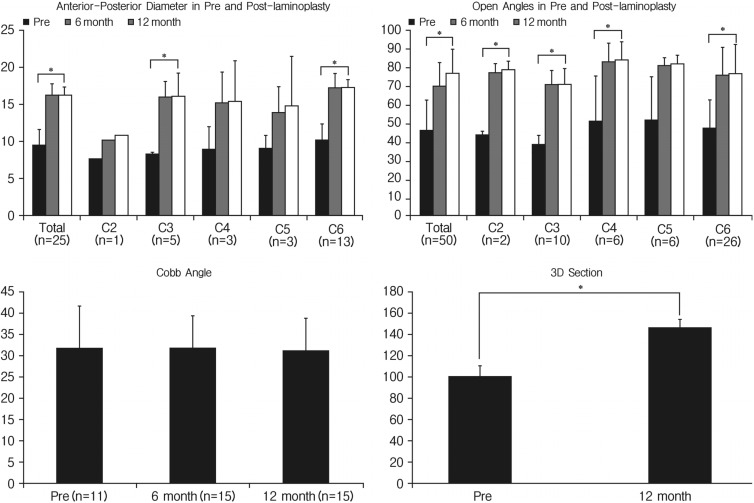
The change of anterior-posterior diameter, open angle, C2-7 angle by Cobb methods, and expansion area of the spinal canal before and after cervical laminoplasty with MAXPACER®
Postoperative clinical scores were excellent compared to the preoperative status in terms of VAS (Neck), VAS (Arm), NDI, and JOA (Fig. 6). The VAS for neck and arm pain decreased from 3.1 and 3.8 in the preoperative period to 0.6 and 1.41 postoperatively (both p<0.001), respectively. Preoperative NDI was 18.5±5.6 and decreased to 8.6±4.5 in the postoperative period (p<0.001), and JOA was also improved from 11.9±4.2 to 14.3±2.1 (p=0.023). The SF-12 for physical and mental health composite scale (PCS and MCS) were shown as 24.4±4.2 and 27.9±3.5 at postoperatively. Odom's score showed excellent results in five cases, good result in six cases, and fair results in four cases.
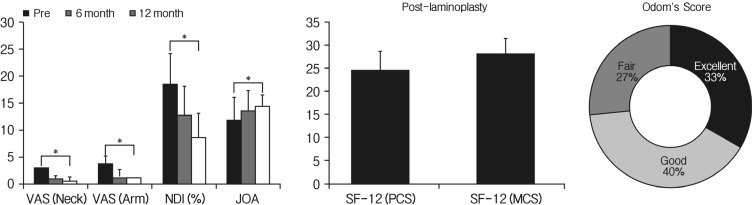
The change of clinical parameters (VAS, NDI, JOA, SF-12, and Odom's score) before and after cervical laminoplasty with MAXPACER®.
The MAXPACER® constructs did not fail during the 12-month follow-up period, and the decompression was maintained. The only observed complication related to the medical device was the displacement of screws in two cases, which occurred during the surgical procedures (Fig. 7). Serious compli cations such as instability and kyphosis did not develop in any cases.
DISCUSSION
In conditions such as degenerative cervical spondylosis, herniated cervical disc and OPLL, laminoplasty is considered as the one of the most useful surgical treatments42326). Numerous studies have reported satisfactory surgical outcomes with laminoplasty, and many technical modifications have been made to the procedure131415212425). Generally speaking, there are two types of laminoplasty methods. The first is the open-door method and the other is the French-door method. French-door laminoplasty was originally devised by Kurokawa192328). In this procedure, the expansion of the spinal canal and preservation of the posterior structures for stability of the cervical spine are important18). French-door laminoplasty allows for easy placement of spacers and performance of bilateral decompression1891032). And, French-door laminoplasty has shown superior clinical and radiological outcomes compared to open-door laminoplasty, in addition to preserving cervical muscle strength after laminoplasty172022). Both open-door and French-door laminoplasties might be similarly effective in decompression of the spinal cord; however, axial pain was improved more and the cervical lordotic angle was increased more in the French-door group after surgery22).
French-door laminoplasty can be divided into two methods, including the conventional spinous process-splitting method and the lamina-splitting method27). Because the spacers are inserted between the split laminae, the desired enlargement of the spinal canal can be obtained simply by choosing a spacer of the appropriate width27). Spacers may be less stable in the lamina-splitting method, because the contact area between the laminae and the spacers is less than that between the spinous processes and the spacers in the spinous process-splitting method. Sufficient stability could be obtained by several techniques, including the 4-point suture. Spacer fixation with screws is advantageous because of its relative ease, the speed of the procedure, and strength to pull-out27). Herein, the MAXPACER® was secured by 8mm sized titanium screws between both laminae.
Few studies compare the spinal canal area among the different types of laminoplasty. Hirabayashi et al. reported that the open-door laminoplasty with hydroxyapatite resulted in a significantly larger expansion ratio than the double-door laminoplasty6). In the study by Kim et al., they compared the amount of canal expansion among the three major types of implants used in laminoplasty15). The major finding to come from this comparison is that box-shaped laminoplasty with miniplates allows the widest canal expansion of the three implant types. The canal expansion rate with miniplates was 76.5%, while that of hydroxyapatite was 49.8%, and that achieved with Centerpiece was 50.6%. In addition, there were no reported complications associated with the miniplates. We obtained similar results in our study. Anterior-posterior diameter (9.4±2.2 to 16.2±1.1mm), the open angles (46.5° to 77.2°), and the expansion area of the spinal canal (100.5±0.7 to 146.5±4.9 cm2) were significantly increased postoperatively by using the MAXPACER®. Indeed, the C2-7 angle by Cobb methods was preserved after laminoplaty using MAXPACER® (31.7±10.0° to 31.2±7.6°). The clinical results were also excellent in term of VAS (Neck), VAS (Arm), NDI, JOA, and SF-12 (PCS and MCS).
But, the relationship between the degree of spinal canal expansion and clinical results after laminoplasty remains unclear. It was noted that spinal cord function can be regained with a minimal degree of enlargement of the spinal canal and that 4mm enlargement of the spinal canal is generally ideal11). It has also been reported that patients with a postoperative cross-sectional area of >160mm2 achieve a better outcome5). It was believed that the optimal enlargement of the stenotic canal by laminoplasty is greater than 4-5mm in the sagittal diameter7). However, although the spinal canal area can be greatly increased during laminoplasty, excessive opening of the lamina may cause problems. The kinking of the nerve root induced by maximal decompression might be related to the occurrence of postoperative C5 nerve root palsy and radiculopathy29). Excessive opening also creates epidural space and can lead to the formation of more epidural scar tissue than expected81015).
Device-related complications were very low in using the MAXPACER®, as our study showed only two cases with screw malposition (4% among total 50 screws in MAXPACER®). This complication was occurred during the operation while the surgeon was learning the technique. The screw displacements did not disturb the clinical outcomes in this retrospective study. In summary of this study and the literatures, MAXPACER® showed several advantage compared to conventional laminoplasty such as excellent surgically accessibility with easy handling27), sufficient increase of spinal canal volume1527), reasonable clinical outcome27), very low chance of intraoperative device failure with the device stability after even though device mal-positioned, and the consistency of device after plate implanted. Therefore, combined French-door laminopalsty using MAXPACER® is a useful and safe surgical technique, as long as the surgeon assures that the screws are properly positioned in the posterior laminaes.
Despite our findings, this study had several limitations. In particular, the number of patients was small and the follow-up period was too short to allow a generalization of our results. Indeed, we only performed an observational study and did not compare our results with those of alternative techniques. So, additional study is required to compare the French-door laminoplasty with the medical spacer in cases with identical operative indications.
CONCLUSION
Combined cervical expansive laminoplasty using a MAXPACER® with French-door laminoplasty is an effective treatment for multi-level cervical stenosis. Despite the small cohort and short follow-up duration in our study, future studies will be able to further confirm our findings.
ACKNOWLEDGMENT
This study is External funding was received from SeohanCare (Manufacture of MAXPACER®, Gyeonggi-do, Korea), and the authors declare that they have no proprietary, commercial, or financial interests that could be construed to have in appropriately influenced this study.
Notes
This study has not been presented in part elsewhere.

