 |
 |
- Search
Abstract
We report the case of a 47-year-old man who presented with progressive paraparesis and sphincter changes over 2 weeks. Magnetic resonance imaging revealed a spinal epidural mass from T9 to L2. We performed a decompressive laminectomy and mass removal. The histopathology was consistent with a small lymphocytic lymphoma. No metastatic lesion was noted in the chest and abdomen-pelvic computerized tomography (CT) and positron emission tomography computerized tomography (PET-CT) scan. The final diagnosis was primary spinal lymphoma, so we performed chemotherapy combined with radiotherapy. At one year follow-up, he had no neurological deficit and no recurrence on neurologic and radiologic exams. Primary spinal cord lymphomas should be considered in the differential diagnosis of spinal cord tumors. Early surgical management is mandatory to achieve a recovery of neurologic function, especially if the patient has a neurological deficit.
Malignant lymphoma (ML) can involve the central nervous system either primarily or by secondary spread, which tends to occur late in the disease as part of widespread dissemination8,18). Lymphoma presenting as primary tumors of the spinal cord are extremely uncommon. The rate of epidural mass formation in all cases of MLs is 0.8-2.86,14,19,21). Owing to this rate of occurrence, a small number of research studies and case reports dominate the literature. We report a rare case of primary spinal epidural lymphoma that presented with progressive myelopathy.
A 47-year-old man presented with progressive lower extremity weakness and numbness associated with fecal and urinary incontinence over 2 weeks. The patient did not complain of fevers, night sweats, or weight loss. A physical examination revealed hypoesthesia on the medial aspect of both thighs with bilateral motor weakness (Gr 4-4+). Anal tone decreased and urinary incontinence was observed. The vibration senses were reduced below the T10 level. There was no abnormality of laboratory findings except the erythrocyte sedimentation rate was elevated (ESR-32mm/h; reference value - less than 20mm/h). Preoperative magnetic resonance imaging (MRI) of the thora columbar region showed an isointense spinal medulla on T1-weighted images and a slightly hyperintense T2-weighted images of the spinal cord extending from T9-L2. The mass was of the homogeneous signal intensity and exhibited diffuse enhancement with gadolinium(Fig. 1A-E). The patient underwent a decompressive partial hemilaminectomy of T9-L2 and tumor removal. Intraoperatively, a grayish epidural mass was identified and resected to decompress the cord and cauda equina. Histopathologic analysis of the tumor specimen showed infiltration of a small lymphocytic lymphoma. Proliferation of small blastic cells, with a lymphoblastic aspect and starry sky pattern, was seen. Further immunohistochemical characterization showed that results from tumor testing were positive for CD20, CD3, CD5, Bcl-2, Cyclin D1 and negative for CD23, CD10 and T-cell markers (Fig. 2). We performed the further evaluations because of indicated lymphoma characteristics. The MRI of the brain and cervical spine were normal. Further work-up, including computed tomography (CT) scans of the chest and the abdomen, iliac crest bone marrow biopsy, and ultrasound of the lymph nodes showed no extramedullary lymphoma manifestation. The patient had no history of immune disorder, and human immunodeficiency virus (HIV) testing was negative. The treatment regimen consisted of radiotherapy, 40Gy administered in 20 fractions, from T9-L2, followed by combination chemotherapy with cyclophosphamide, adriamycin, oncovin, and prednisone (CHOP). A follow-up MRI one year after the surgery showed that the lesions of lymphoma had disappeared (Fig. 1F). After surgery, all preoperative symptoms completely resolved. At one year postoperatively, the patient was faring well with no evidence of local recurrence or new lesions at any other site.
Primary central nervous system lymphoma is a rare form of extranodal lymphoma, particularly isolated primary spinal lymphoma7,8). Compression of the spinal cord as the first manifestation of the primary spinal lymphoma is particularly rare, with an incidence of <5%15). In our report, the patient initially presented with compression of the spinal cord manifesting as hypoesthesia and motor weakness of the lower extremities. Although the mechanism of epidural mass formation without systemic lymphoma has not been clarified, the origin of the mass is thought to be bone marrow11). A small amount of lymphocytic cells can migrate to the spinal epidural space directly through the harversian canals of the vertebral bone or hematogenously via the epidural venous plexus, followed by a mass formation even before systemic lymphoma.
Findings from the CT and MRI provided only indirect diagnostic evidence. On T1-weighted MRI, primary spinal lymphomas are most frequently isointense to hypointense, while T2-weighted images are commonly isointense to hyperintense, with contrast enhancement4). Primary spinal lymphomas are often confused with other spinal tumors, especially metastasis12). In the present case the spinal lesion was originally thought to be a metastatic tumor. The MRI demonstrated an epidural mass lesion extending from T9 to L2, causing spinal cord compression. The lesion appeared slightly hyperintense on the T2-weighted image and exhibited continuous homogenous enhancement after gadolinium injection. The lesion was located mainly in the posterior epidural space. This is similar to the description by Negendank et al of lymphomas elsewhere in the body and Li et al of spinal epidural lymphomas12,17).
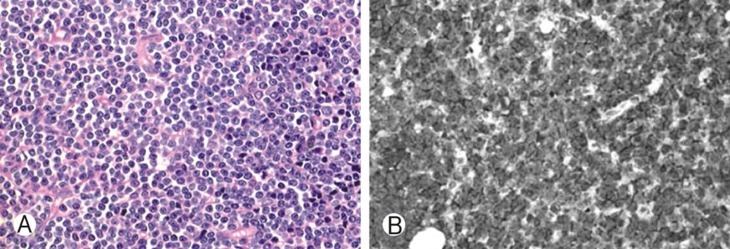
Most primary spinal lymphomas are comprised of diffuse large B-cell lymphomas with a minority being follicular lymphoma, precursor B-lymphoblastic lymphoma, Ki-lymphoma, diffuse lymphoblastic lymphoma, small lymphocytic lymphoma or T-cell lymphoma5). Small lymphocytic lymphoma is composed mostly of small mature appearing lymphocytes with round nuclei and scant basophilic cytoplasm9). In our case, immunohistochemical studies demonstrated that the tumor cells expressed the B cell markers CD19 and CD20, with coexpression of the T cell marker CD5 (Fig. 2). Expression of surface immunoglobulin M and immunoglobulin D was also noted. The tumor cells had stained positively for bcl-2 but not for CD10, CD23. Cyclin D1 staining yielded positive results for both the primary lesion and the recurrence1). Haddad et alobserved that patients with the histopathologic subtype of mixed histiocytic and small lymphocytic lymphoma seemed to have a better survival than the other subtypes (61% at 10 years), but a small number (n=17) of patients in this group did not allow statistical confirmation of this apparent observation10).
In the study by Monnard et al, out of 52 patients, 48 (92%) underwent a laminectomy, with a partial resection performed in 22 cases (42%) and a complete resection for 7 cases (13%). In surgery, each lesion was brownish or reddish purple and soft to firm in consistency. It does not usually adhere to the dural sac16,20). During an operation, we performed a hemilaminectomy of T9-L2 and a partial resection of a grayish epidural mass. However, a surgical resection does not have the survival benefits and the reported median overall survival durations with surgery alone are similar to those of untreated patients2,3). Some reports suggest that well-established treatments, such as radiotherapy and chemotherapy or a combination of both should remain as the mainstays and that surgery should be used in combination with radiotherapy and/or chemotherapy13).
Therefore, concurrent chemoradiotherapy treatment was provide (CHOP; cyclophosphamide, adriamycin, oncovin, and prednisone+radiotherapy; 40Gy administered in 20 fractions) after pathologic confirmation. Monnard et al observed local control of 88% and a 5-year overall survival of 69% with combined modality treatment. In a multivariate analysis, they found that the combined modality treatment was statistically superior to RT alone16). Also, they reported a post-treatment complete neurologic response in 25% of patients who had initial motor deficits. The remaining 75% had partial recovery. In their multivariate analysis, they have found that complete neurologic response was the most significant favorable prognostic finding with regard to overall survival (p=0.001)16). In the present case, because of the manifestations of compression, timely surgical decompression was required to allow for a recovery of nerve function even before the diagnosis had been made. After surgery, the neurologic function in our patient was completely recovered. Therefore, every effort should be made to recognize and treat this disease appropriately.
Primary small lymphocytic lymphomas are rare, especially with an initial presentation of paraparesis. A primary small lymphocytic lymphoma of the spine should be considered in the differential diagnosis of primary spine tumors. Surgical decompression and tumor removal are mandatory to recover neurologic function, if the patient had a neurologic deficit. The combined modality treatment after surgical decompression resulted in improvement of the neurologic symptoms and remission of the lymphoma.
References
1. Barnard M, Perez-Ordonez B, Rowed DW, Ang LC. Primary spinal epidural mantle cell lymphoma: case report. Neurosurgery 2000 47:1239-1241. PMID: 11063119.


3. Bellinzona M, Roser F, Ostertag H, Gaab RM, Saini M. Surgical removal of primary central nervous system lymphomas (PCNSL) presenting as space occupying lesions: a series of 33 cases. Ejso 2005 31:100-105. PMID: 15642434.


4. Boukobza M, Mazel C, Touboul E. Primary vertebral and spinal epidural non-hodgkin's lymphoma with spinal cord compression. Neuroradiology 1996 38:333-337. PMID: 8738091.


5. Chiodo A. Spinal cord injury caused by epidural B-cell lymphoma: report of two cases. J Spinal Cord Med 2007 30:70-72. PMID: 17385273.



6. Lee DY, Chung CK, Jahng TA, Kim HJ. Treatment of thoracolumbar junction metastasis: does posterior approach suffice? Korean J Spine 2004 1:207-211.
7. Ebus SC, Bernsen HJ, Norel Van GJ, Donk R. Primary non-hodgkin's lymphoma in multiple vertebrae presenting as a lumbar radicular syndrome: a case report. Spine (Phila Pa 1976) 2002 27:E271-E273. PMID: 12004189.


8. Friedman M, Kim TH, Panahon AM. Spinal cord compression in malignant lymphoma. Treatment and results. Cancer 1976 37:1485-1491. PMID: 1260667.


9. Gupta D, Lim MS, Medeiros LJ, Elenitoba-Johnson KS. Small lymphocytic lymphoma with perifollicular, marginal zone, or interfollicular distribution. Mod Pathol 2000 13:1161-1166. PMID: 11106071.


10. Haddad P, Thaell JF, Kiely JM, Harrison EG, Miller RH. Lymphoma of the spinal extradural space. Cancer 1976 38:1862-1866. PMID: 991100.


11. Higashida T, Kawasaki T, Sakata K, Tanabe Y, Kanno H, Yamamoto I. Acute lymphocytic leukemia recurring in the spinal epidural space. Neurol Med Chir (Tokyo) 2007 47:375-378. PMID: 17721056.


12. Li MH, Holtas S, Larsson EM. MR imaging of spinal lymphoma. Acta Radiol 1992 33:338-342. PMID: 1633044.


13. Liu BL, Cheng JX, Zhang X, Zhang W, Cheng H. Limited role of surgery in the management of primary central nervous system lymphoma (Review). Oncol Rep 2009 22:439-449. PMID: 19639187.

14. Mak KS, Lee LK, Mak RH, Wang S, Pile-Spellman J, Abrahm JL, et al. Incidence and treatment patterns in hospitalizations for malignant spinal cord compression in the United States, 1998-2006. Int J Radiat Oncol Biol Phys 2011 80:824-831. PMID: 20630663.


15. McDonald AC, Nicoll JA, Rampling RP. Non-hodgkin's lymphoma presenting with spinal cord compression; a clinicopathological review of 25 cases. Eur J Cancer 2000 36:207-213. PMID: 10741279.


16. Monnard V, Sun A, Epelbaum R, Poortmans P, Miller RC, Verschueren T, et al. Primary spinal epidural lymphoma: patients' profile, outcome, and prognostic factors: a multicenter Rare Cancer Network study. Int J Radiat Oncol Biol Phys 2006 65:817-823. PMID: 16542791.


17. Negendank WG, al-Katib AM, Karanes C, Smith MR. Lymphomas: MR imaging contrast characteristics with clinical-pathologic correlations. Radiology 1990 177:209-216. PMID: 2399318.


18. Pels H, Vogt I, Klockgether T, Schlegel U. Primary non-hodgkin's lymphoma of the spinal cord. Spine (Phila Pa 1976) 2000 25:2262-2264. PMID: 10973412.


19. Perry JR, Deodhare SS, Bilbao JM, Murray D, Muller P. The significance of spinal cord compression as the initial manifestation of lymphoma. Neurosurgery 1993 32:157-162. PMID: 8437651.


20. Samadian M, Vahidi S, Khormaee F, Ashraf H. Isolated, primary spinal epidural hodgkins disease in a child. Pediatr Neurol 2009 40:480-482. PMID: 19433288.


21. Zheng JS, Wang M, Wan S, Zhou YQ, Yan M, Chen QF, et al. Isolated primary non-hodgkin's lymphoma of the thoracic spine: a case report with a review of the literature. J Int Med Res 2010 38:1553-1560. PMID: 20926031.


Fig. 1
Pre and postoperative Magnetic resonance images of the epidural spinal mass. (A) and (B): Sagittal T2-weighted images, in which the lesion is slightly hyperintense. (C): T1-weighted image with slight hypointensity. (D): Ho mogeneous enhancement by a contrast medium from level T9 to L2. (E) Axial image with contrast medium at the level of T11, compressing the spinal cord to the right side. (F): After one year postoperative sagittal T2-weighted images, there was no recurrence in the previous thoracolumbar region.

Fig. 2
(A) Photomicrograph of a monomorphous specimen and comprised of small lymphocytes with a blend cytological appearance. A heterogenous mixture of cells including prolymphocytes, lymphoplasmacytoid cells or features of disease progression with an increased number of larger cells (paraimmunoblasts) and mitoses or an aberrant phenotype should be considered in the small lymphocytic lymphoma (Haematoxylin and eosin, ×400.) (B) Immunohistochemical staining showing tumor cells positive for CD20 (EnVision, original magnification ×200).

- TOOLS
-
METRICS

-
- 6 Crossref
- Scopus
- 6,283 View
- 90 Download

























