The Effect of Recombinant Tyrosine Hydroxylase Expression on the Neurogenic Differentiation Potency of Mesenchymal Stem Cells
Article information
Abstract
Objective
Tyrosine hydroxylase (TH) is a rate-limiting enzyme in dopamine synthesis, making the enhancement of its activity a target for ensuring sufficient dopamine levels. Rat bone marrow mesenchymal stem cells (rBM-MSCs) are known to synthesize TH after differentiating into neuronal cells through chemical induction, but the effect of its ectopic expression on these cells has not yet been determined. This study investigated the effects of ectopic recombinant TH expression on the stemness characteristics of rBM-MSCs.
Methods
After cloning, a cell line with stable TH expression was maintained, and the proliferation, the gene expression profile, and differentiation potential of rBM-MSCs were analyzed. Analysis of the cells showed an increment in the proliferation rate that could be reversed by the neutralization of TH.
Results
The constitutive expression of TH in rBM-MSCs was successfully implemented, without significantly affecting their osteogenic and adipogenic differentiation potential. TH expression improved the expression of other neuronal markers, such as glial fibrillary acidic protein, β-tubulin, nestin, and c-Fos, confirming the neurogenic differentiation capacity of the stem cells. The expression of brain-derived neurotrophic factor (BDNF) and ciliary neurotrophic factor (CNTF) significantly increased after the chemical induction of neurogenic differentiation.
Conclusion
In this study, the expression of recombinant TH improved the neuroprotective effect of MSCs by upregulating the expression of BDNF and CNTF. Although the neuronal markers were upregulated, the expression of recombinant TH alone in rBM-MSCs was not sufficient for MSCs to differentiate into neurogenic cell lines.
INTRODUCTION
Tyrosine hydroxylase (TH) is a member of the aromatic amino acid hydroxylase family and is the rate-limiting enzyme in the biosynthesis pathway of the catecholamines. For that reason, TH expression plays a key role in some neurological disorders, such as Parkinson disease (PD) [1]. To compensate the decreased level of dopamine, many strategies were developed to sustain the required level, such as transplanting fetal mesencephalic dopamine cells [2,3], genetically modified cells expressing TH [4-6], and direct transfer of TH-producing gene cassettes to endogenous striatal cells [7-9].
Mesenchymal stem cells (MSCs) have the potential to differentiate into various cell types including neuronal cells. However, the neuronal differentiation potential of these cells is closely related to the cell origin, and it was previously demonstrated that dental pulp-MSCs possess higher differentiation capacity into neuronal cell lineages than bone-marrow derived MSCs [10]. The improvement of the differentiation characteristics of MSCs into dopaminergic could be achieved by gene transformation [11]. Genetic modification of MSCs was shown to provide an attractive approach to produce therapeutically important proteins, like brain-derived neurotrophic factor (BDNF) [12].
In this study, rat bone marrow-derived MSCs (rBM-MSCs) were modified by transfection of rat TH gene. The extracellular production of TH was aimed to analyze the effect of the enzyme on the differentiation potential of stem cells into neuronal cell lineages. The changes in cell proliferation and other stem cell characters after modification were also evaluated in this context.
MATERIALS AND METHODS
1. Isolation and Culture of rBM-MSCs
The bone marrow of Wistar Albino rat (n=5) was used to isolate MSCs. The methods used in this study were approved by Kocaeli University Ethics Committee for Animal Experiments (KOU HADYEK 6/4-2011). Isolation and culture of rBM-MSCs were performed as previously described [13]. Under sterile conditions, both rat femur and tibiae were excised, and cells were separated by density centrifugation by Ficoll-histopaque (1.077 g/mL), and the cell pellet was resuspended in L-dulbecco's modified eagle's medium (L-DMEM) (Gibco, Invitrogen, Paisley, UK). The cells were seeded in plastic tissue culture flasks and incubated at 37°C in humidified air with 5% CO2. After the cells reached 70%–80% confluence, were subcultured using 0.25% trypsin ethylenediaminetetraacetic acid (EDTA) solution (Gibco).The culture media was refreshed once every 3 days.
2. Flow Cytometry Analysis
The isolated cells were characterized with respect to following antigens in cytometer: CD29, CD45 CD90, CD54, CD106, major histocompatibility complex (MHC) Class I and MHC Class II, as previously described [14]. All antibodies were supplied by BD Biosciences (San Diego, CA, USA). Flow cytometry was performed using a FACSCalibur (BD Biosciences), and data were analyzed with Cell Quest software (BD Biosciences).
3. Differentiation of TH+ rBM-MSCs
Adipogenic and osteogenic differentiation were performed according to the protocol mentioned previously [14]. To induce adipogenic differentiation, cells (3,000 cells/cm2) were cultured in L-DMEM (Gibco) supplemented with 10% fetal bovine serum (FBS) (Gibco), 0.5 mM isobutyl-methylxanthine (IBMX) (Sigma, St. Louis, MO, USA), 1 μM dexamethasone (Sigma), 10-μg/mL insulin (Gibco), 200 μM indomethacin (Sigma), and 1% Pen/Strep (Gibco) for 3 weeks. The presence of intracellular lipid droplets was confirmed by staining with 0.5 % Oil red O (Sigma). For osteogenic differentiation, cells (3,000 cells/cm2) were cultured in L-DMEM supplemented with 0.1 μM dexamethasone, 0.05 μM ascorbate-2-phosphate (Wako Chemicals, Richmond, VA, USA), 10 mM β-glycerophosphate (Sigma), 1% Pen/Strep and 10% FBS. After 4 weeks, osteogenic differentiation was assessed via staining with 2% alizarin red (pH 4.1–4.3; Fluka, Buchs, Switzerland).
For neurogenic differentiation, cells on collagen (type-I) coated coverslips were cultivated until 70% confluency. Cells were further cultured in differentiation medium (L-DMEM supplemented with 0.5 mM IBMX), epidermal growth factor (Biological Industries, Kibbutz Beit Haemek, Israel), basic fibroblast growth factor (Biological Industries), neural stem cell proliferation supplements (StemCell Technology, British Columbia, Canada), and 1% Pen/Strep.
4. Isolation of TH Gene From Rat Brain Tissue
The tissue was obtained from Wistar albino rat (4 months) by excision of the brain cortex. The tissue was transferred in RNA Later Solution (Qiagen, Hilden, Germany). Total RNA was isolated by the High Pure RNA Isolation Kit (Roche, Mannheim, Germany), according to the manufacturer’s instructions. The concentration and purity were detected by measurements at 260 nm and 280 nm. Complementary DNA (cDNA) synthesis was performed by Transcriptor High Fidelity cDNA Synthesis Kit (Roche).
5. Cloning of TH Gene
The second strand DNA synthesis and subsequent TH gene amplification were performed by Phusion DNA polymerase (Thermo, Braunschweig, Germany). The reaction mixture included 1X polymerase chain reaction (PCR) buffer, 0.2 mM deoxynucleoside triphosphate, 0.5 μM of each primer, 1.25 mM MgCl2, and 2 μL of cDNA from the first strand reaction. An initial 5-minute denaturation step at 94°C was followed by cDNA amplification cycles including denaturation at 94°C, annealing at an appropriate temperature at 60°C and elongation at 72°C. The gene separated in agarose gel (Roche, 1%) was purified by Agarose GelExtract Mini Kit (Prime5, Hamburg, Germany), restriction digested with HindIII and BamHI (Thermo, Fermentas, Vilnius, Lithuania) and ligated by T4 DNA Ligase (Thermo, Fermentas) into pFLAG-3 (Sigma) downstream of cytomegalovirus (CMV) promoter. Following subcloning and isolation by EndoFree plasmid isolation maxi kit (Qiagen), the plasmid was transfected by electroporation (Neon Transfection System, Invitrogen, Carlsbad, CA, USA) with respect to the instructions provided by the manufacturer. The transfection parameters for rBM-MSCs were 990 V/40 ms/2 pulses. After 48 hours of culture in L-DMEM with 10% FBS, the transformed cells were selected with respect to their resistance against 400 μg/mL G418 (Roche) under standard culture conditions.
6. Immune Staining
Samples were fixed in ice-cold methanol for 10 minutes and treated with 0.025% Triton X-100 (Merck, Darmstadt, Germany) for permeabilization. Cells were incubated with 1.5% blocking serum solution (Santa Cruz Biotechnology, Heidelberg, Germany) for 30 minutes at 37°C, and incubated overnight at 4°C with the primary antibodies. Samples were incubated with appropriate secondary antibodies for 25 minutes and covered with mounting medium containing DAPI (4´,6-diamidino-2-phenylindole) (Santa Cruz Biotechnology). The mounted cells were immediately examined under a fluorescence microscope (Leica DMI 4000B, Wetzlar, Germany).
7. Real-Time PCR Analysis
The gene expression levels were detected by LightCycler 480 DNA SYBR Green I Master (Roche) with gene-specific primers on LightCycler 480 real-time PCR instrument, according to the manufacturer’s protocol. Melting curve analysis for determining the dissociation of PCR products was performed from 60°C to 95°C to confirm the presence of a single peak. The second derivative maximum method was performed to calculate Cp values.
To profile the gene expression in MSCs before and after gene transfer, real-time (RT) [2] Profiler PCR Array for rat stem cells (PARN-405F, SABioscience, Qiagen, Germany) was used according to the manufacturer’s protocol.
8. Western Blot Hybridization
Sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) was performed according to Laemmli [15]. SDS-PAGE was resolved in 11% polyacrylamide resolving gel with a 4% stacking gel. Protein samples were denatured by mixing with sample buffer (2% SDS, 10% glycerol, 0.1 M Tris–HCl at pH 6.8, 1% β-mercaptoethanol, traces bromophenol blue) and heated in boiling for 3 minutes. Gels were run in a Mini-PROTEAN Tetra Cell system (Bio-Rad, Herts, UK). The gels were run at 100 V. For Western blotting, proteins were transferred onto nitrocellulose membrane by i-Blot instrument (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s instructions. The membranes were blocked in phophate buffered saline supplemented with 3% skim milk and were incubated with antibodies against TH (Santa Cruz Biotechnology, sc-73152) or FLAG-Protein Expression System (Sigma, F3165) at 4°C overnight. Following incubation with the specific secondary anti-mouse peroxidaseconjugated anti-IgG antibody (Santa Cruz Biotechnology, sc-2005). The bands were visualized by enhanced chemiluminescence (ECL Plus; GE Healthcare, Uppsala, Sweden).
9. ELISA for TH
The level of rat TH in media was detected by enzyme-linked immunosorbent assay (ELISA) (USCN Life Science Inc., Wuhan, China) according to the manufacturer’s recommendation. The minimum detectable dose of TH was 59 pg/mL. All these experiments were repeated at least three times. The differences in the absorbance were measured by a ultraviolet-visible spectrophotometer (VersaMax microplate reader, Molecular Devices, Sunnyvale, CA).
10. Cell Proliferation and Cell Cycle Analysis
The water soluble tetrazolium-1 (WST-1) assay was used to estimate the proliferation rate of the cells in culture media. The cells were seed in 96-well plate, and cultured for 48 hours, before reaching 70% confluence. The culture media was removed and the basal media supplemented with WST-1 (10%; Roche) was added to the well. After incubation at 37°C for 1 hour in dark, the color formation was and quantified at 450 nm with a microplate reader.
CycleTEST PLUS DNA Reagent Kit (BD Biosciences) was used to perform the DNA ploidy analysis in different rBMMSCs following the manufacturer’s protocol. The cells were detached, and later the DNA content was labeled with propidium iodide. The analyses were performed by flow cytometer. Peripheral blood mononuclear cells with known DNA amount were used as a control.
11. Neutralization Assay for TH
For neutralization of TH protein, the antibody against TH (Santa Cruz Biotechnology, sc-73152) was added in the culture medium at the amount of 0.2 μg/mL. The cells were cultured in this media for 8 days, and the medium was refreshed once every 2 days.
12. Statistical Analysis
All experiments were performed in triplicates. Data were reported as means±standard deviation. The statistical analyses were performed by SPSS ver. 10.0 (SPSS Inc., Chicago, IL, USA). Data were analyzed using 1-way analysis of variance or paired t-test. Differences between the experimental and control groups were declared statistically significant when p<0.05.
RESULTS
1. Expression of rat TH Gene in rBM-MSC
TH gene of 1,497-kb length was isolated from rat brain tissue and ligated first to pUC19 for sequence analysis. The sequence of the fragment was checked by comparing it with TH gene sequence in the database (GeneBank Acc. No.: NM 012740) for any mutation. The fragment was later ligated into a pFLAG-3 expression vector, and the construct was transferred into rBMMSCs in passage 3 (P3). The presence of gene integration was confirmed by green fluorescent protein (GFP) expression in the cell (Fig. 1A). The expression of FLAG-Protein Expression System fragment, which should be coexpressed with TH gene as fusion protein under the control of CMV promoter, was detected in the cells (Fig. 1B). After the selection with G418 for 3 consequent passages, the ratio of GFP positive cells expressing was estimated by flow cytometry to be 92.09%±5.88%. This indicates that more than 90% of the cells are coexpressing TH gene with GFP (Supplementary Fig. 1). Western blot analysis for TH and FLAG protein fragments showed 2 fragments of nearly 60 kDa (Fig. 1C). The size of FLAG peptide did not significantly affect the total size of TH protein on the blotting membrane. The time course of TH gene expression was shown that the expression altered during the culture (Fig. 1D). TH was not detected in the cell lines without TH gene transfection (wild-type and empty vector transfections). After one day of culture, the weak protein expression was measured in the transfected cells. The level of the expression was increased in the second day but sharply dropped down in the following day. The highest level of TH expression was found 0.17±0.01 ng/mL on day 2.

(A) The expression of the transgenic gene in rBM-MSCs. Following transfection, most of the cells showed GFP expression after 3 days (scale bar: 200 µm). (B) The immunostaining against FLAG peptide (blue color) was performed to show the coexpressed TH gene (scale bar: 50 µm). (C) Western blot analysis against TH (1) and FLAG (2) showed the protein expression in size of about 60 kDa. (D) TH level was determined highest on the second day in the culture medium of TH+ rBM-MSCs. rBM-MSC, rat bone marrow mesenchymal stem cells; GFP, green fluorescent protein; FLAG, protein expression system; TH, tyrosine hydroxylase.
2. Effect of TH Gene Expression on rBM-MSC
After gene transfection, significant alteration in cell morphology was not observed. The flow cytometer analysis for both, rBM-MSCs and TH+ rBM-MSCs, demonstrate no significant change in the surface markers (data not shown). However, the proliferation rate of transfected cells increased. The cell cycle status was evaluated to estimate any change (Fig. 2A). The analysis showed that the ratio of TH+ rBM-MSCs in S phase was increased slightly to 18.67% from 14.77%, and the cells in G2/M phase and G1 phase was changed from 8% to 6.08% and from 77.23% to 75.26%, respectively. The expression of neuronal markers was analyzed in rBM-MSCs and TH+ rBM-MSCs. For this purpose, the immune staining was performed on Nestin, c-FOS, GFAP, TUBB, NF, and TUBB3 expressions (Fig. 3). Nestin, GFAP, and TUBB3 staining were found positive. However, immune staining of cells against c-FOS, NF, or TUBB was negative. Epithelial marker (E-cadherin), pancreatic endoderm marker (Pdx1) and chondrogenic markers (Col2a1a, BMP3, and Acan) were downregulated in TH+ rBM-MSCs compared to the cells before transformation (Fig. 2B). On the other hand, neural markers (N-Cadherin, Jag1, and Tubb3) and osteoblast marker (Col1a1) were increased. Dll3, which can inhibit the primary neurogenesis, was decreased, but Wnt1, which is a signaling molecule important in neural development, was also decreased.

(A) The cell cycle status of rBM- and THM+ rBM-MSCs. Cell cycle distribution of transfected and untransfected cell lines was measured by flow cytometry after propidium iodide staining. All experiments were repeated at least 3 times. (B) Changes in expression of differentiation markers in TH+ rBM-MSCs with respect to untransformed cells. TH, tyrosine hydroxylase; rBM-MSC, rat bone marrow mesenchymal stem cells.
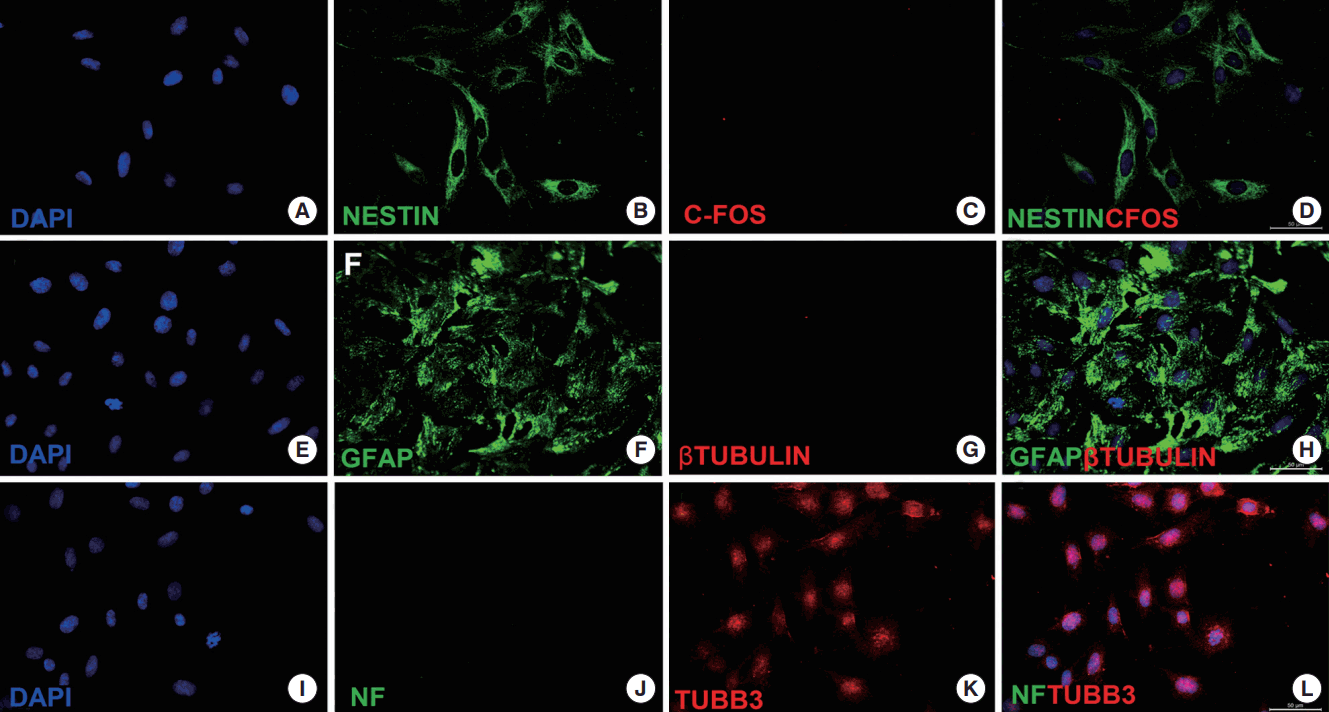
Immunophenotype analysis of TH+ rBM-MSCs for neurogenic markers. Representative staining patterns are shown for Nestin (B, D), c-Fos (C, D), GFAP (F, H), β Tubulin (G, H), NF (J, L), TUBB3 (K, L). Nuclei were labeled with DAPI (A, E, I; blue). Only Nestin, GFAP, and TUBB3 staining were found positive. Scale bars represent 50 µm. TH, tyrosine hydroxylase; rBM-MSC, rat bone marrow mesenchymal stem cells; DAPI, 4´,6-diamidino-2-phenylindole.
3. Alteration of Differentiation Potential After Gene Transfer
The differentiation potential of cells into osteogenic, adipogenic and neurogenic cell lineages was further analyzed by histochemical staining, and impact of TH gene expression on stemness character of cells was demonstrated. rBM-MSCs showed superior osteogenic differentiation capacity, and this feature of cells continued after gene transfer. After 28 days in osteogenic differentiation medium, mineral deposits were observed to form in TH+ rBM-MSCs (Fig. 4B). Later these deposits were stained with Alizarin Red S. There is no significant difference in the staining patterns with this dye in the cell lines before and after gene transfection (Fig. 4C). In the adipogenic differentiation medium, rBM- and TH+ rBM-MSCs were differentiated to the same degree. The oil droplets formed were demonstrated by Red Oil O staining (Fig. 4E). Neurogenic differentiation capacity of TH+ rBM-MSC was analyzed after the incubation in neurogenic differentiation medium for 20 hours, 30 hours, 60 hours, and 10 days. Stem cells exposed to neural differentiation medium underwent dramatic morphological changes reminiscent of neuritis (Fig. 5C). The morphological changes were significant in the cells after 20 hours of incubation, but the expression of neural cell line related markers did not appear before 30 hours of incubation. TH and coexpressed FLAG peptide expression were visible before the differentiation process, but significant increase in TH was detected after 30 hours of induction (Fig. 5D-O). This sudden increase in the staining could be related to the induction of endogenous TH expression. The neurogenesis markers, Nestin and GFAP, were detected throughout the induction period, but Tubb3 and c-Fos expressions were only visible after 30 hours, disappeared after 60 hours of induction and not visible after 10 days (Fig. 6). The Tubb3 and c-Fos were expressed at the same time when endogenous TH expression was at the highest point. Similarly, the expression of neurotrophic factors, BDNF, and CNTF, were also observed in the same time period (Fig. 7A-H). The gene expression analysis of neurogenic markers in differentiated/undifferentiated TH+ rBM-MSCs supported our findings with respect to increased levels of TH, c-Fos, and Sox9 (Fig. 7J). TH gene showed 4 times higher expression level in differentiated TH+ rBM-MSCs than in undifferentiated TH+ rBM-MSCs. The neurogenic markers, c-Fos, and Sox9, expressions were increased almost 6 and 3 fold, respectively.
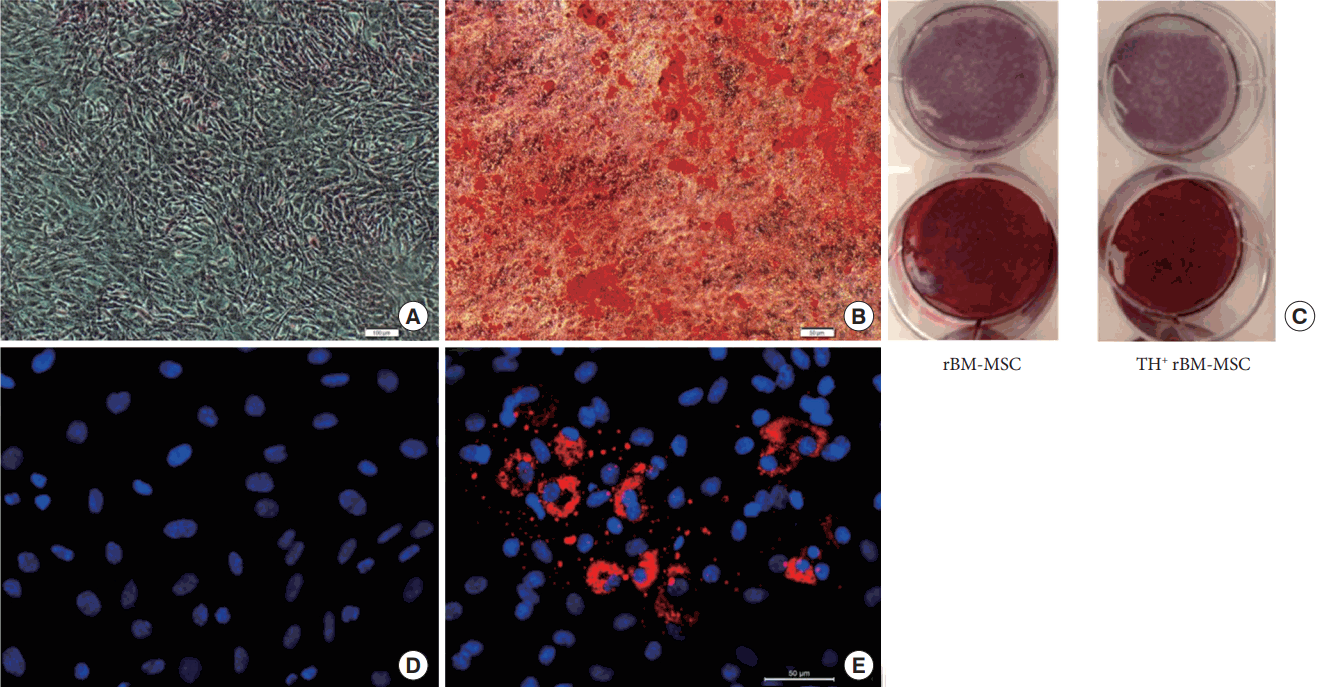
Differentiation of TH+ rBM-MSCs into osteogenic (A–C) and adipogenic (D–E) lineages. After osteogenic differentiation cells were stained with Alizarin Red-S (B) and control (A) (scale bar: A, B, 200 µm). (C) There is no difference in the staining pattern between rBM- and TH+ rBM-MSC. After induction to differentiate into adipogenic cell lineage, TH+ rBM-MSC were stained with Oil Red O (E) and control (D) (scale bar: 50 µm). In the adipogenic differentiation medium, rBM- and TH+ rBM-MSCs were differentiated to the same degree. (E) The oil droplets formed were demonstrated by Red Oil O staining. TH, tyrosine hydroxylase; rBM-MSC, rat bone marrow mesenchymal stem cells.
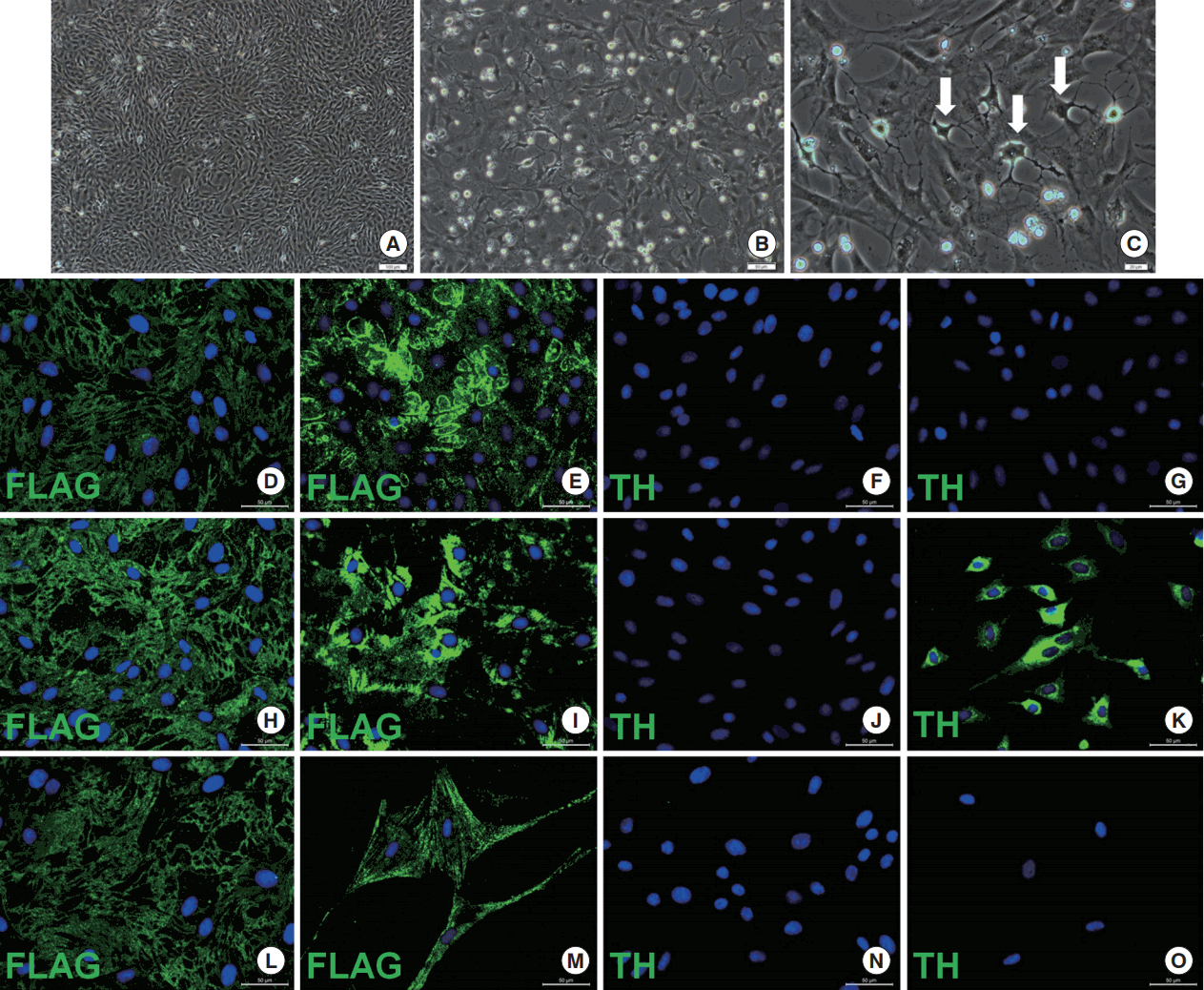
Differentiation of TH+ rBM-MSCs into neuron-like cells. After 30 hours of induction in the neuronal differentiation medium, cells displayed neuron-like morphologies (arrows) (B, C). (A) There was no neuronal differentiation in control culture. Scale bar: A, 200 µm; B, 50 µm; C, 20 µm. Immune fluorescence analysis of neural differentiated TH+ rBM-MSCs for distinct time courses of 20 hours (D–G), 30 hours (H–K), and 10 days (L–O). The expressions of FLAG (D, E, H, I, L, M) and TH (F, G, J, K, N, O) were detected in differentiated (E, I, M, F, J, N) and undifferentiated (D, H, L, F, J, N) TH+ rBM-MSCs. Nuclei were shown by DAPI in blue staining. Scale bar: D–O, 50 µm. TH, tyrosine hydroxylase; rBM-MSC, rat bone marrow mesenchymal stem cells; FLAG, protein expression system; DAPI, 4´,6-diamidino-2-phenylindole.
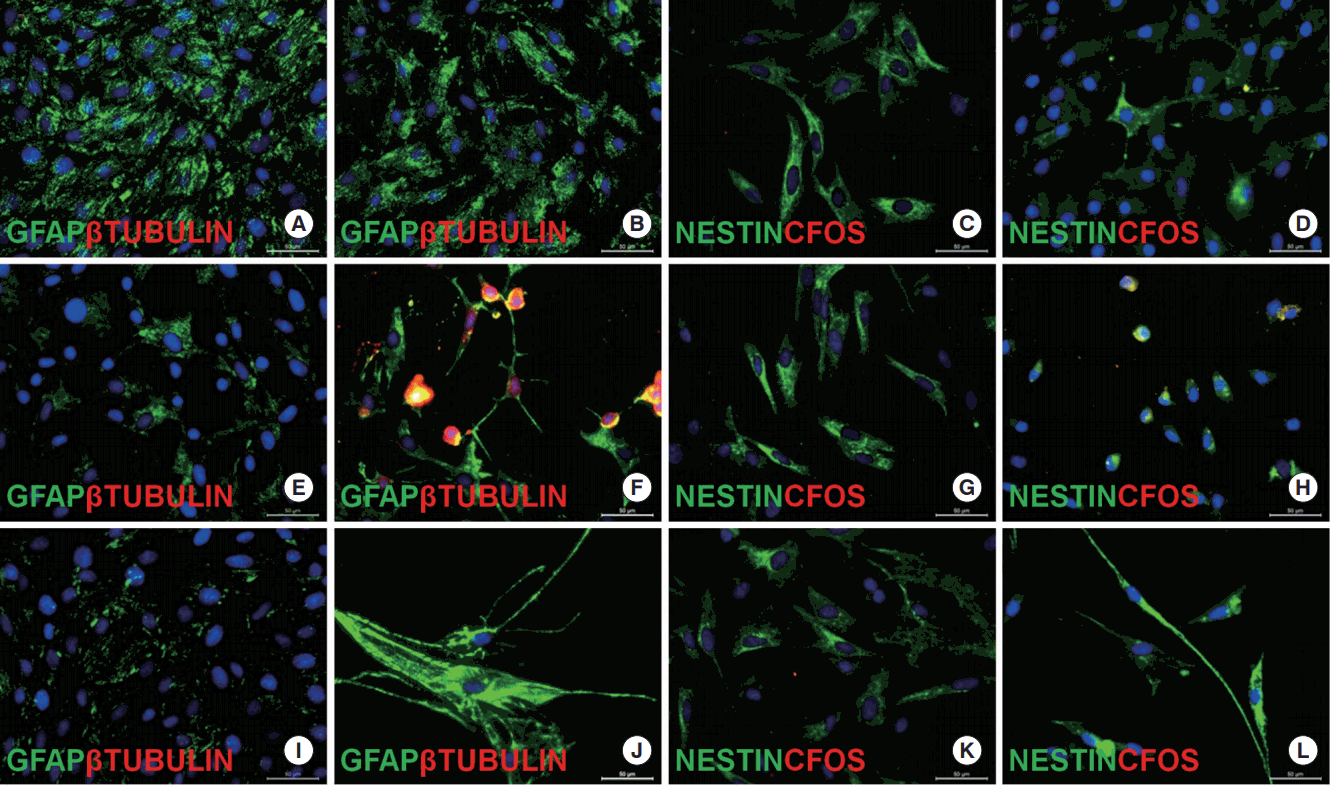
Immune fluorescence analysis of neural differentiated TH+ rBM-MSCs for the time courses of 20 hours (A–D), 30 hours (E–H), and 10 days (I–L). The expressions of GFAP, βTubulin, Nestin, and c-Fos were detected in differentiated (B, F, J, D, H, L) and undifferentiated (A, E, I, C, G, K) TH+ rBM-MSCs. Nuclei were shown by DAPI in blue staining. Scale bar: 50 µm. TH, tyrosine hydroxylase; rBM-MSC, rat bone marrow mesenchymal stem cells; DAPI, 4´,6-diamidino-2-phenylindole.
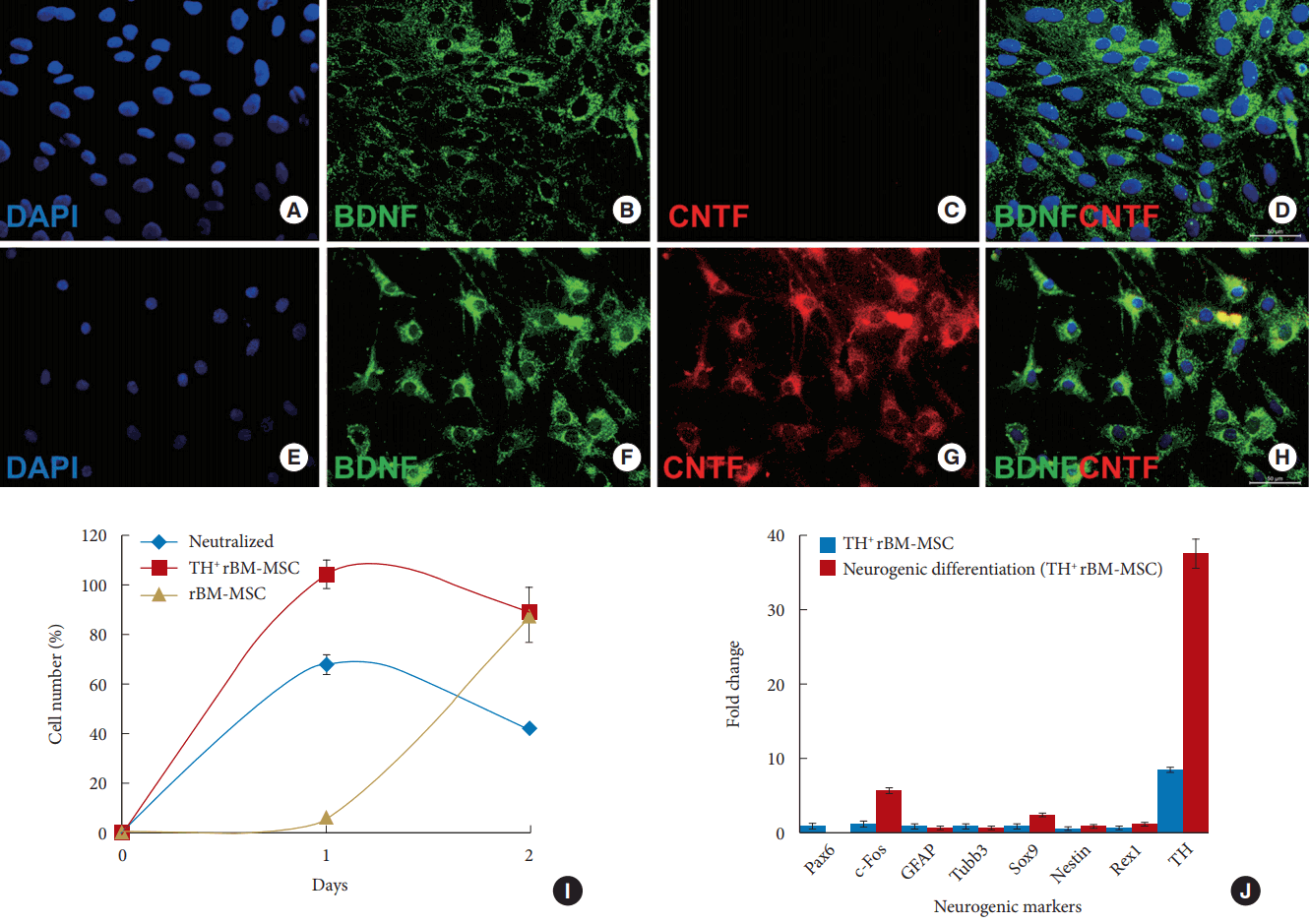
Expression of neurotrophic factors, BDNF, and CNTF, in undifferentiated (A–D) and in neurogenic differentiated TH+ rBM-MSCs for 30 hours (E–H). Nuclei were shown in blue by DAPI staining. (I) Cell proliferation analysis for neutralized TH+ rBM-MSCs, untreated TH+ rBM-MSCs, and rBM-MSCs. (J) Expressions of neurogenic markers in neurogenic differentiated/undifferentiated TH+ rBM-MSCs were estimated by real-time polymerase chain reaction. TH showed 4 times higher expression level in neurogenic differentiated TH+ rBM-MSCs. Scale Bars: 50 µm. BDNF, brain-derived neurotrophic factor; CNTF, ciliary neurotrophic factor; TH, tyrosine hydroxylase; rBM-MSC, rat bone marrow mesenchymal stem cells; DAPI, 4´,6-diamidino-2-phenylindole.
4. Neutralization of the TH Effect in Transfected Cells
The proliferation rate of TH+ rBM-MSCs increased 2 fold after TH gene transfection. To prove that this alteration is due to the TH ectopic expression, the activity of TH was suppressed by the antibody (anti-TH). The culture of cells in the complete medium supplemented with anti-TH decreased the rate of proliferation significantly by 40% compared to unneutralized cells. But proliferation rate was still higher with respect to untransformed cells (Fig. 7I). This data supports the results of cell cycle analysis, in which the S phase of the cells was elevated (Fig. 2A). Besides, the expression of proliferation stimulating chemokine, CXCL12, was increased almost by 8 fold (Fig. 2B).
DISCUSSION
PD is the second most common neurodegenerative disease of the aging central nervous system after Alzheimer dementia [16]. Stem cell-based therapies aim to provide a long-lasting symptomatic relief for PD patients. Stem cells are defined as immature cells with a capacity of self-renewal and depending on their origin, can differentiate into specialized cell types or retain the potential to differentiate into somatic cells, including dopaminergic neurons [17]. MSCs are highly promising candidates for PD treatment approaches. The application of MSCs might be effective in a PD therapeutic strategy [18,19]. Intrastriatal grafting of MSCs into adult intact rats resulted in a significant elevation of TH expression and dopamine levels in the striatum [20]. In other reports showed the functional recovery in PD rodent models following transplantation of MSCs that are indicative of a neuroprotective role in vivo [21,22].
In this study, TH gene was transferred to rBM-MSCs, and stable gene expression was obtained in the cell line by selection with G418. The continuous expression caused the alteration in cellular physiology of TH+ rBM-MSCs becoming much smaller in size. This modification might be explained by the increment of the proliferation rate, which was slightly increased after gene transfer. The expression level of TH was estimated as 0.17 ng/mL in the culture on the second day. This level is significantly lower compared with the neuronal differentiated MSC, in which TH level was estimated as 0.53 ng/mL [19]. On the other hand, the TH expression also affected the other properties of cells, like proliferation and differentiation potential.
Other studies involving the transfer of TH gene into other stem cells focus on their therapeutic effect in neurodegenerative diseases [11,23,24]. However, the impact of TH expression in stem cells on cell characteristics was sparsely investigated. In the study by Lu et al. [11], TH gene was delivered into MSCs by adeno-associated virus, and the cells were transplanted into the striatum of PD rat for functional analysis. Following injection, dopamine level was significantly improved with respect to the control rats, but the dose and timing of gene expression in the brain were found to be not regulated, as it was observed in normal physiology state. These results propose that the expression of TH in these cells might not induce to differentiate into dopaminergic or neuronal cells, as proposed by other studies [25]. Similarly, the genetically engineered rBM-MSCs with TH gene was demonstrated to contribute to regeneration process, but rather differentiation they migrated in brain tissues and increased the survival rate of surrounding cells [24]. Studies regarding in vivo differentiation of rBM-MSCs into neuronal cells have shown unsatisfactory results in parallel [26]. Their supportive effects observed following transplantation mainly occurred independently of tissue integration, and these secreted stimulating factors by the transplanted cells might be the source [27,28]. These secreted factors contributing to the regeneration process of stem cells are the neurotrophic factors: BDNF and CNTF. The expression of these proteins was increased notably in the neurogenic differentiation medium. Although these markers were not markers for mature neural cell lines, they are important for the regeneration process after neurodegenerative diseases. These are known to have function through activation of separate and specific receptors, and subsequent signaling pathways. The protective effect was reported to correlate with the endogenous up-regulation of these neurotrophic factors, which induce the activation of ERK1/2 and AKT pathways and the downstream effect on the transcription factor CREB [29]. These factors would affect the other cells in the same microenvironment as neuroprotective against apoptosis. The BDNF expression in TH+ rBM-MSCs was observed even before the neurogenic induction, but the CNTF appeared after 30 hours and went down before 60 hours of culture in differentiation medium. The cells might also respond similarly in the neural microenvironment by increasing the secretion of these factors the following transplantation into the brain tissue of animal disease model. In the comparable in vivo study related to the spinal cord injury, this paracrine effect was also observed [30].
Nestin, GFAP, and TUBB3 expressions were detected in TH+ cells without any chemical induction. The presence of these markers might provide the suitable conditions for high efficient neurogenic differentiation. Other markers, for example, Tubb and c-Fos, were expressed only after 30 hours under neurogenic differentiation condition when the neuron-like structures were observed to form. TH+rBM-MSCs could differentiate into neuron-like cells with the morphology similar to neurons in the first days, but the number of viable cells was drastically decreased after 10 days, indicating a possible elimination of cells from culture. In 10 days neurogenic differentiated group, the cells were still viable, although the number of cells was decreased considerably. To maintain the viability, the components of neurogenic differentiation media might be improved better to sustain their expressions.
Under the normal culture conditions, we did not observe any morphological difference between rBM-MSCs and TH+ rBM-MSCs. However, the cells formed reticular-like structures in the culture. Increased N-Cadherin expression, which is present in neuronal cells [31], and decreased expression of the epithelial marker, E-Cadherin, was observed. Despite the stem cell-specific surface markers were not significantly changed, the upregulation of N-Cadherin expression could indicate that TH+ rBM-MSCs are close to neuron-like cell character after TH gene transfection.
In the neurogenic differentiation, some pathways regulate these processes. It was shown that activation of Wnt/Shh pathway blocks the neurogenic differentiation [32]. In the present study, down-regulation by 4 times was observed in Wnt1 gene expression in TH+ rBM-MSCs, which could suggest that the Wnt pathway might weakly influence the inhibition of neurogenic differentiation. Besides the constitutive recombinant expression, the endogenous TH expression also observed after neurogenic induction to differentiate. Therefore, the level of TH expression was further improved during neurogenic differentiation by repression of Wnt pathway. On the other hand, Wnt signaling was reported to increase the proliferation and to impair differentiation of stem cells [33]. This contradiction in TH+ rBM-MSCs could be explained by the acceleration of cell cycle by TH gene through affecting intracellular mechanisms. This effect of TH gene could be reversed by neutralization of gene product. We found that the ratio of cell in S phase increases while that of cells in G1 phase decreased in TH+ rBM-MSCs culture, which indicates the promoting effect of TH gene transfection on cell proliferation. Furthermore, down-regulation of Cyclin E gene expression, which regulates the G1/S phase transition, might have an effect on growth. Under normal conditions, the expression of this protein is related to the increase the number of cells in G1 phase and decreases those in S phase [34]. In fact, decrease G1 phase and increase S phase could be attributed to up-regulation of CXCL12 by 8 times. Accelerated G1/S transition in cell cycle might increase the proliferation rate by the interaction of CyclinD1 and CXCL12 [35]. Eventually, up-regulation of Jag1 gene expression, which involves in G1/S phase transition by interaction with Cyclin D1, and down-regulation of Dll3 gene expression promotes the activation of Notch pathway [36]. The differentiation potential of TH+ rBM-MSCs toward neurogenic cell lineages was higher compared to the naive rBM-MSCs. However, down-regulation of Coll2a1, Aggrecan and BMP3 point out the decrease in chondrogenic differentiation potential. Similarly, Pdx1 gene down-regulation indicates the decrease in endodermal differentiation potential.
CONCLUSION
In conclusion, recombinant TH gene expression was not sufficient to transform the rBM-MSCs into neural cell lineages, but it still has the capacity to function as dopaminergic cells. After induction of neurogenic differentiation, the expression of endogenous TH gene was induced in TH+ rBM-MSCs, and the regulation of TH synthesis could be maintained, which is important for the regulation of the dopamine synthesis. The TH expression converted the rBM-MSCs into dopamine-secretingcell like cells with the differentiation potential into neural cell lineages. The TH+ rBM-MSCs had been shown previously that they could be used in the treatment of neurodegenerative diseases, like PD, but the terminal differentiation was not observed. Ectopic TH expression in rBM-MSCs improved the early neuronal differentiation markers, but rather differentiation into functional cells, the cells have the potential to be used as a neuroprotector.
Notes
The authors have nothing to disclose.
Acknowledgements
This study was supported by a grant of the Scientific and Research Council of Turkey (TÜBİTAK, Grant No.111M443). We thank Gülay Erman for her help in flow cytometry analysis and Özlem Sağlam for her technical assistance in Western Blot analysis.
SUPPLEMENTARY MATERIALS
Supplementary Fig. 1 can be found via (https://doi.org/10.14245/ns.1836010.005).
