Diagnosing Pseudoarthrosis After Anterior Cervical Discectomy and Fusion
Article information
Abstract
Radiographic confirmation of fusion after anterior cervical discectomy and fusion (ACDF) surgery is a critical aspect of determining surgical success. However, there is a lack of established diagnostic radiographic parameters for pseudoarthrosis. The purpose of this study is to summarize the findings of previous studies, review the advantages and disadvantages of frequently employed diagnostic criteria, and present our recommended protocol of fusion assessment. This study identified randomized controlled trials, case-control studies, and prospective and retrospective cohort studies reporting on spinal fusion and how successful fusion after ACDF. Among the 39 articles reviewed, bridging bone across the operated levels on static radiographs was the most commonly used criteria to confirm fusion (31 of 39, 79%). Dynamic flexion-extension radiographs were used to assess for interspinous movement (ISM) (22 of 39, 56.4%) and change in Cobb angle (12 of 39, 30.8%). Computed tomography (CT) based findings (21 of 39, 53.8%) were employed in ambiguous cases with improved sensitivity and specificity. Reconstructed CT scans were used to assess for intragraft bridging bone and extragraft bridging bone (ExGBB). ExGBB were proved to have the highest diagnostic sensitivity and specificity for pseudoarthrosis detection when compared to all other radiographic criteria. The ISM <1 mm on dynamic flexion-extension radiographs had high diagnostic sensitivity and specificity as well. After our reviewing, we recommend using dynamic lateral flexion-extension cervical spine radiographs at 150% magnificationin which the interspinous motion <1 mm and superjacent interspinous motion ≥4 mm confirms fusion. In ambiguous cases, we recommend using reconstructed CT scans to evaluate for ExGBB.
INTRODUCTION
The aim of anterior cervical discectomy and fusion (ACDF) surgery is to provide the patient with adequate decompression and rapid fusion to treat cervical degenerative disease, ultimately reducing symptoms of neck pain, radiculopathy, and myelopathy. Since its introduction in the 1950s, ACDF has a proven track record of high fusion rates [1]. However, pseudoarthrosis and adjacent segment disease are known complications of the procedure that may lead to persistent symptoms requiring further revisions surgery which is complicated by prolonged hospital stay and increased morbidity [1-4]. The true etiology of pseudoarthrosis is difficult to ascertain, but there are known risk factors, which include patient factors and surgical factors such as multilevel fusions, instrumentation choice and bone grafts used for the case [5-7]. The reported fusion rate following anterior cervical spine surgery with fixation is as high as 96% [8], but the majority of reoperation after the anterior approach was due to pseudoarthorsis [9]. Numerous imaging modalities and diagnostic radiographic criteria to determine the fusion status following ACDF have been described and used in recent literature. However, only a few of these radiographic parameters were rigorously analyzed to validate their accuracy and reliability so far. This inconsistency in reporting pseudoarthrosis following ACDF stems from a lack of established radiographic parameters in the literature. This current study aims to review recent literature over the last decade (2008 to 2018) to evaluate the different imaging modalities and radiographic parameters used in prior works. In this review article, we will summarize the findings of previous studies, review the advantages and disadvantages of frequently employed diagnostic criteria, and present our recommended protocol of fusion status assessment with a case example.
MATERIALS AND METHODS
1. Data Source
This study identified longitudinal studies including randomized controlled trials (RCT), observational case-control studies, and prospective and retrospective cohort studies reporting on the success of spinal fusion and how successful fusion was identified and defined after ACDF. The National Institutes of Health PubMed database was queried using a combination of free and medical subject headings search parameters related to the surgical intervention (e.g., “anterior cervical discectomy and fusion,” “ACDF,” “cervical arthrodesis”) and outcome of interest (e.g., “fusion,” “pseudoarthrosis,” “nonunion,” “treatment outcomes”) in major journals. There were no language restrictions on potential studies. Studies that were published within the approximately 10-year period between January 1, 2008 and June 30, 2018 were included for inclusion in this study to assess for recent trends in radiographic pseudoarthrosis diagnosis. Studies without clear radiographic fusion criteria, literature review, case reports, and fusion assessment of upper cervical spine and craniocervical junction were excluded. A list of relevant articles was identified using these search terms by 3 authors (WL, AH, VB) and were manually screened for inclusion in this current review. All longitudinal studies that reported on a cohort of patients who underwent ACDF for any indication and who were followed postoperatively for fusion status were included in this study.
2. Data Extraction
After an initial screen of abstracts and article titles, we obtained full text articles of all potential studies. Three reviewers independently assessed the articles using the inclusion criteria until a consensus was reached. Relevant data identified from each article was the type of study (RCT, case-control, cohort studies), level of evidence, number of patients included in the study, number of intervertebral levels assessed in the study, imaging modality used to assess spinal fusion (e.g., plain films, computed topography [CT], etc.), and how fusion was assessed from imaging (e.g., trabecular bone bridge, motion, etc.).
RESULTS
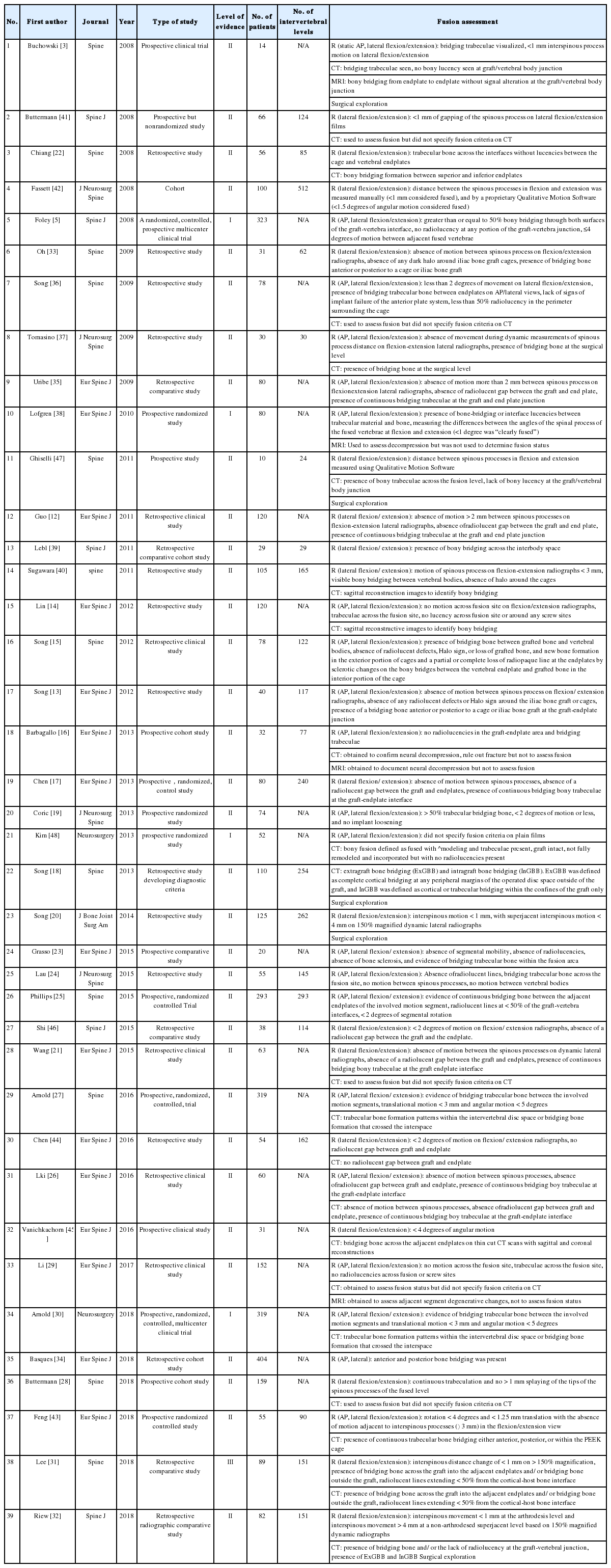
In total, 39 articles (Table 1) published between January 1, 2008 and June 30, 2018 met our inclusion criteria [10-49]. The various imaging modalities used to assess fusion status in these studies included radiographs (static and dynamic lateral) and CT imaging. No studies were identified that reported cervical spine fusion or pseudoarthrosis based solely on history or physical examination.
1. Radiographic Assessment
The most frequently used radiographic diagnostic criteria for pseudoarthrosis after ACDF was an absence of bridging trabeculae across the fused levels on static radiographs (31 of 39 used this definition, 79%) [10-40]. Fusion status was also frequently assessed using dynamic lateral radiographs. Using this imaging technique, authors measured ISM (22 of 39, 56.4%) [10,13,14,16,19-22,25,28,29,31,33-39,41-43] or used the Cobb angle method (12 of 39, 30.8%) [11,15,17,27,32,34,37,42-46] to assess cervical fusion. No consensus was reached regarding the amount of motion for evaluation of cervical fusion on dynamic lateral radiographs. For the interspinous process method, no motion (9 of 22, 40.9%) [13,14,17,21,24,26,29,33,37], under 1 millimeter (mm) (7 of 22, 31.8%) [10,20,28,31,32,41,42], under 2 mm (2 of 22, 9.1%) [12,35], and under 3 mm of motion (4 of 22, 18.2%) [27,30,40,43] between spinous process were all used as cutoff values for a definition of fusion. Using the Cobb angle method, changes of 1 degree (1 of 12, 8.3%) [38], 1.5 degrees (1 of 12, 8.3%) [42], 2 degrees (5 of 12, 41.7%) [19,25,36,44,46], 4 degrees (4 of 12, 25.0%) [11,43,45], and 5 degrees (2 of 12, 16.7%) [27,30] were all used in various studies.
2. CT Assessment
CT scans were the most commonly used advanced imaging modality to assess fusion status (21/39, 53.8%) [10,12,15,16,19,22,26,29,33-38,41,43,45-49]. Fusion was identified in these studies by assessing for continuous bridging bone at the cage or graft endplate interface. Pseudoarthrosis was identified as a radiolucent gap across the fused levels. Reconstructed multi-axial CT scans were also compiled to assess for intragraft bone bridging (InGBB) and extragraft bone bridging (ExGBB) [18,32].
DISCUSSION
Achieving fusion after ACDF is critical to attain predictable postoperative pain relief and functional recovery. Pseudoarthrosis is an uncommon, but known complication after ACDF that leads to persistent unresolved symptoms, which often requires revision surgeries [1-4]. Although the gold standard to assess pseudoarthrosis is operative exploration of fusion mass, the preoperative radiographic options to determine fusion status is still poorly described in the literature. The pseudoarthrosis rate is affected by patient factors (diabetes, smoking, etc.), fusion levels, graft choice, and surgical instrumentation, but the true etiology remains difficult to establish given high rates of asymptomatic patients and inconsistent diagnostic radiographic parameters [32,50-53]. Therefore, the purpose of this review study was to inspect the articles published over the past decade to assess which imaging modality and radiographic parameters were used to diagnose fusion after ACDF to identify current trends and consensus protocols for radiographical assessment of fusion status.
In this systematic review study of 39 articles published between January 1, 2008 and June 30, 2018, the fusion status after ACDF was confirmed with different imaging modalities in addition to the history and physical exam of the patients. All the studies included cervical spine radiographs as one of the tools used to assess the fusion status and most studies used more than one criterion, which suggests a more stringent criteria for fusion assessment since our previous review article [53]. The use of cervical spine films as a first line to diagnose pseduoarthrosis stems from low costs, easy accessibility, and low radiation for the patient.
Static anteroposterior and lateral cervical spine films are consistently described as the initial approach to assess ACDF postoperative fusion status in recent literature. These films expose the patients to low-dose radiation and provide the clinician with valuable information regarding fusion status. In the static films, fusion is confirmed with formation of osseous bridging between the graft and vertebral body across the fused levels. Pseudoarthrosis is diagnosed with static films when there is an absence of this osseous bridging and a presence of haloing or lucency around the graft [10,33]. However, pseudoarthrosis diagnosed in plain films only correlates between 43%–82% with pseudoarthrosis detected during operative exploration of fusion mass, which is most likely secondary to the inability of films to accurately assess bony morphology with an implant in place [54]. Among the 39 included articles, only two studies determined cervical spine fusion status or pseudoarthrosis based solely on static radiographs as the only modality [23,40]. Given the lower rate of pseudoarthrosis detection using just the static films, the recent trends for pseudoarthorsis diagnosis have moved from relying on static radiographs to dynamic radiographs and advanced imaging. The dynamic lateral flexion-extension cervical spine films provide an improved method of assessing postoperative ACDF fusion status. The dynamic films can either evaluate for the ISM or the change in Cobb angle, both of which have a wide range of diagnostic radiographic parameters in literature. The ISM measurement was much more prevalent in recent works, which accounted for more than half of the literature reviewed in this study. The ISM measurement leading to no movement (40.9%) [13,14,17,21,24,26,29,33,37] and the Cobb angle change of less than 2 degrees (41.7%) [19,25,36,44,46] were the most common method to confirm fusion after ACDF in recent literature. Song et al. [20] evaluated the accuracy of different radiographic parameters used to assess fusion status after ACDF and reported that less than 1 mm of ISM showed the best accuracy and agreement with intraoperative exploration. In this study, he demonstrated increased inter and intraobserver reliability of ISM measurement using magnified 150% radiographs compared to the 25% and 100% magnification, which was observed in future studies as well [20,31]. Another comparative study conducted by Riew et al. [32] demonstrated that <1 mm ISM cutoff value showed a reliable accuracy compared with conventional CT based bridging bone criteria and showed acceptable sensitivity and specificity second only to ExGBB. Although no motion in ISM is frequently used in recent literature to validate fusion, there is concern that this method may increase the number of reported pseudoarthrosis since micromotion still exists within the solidly fused levels on dynamic films. On the other hand, the cutoff value of less than 2 mm of ISM measurement may overestimate fusion rate, leading to missed diagnosis of pseudoarthrosis. Also, there is no literature confirming the validity of no motion or less than 2 mm of ISM as a reliable method of pseudoarthrosis detection. An important aspect of measuring the ISM is to also be mindful of the superjacent ISM which should be more than 4 mm to increase negative predictive value and sensitivity. Although the no motion of ISM is frequently described in recent literature as a method of pseudoarthrosis diagnosis, only the ISM less than 1mm have been shown to have reliable accuracy.
The dynamic flexion-extension films can also assess for the change in Cobb angle between the adjacent fused vertebrae to determine postoperative fusion status. Recent literature frequently employed the change in Cobb angle as a criteria to confirm fusion (30.8%) [11,15,17,27,32,34,37,42-46], but the radiographic parameters ranged from 1.5 to 5 degrees. Cannada et al. [4] showed that changes in Cobb angle of 2 degrees lead to a sensitivity of 82% and specificity of 39%. This significantly improved with the Cobb angle change of 4 degrees that resulted in a specificity of 100% [4]. However, the Cobb angle measurement may be a less reproducible form of radiographic parameter compared to the ISM measurement. The Cobb angle measurement is closely associated with the instantaneous center of rotation while obtaining the dynamic cervical spine films, making an accurate and consistent angle measurement difficult. Although there is evidence that combining quantitative motion analysis software with dynamic radiographs may yield objective and reliable numbers compared to manual or subjective measurements, the limitation of specialized technology and software availability make this less useful [42,47]. The difficulty of consistently reproducing the Cobb angle measurement makes this a less appealing method to diagnose pseudoarthrosis for the authors.
In addition to plain radiographs, CT scans were frequently used (53.8%) [10,12,15,16,19,22,26,29,33-38,41,43,45-49], to assess pseudoarthrosis following ambiguous radiographic findings. The interobserver reliability of predicting pseudoarthrosis is better using the CT scan despite the metal artifacts compared to dynamic cervical spine films [10]. The radiographic parameter for CT scan based fusion diagnosis is still in flux in recent literature. Kim et al defined bony fusion as “fused with remodeling and trabeculae present” or “graft intact, not fully remodeled and incorporated, but no lucency present,” which is more of a general vague description of fusion status [48]. Other CT scan based parameters used the lack of motion in fused segments and at times no specific parameter was described for fusion assessment [14,28,29,37,47,55]. Song et al. [18] first described the ExGBB and InGBB on CT scans to subcategorize the areas of achieved fusion in 2013. Riew et al. [32] further evaluated the ExGBB and InGBB in multiaxial reconstructed CT scans and reported that ExGBB had the highest sensitivity and specificity and acceptable accuracy to detect pseudoarthrosis, but InGBB was worse than guessing. Although the CT scan is superior to standard films in evaluating bony fusion status, it is limited to findings derived from a static moment in time. It fails to assess for dynamic changes in the cervical spine during motion, which may leave out some cases of pseudoarthrosis seen only with movement. Also, the CT scan based fusion status is based on subjective interpretation compared to the objective measurement findings on standard films, which make the results more vulnerable to both type I and type II errors [47]. In addition, because of the imaging features of cortical allografts, the CT evaluation may actually omit the nonunion and overstate the fusion rates, particularly during the early post-operative follow-up period. The published CT based pseudoarthrosis diagnosis is inconsistent with other radiographic nonunion indicators including internal fixation failure, peri-instrument halo signs, and cystic changes around the grafts [20]. Overall, the CT scan is a valuable tool used to confirm equivocal findings on standard radiographs given its improved pseudoarthrosis detection rate, but care must be taken prior to obtaining the imaging given its limitations.
CONCLUSION
Diagnosing pseduoarthrosis after ACDF is controversial and the literature is inconsistent in objectively evaluating postoperative radiographic findings. After reviewing the recent studies, we find that no one single method is absolutely perfect for the diagnosis of pseudoarthrosis. After taking the advantages and disadvantages into consideration, we recommend first using the dynamic lateral flexion-extension cervical spine films in 150% magnification as the initial method for evaluation, since this is economically prudent, quick, and most informative of dynamic cervical spine movements with low radiation for the patient. The interspinous motion <1 mm and superjacent interspinous motion ≥4 mm confirms the fusion diagnosis in the dynamic films (Fig. 1). In ambiguous cases (Fig. 2), we recommend using the reconstructed multiaxial CT scans to evaluate for ExGBB given its superior diagnostic qualities (Fig. 3).

Measurement of interspinous movement (ISM) at superjacent level (C4–5) and operated levels (C5–7) on the 150% magnified flexion and extension radiographs. The superjacent ISM at C4–5 (A and a) is 10.2 mm, which indicates adequate dynamic motion ( > 4 mm). ISM at C5–6 (B and b) is 0.21 mm, which is consistent with our definition of fusion ( < 1 mm). ISM at C6–7 (C and c) is 5.13 mm, which indicates pseudoarthrosis ( > 1 mm).

Evaluating bone bridging on multiaxial reconstructed coronal and sagittal computed tomographic images. It appears to be fused with bone bridging formation at both C5–6 (A and a) and C6–7 (B and b) levels.

Evaluating extragraft bridging bone (ExGBB) on multiaxial reconstructed coronal and sagittal computed tomographic images. The top level (C5–6) shows ExGBB on both the coronal and sagittal images (A and a). The bottom level (C6–7) shows intragraft bone bridging on the coronal view (B) but demonstrates a cleft and no ExGBB on sagittal image (b), which indicates pseudoarthrosis. This is consistent with the results of interspinous movement evaluation on X-rays and was confirmed to be pseudoarthrosis with intraoperative exploration.
Notes
The authors have nothing to disclose.