 |
 |
- Search
|
|
||
Abstract
Determining the optimal surgical method for cervical ossification of the posterior longitudinal ligament (OPLL) is challenging. The surgical indication should be made based on not only radiological findings, but also the patientŌĆÖs age, preoperative neurological findings, social background, activities of daily life, and the presence or absence of comorbid diseases. Anterior resection for OPLL with or without wide corpectomy and fusion, posterior decompression with or without relatively long fusion, or anterior and posterior combined surgery may be considered. When evaluating the clinical condition of patients with cervical OPLL before surgery, various radiological parameters should be carefully considered, including the number of spinal segments involved, the cervical alignment or tilt angle, the relationship between OPLL and the C2ŌĆō7 line (termed the ŌĆ£K-lineŌĆØ), the occupying ratio of OPLL, and the involvement of dural ossification. The objective of this article is to review the radiological parameters in current use for deciding upon the optimal surgical strategy and for predicting surgical outcomes, focusing on cervical OPLL.
Cervical ossification of the posterior longitudinal ligament (OPLL) is generally clinically silent or gradually progressive but can be acute after even minor trauma [1]. Surgical intervention is usually necessary if there are moderate or severe neurological deficits. However, determining the optimal surgical method for cervical OPLL is not straightforward. Anterior resection for OPLL with or without wide corpectomy and anterior fusion may provide a useful option (Fig. 1A, B) [2,3]. However, anterior surgery carries surgery-related risks such as cerebrospinal fluid (CSF) leakage, spinal cord damage, instrumentation-related complications, and dyspnea or dysphagia. In contrast, posterior decompression with or without relatively long fusion offers an alternative that may allow decompression of the spinal cord and correction of spinal alignment to some extent (Fig. 1C, D) [4-6]. Although posterior surgery may reduce the surgical risks associated with anterior cervical surgery, it still carries a high risk of nerve root tethering or cervical foraminal stenosis or additional neurological deficit related to the remaining OPLL [7,8]. Anterior and posterior combined surgery may be another surgical option (Fig. 1E, F) [9,10]. Various radiological parameters are used to evaluate cervical OPLL and to determine the surgical strategy. Factors such as the number of involved spinal segments, cervical alignment or T1 slope, the relationship between OPLL and the C2ŌĆō7 line (termed the ŌĆ£K-lineŌĆØ), occupying ratio of OPLL, and involvement of dural ossification need to be carefully considered before surgery (Fig. 2A, B). The objective of this article is to review the radiological parameters in current use for deciding the optimal surgical strategy and for predicting the surgical outcome, focusing on cervical OPLL. This review article includes classification of cervical OPLL based on lateral radiograph or sagittal computed tomography (CT) images, sagittal relationship between OPLL and the ŌĆ£K-line,ŌĆØ cross-sectional shape of spinal cord, dural ossification associated with OPLL, thickness and width of the ossified mass with ŌĆ£Rule of Nine,ŌĆØ the radiological parameters for deciding the optimal surgical strategy and discussion.
The Investigation Committee for Ossification of the Spinal Ligaments (part of the Japanese Ministry of Health, Labor and Welfare) developed a classification system for cervical OPLL [11] comprising 4 types: (1) continuous: a long lesion extending over several vertebral bodies; (2) segmental: one or several separate lesions behind the vertebral bodies; (3) mixed: a combination of the continuous and segmental types; and (4) localized (focal or circumscribed): located mainly just behind a disc space, ossification surrounding intervertebral disc herniation (Fig. 3). The frequency of each type has been reported as continuous, 25.97%; segmental, 34.26%; mixed, 32.47%; and localized, 7.30% [12]. In a different system, Iwasaki et al. [5] classified OPLL as either plateau-shape or hill-shape type based on the outline of the OPLL on sagittal CT images or a lateral radiograph of the cervical spine (Fig. 4). In plateau-shape ossification, the spinal canal is usually relatively narrow without localized massive ossification, whereas in hill-shape type, OPLL is seen as a massive beak-shaped ossification localized at certain levels. Iwasaki et al. [5] described poor surgical outcomes for laminoplasty in the case of an OPLL occupying ratio >60%, and/or when the ossified lesion is hill-shaped with sharp angulation of the spinal cord.
Fujiyoshi et al. [13] proposed the K-line concept for deciding the surgical approach for cervical OPLL (Fig. 2B). The K-line is a straight line marked on a neutral lateral radiograph of the cervical spine and connects the midpoints of the spinal canal from C2 to C7. In patients whose C7 vertebra cannot be visualized on the radiograph because of overlying shoulder or scapula, the K-line is drawn on midsagittal T2-weighted magnetic resonance imaging (MRI). Cervical OPLL can be divided into 2 groups based on the K-line: K-line positive (+) and K-line negative (ŌĆō). In the K-line (+) group, OPLL exceeds the K-line; in the K-line (ŌĆō) group, OPLL exceeds the K-line. Fujiyoshi et al. [13] suggested that in the K-line (ŌĆō) group, posterior decompression surgery would be unable to achieve a satisfactory posterior shift of the spinal cord and thus satisfactory neurologic improvement. Dynamic changes in the cross-sectional area of the spinal cord can be significantly affected by OPLL. At the level of most severe stenosis, the spinal cord becomes narrower at all disc levels during extension. In patients with kyphosis and the K-line (ŌĆō) group, the spinal cord becomes narrower during flexion. Cervical flexion may induce more significant spinal cord compression in patients with kyphosis and K-line (ŌĆō) patients [13-15]. The K-line concept appears to be a simple and practical method for determining the optimal surgical approach.
The cross-sectional shape of the spinal cord on T2-weighted MRI at the level of maximal compression can be divided into 3 types: boomerang, teardrop, and triangular [16]. Matsuyama et al. [16] described the boomerang type as having a convex posterior surface and concave anterior surface with smooth rounded corners; the teardrop type as having a convex posterior surface and concave anterior surface with a smooth round corner on only one side; and the triangular type as having angular lateral surfaces and a flat anterior surface (Fig. 5). Additionally, Matsuyama et al. [16] reported a contrasting expansion pattern of the spinal cord on intraoperative ultrasonography compared with that seen on postoperative MRI. They suggested that poor postoperative clinical recovery correlates with a lack of postoperative cord expansion on either MRI or ultrasound evaluations. The patients with either teardrop or boomerang deformity demonstrate a relatively good recovery rate, although those with triangular deformity may demonstrate a relatively poor recovery rate. The spinal cord may be severely atrophic or degenerated in case of triangular deformity compared to teardrop or boomerang deformity.
In the case of cervical OPLL with dural ossification, CSF leakage can become highly problematic when performing direct anterior removal of OPLL involving dura mater [17]. Hida et al. [18] first reported the double-layer sign, which indicates dural ossification, as a central hypodense line of hypertrophied ligament between anterior and posterior rims of hyperdense ossification on axial CT. Mizuno et al. [19] further classified cases of dural ossification into 3 types (isolated, double layer, and en bloc) according to their appearance on axial CT (Fig. 6). The isolated type has a small ventral hyperdense mass in the canal, without related OPLL. The double-layer type has anterior and posterior rims of hyperdense ossification separated by a central hypodense area. The en bloc type has a hyperdense mass with a downward and/or upward tail along the dura mater and an en bloc hyperdense mass. They also suggested that isolated dural ossification can occur, in which there is ossification of the dura mater without any correlation to OPLL. En bloc dural ossification is characterized by a single hyperdense mass on CT that cannot be differentiated between dural ossification and a component of OPLL. Sagittal CT reconstruction or polytomography studies that reveal OPLL by the hyperdense meningeal tail sign can diagnose this type of dural ossification. Double-layer dural ossification is the most common type in this radiological categorization system. Yang et al. [20] classified the double-layer sign according to the morphological features of the ossified and central hypodense mass on axial CT into 3 types (A, B, and C), having crescent shape, short-straight shape, and long-straight shape, respectively (Table 1). Type A-crescent shape was the most common (55.4%), followed by type B-short-straight shape (38.1%) and type C-long-straight shape (6.5%). Also, they reported that the incidence of CSF leakage after anterior resection of OPLL was only 3.9% in type A-crescent shape, but 17.1% in type B-short-straight shape and 100% in type C-long-straight shape. They concluded that in patients with type C-long-straight shape of the double-layer sign, postoperative CSF leakage would be almost inevitable following anterior resection of OPLL. Epstein [21] proposed the ŌĆ£C signŌĆØ to indicate dural penetration by OPLL. This sign is characterized by the extremely lateral, curved, and irregular features of the OPLL mass, with a unique hook-like configuration, in addition to the features typical of a large focal OPLL mass. This sign indicates the likely imbrication of the dura mater (Fig. 7).
Yang et al. [22] proposed the ŌĆ£Rule of NineŌĆØ to determine whether cervical OPLL can be resected completely and safely. On an axial image at the maximum ossified level, which is the thickest and widest part of OPLL, the baseline is a straight line along the posterior margin of the vertebral body, extending to the junction of the vertebral body and the pedicle on each side. The distance between the posterior margin of the vertebral body and the root of the spinous process is divided into 3 equal parts by 2 parallel lines, termed the safety and danger lines, to divide the spinal canal (from anterior to posterior) into 3 zones: the safety, intermediate, and danger zones. The baseline is divided lengthwise by 2 perpendicular lines into 3 equal parts, to divide the spinal canal into 9 resection units (Fig. 8). Yang et al. [22] reported that as the thickness and width of the ossified mass increase, the incidence of spinal cord injury and CSF leakage also increases, especially in cases in which the ossified mass exceeds the danger line and extends into 2 resection units.
It is thought that spinal cord cross-sectional area (SCCSA) slowly becomes narrower in patients with OPLL, but the influence of dynamic factors may be another important factor in cervical myelopathy. Liu et al. [23] found that in patients with severely limited range of movement in the cervical spine, cervical myelopathy does not always develop despite spinal stenosis (6 mm <space available for the spinal cord <14 mm). This indicates that in addition to static factors, dynamic factors such as listhesis or hypermobility at the discontinuity of the ossified part also play important roles in the development of myelopathy, especially in mixed and segmental OPLL [24]. Ito et al. [25] studied the change in SCCSA between the neck extension and flexion positions for each type of OPLL. The average change in SCCSA between extension and flexion was 7.4┬▒5.1 mm2 in the connection part (completely connected part which was well seen in the continuous type OPLL), 5.8┬▒6.0 mm2 in the coating part (incompletely connected part which was seen well in the mixed-type OPLL), and 6.7┬▒6.4 mm2 in the nonconnection part (segmental type of OPLL) of the OPLL group. Although there was no statistically significant difference, they concluded that dynamic factors influence spinal cord compression, even in the connection part of OPLL. The dynamic factors can be observed even in cervical myelopathy patients with a continuous type of OPLL. Also, Chen et al. [26] reported fracture of the ossified ligament, termed the ŌĆ£broken sign,ŌĆØ which indicates a degree of dynamic mobility the ossified intervertebral connecting part of OPLL (Fig. 9).
Kawaguchi et al. [27] classified OPLL patients into 2 groups according to ossification of the ligament, including the transverse atlantal ligament at C1 (behind the dens): the C1(+) and C1(ŌĆō) groups. They reported that 25% of patients with cervical OPLL had an ossified lesion in the upper cervical spine. In this C1(+) group, the continuous type of OPLL was common, and the segmental type was rare compared with the C1(ŌĆō) group. Because the C1(+) group more frequently had OPLLs in the thoracic and/or lumbar spine, investigation of the whole spine is recommended in these patients.
In the surgical treatment of cervical OPLL and cervical spondylotic myelopathy, posterior decompression surgeries such as cervical laminoplasty occasionally fail to relieve anterior compression of the spinal cord caused by preoperatively existing cervical kyphosis and/or intervertebral disc bulging [5,28,29]. These anterior components prevent the patientŌĆÖs neurological recovery because the decompression mechanism after laminoplasty depends only on the posterior shifting of the spinal cord [28-30]. Taniyama et al. [31,32] investigated whether a predictive indicator termed ŌĆ£modified K-line,ŌĆØ could determine during the surgical decision-making process whether laminoplasty would be ineffective or incomplete for decompression (Fig. 10). The modified K-line is defined as the line connecting the midpoints of the spinal cord between C2 and C7 on a T1-weighted midsagittal slice of preoperative MRI. They determined the minimum interval between the tip of local kyphosis including disc bulging and the modified K-line. They concluded that a minimum interval of 4.0 mm could be critical to indicate a high likelihood of postoperative residual anterior compression of the spinal cord (such as kyphosis or bulging disc) after laminoplasty.
The Scoliosis Research Society has proposed various radiological parameters for assessment of the cervical spine [33-35]. C2ŌĆō7 angle is measured by formal Cobb methods, between the horizontal line of the C2 lower endplate and the horizontal line of the C7 inferior endplate (Fig. 11). Thoracic inlet angle (TIA), a constant morphological parameter that is not influenced by posture, is defined as the angle formed by intersection of the line perpendicular to the center of the T1 upper endplate and the superior anterior sternum (Fig. 11). As a measure of the influence of posture, T1 slope is defined as the angle formed between a horizontal line and the T1 upper endplate. Neck tilt (NT) is defined as the angle formed by a vertical line passing through the superior anterior sternum and the line connecting the center of the T1 upper endplate with the superior anterior sternum. Geometrically, the formula ŌĆ£TIA=T1 slope+NTŌĆØ can be derived.
Sagittal vertical axis (SVA) values are standard measurements taken to assess deformity in the thoracolumbar spine [35-37]. Previous researchers have concluded that positive sagittal malalignment, defined as a C7 plumb line greater than 50 mm anterior to the posterosuperior aspect of the sacrum, is associated with a deterioration of quality of life in patients with adult spinal deformity [38-46]. It has also been suggested that C2ŌĆō7 SVA value positively correlated with Neck Disability Index scores (Fig. 11) [36]. T1 slope and TIA concerning cervical sagittal balance may be as important as pelvic incidence concerning lumbar lordosis. The shape and orientation of the T1 vertebral body affect the amount of lordosis required to maintain sagittal balance of the cervical spine and maintain upright horizontal gaze. Previous investigators have used X-ray and CT to examine the relationship between T1 slope and cervical sagittal alignment [47-50]. As the T1 slope increases, so does C2ŌĆō7 lordosis to maintain forward gaze, thereby resulting in a greater degree of lordotic curvature. Similarly, C2ŌĆō7 SVA tends to increase as T1 slope increases. Because T1 slope indicates a compensatory change related to thoracolumbar sagittal imbalance, we should take overall spinal sagittal balance into consideration, not only cervical sagittal alignment. It has been reported that the compensatory mechanism of the thoracic spine curve becomes difficult when the T1 slope is >25┬░ [50]. In addition to sagittal balance, evaluation of the preoperative presence of intervertebral instability and preoperative estimation of the development of the posterior neck musculature are also very important in preventing unexpected postoperative neck pain, postoperative malalignment such as kyphotic deformity, and nerve root palsy due to postoperative foraminal stenosis. Furthermore, measurements of K-line and T1 slope on MRI acquired in the sitting position and evaluation on dynamic MRI have also begun [51-53]. These radiological parameters may be useful and informative in estimating the surgical outcome.
In the various classification systems of the spinal cord, factors such as its shape, distribution, and the degree of compression undergo dynamic change. The progression of OPLL, especially ossification of the dura mater, is one of the most important aspects of preoperative radiological evaluation. Patients with dural ossification are at a higher risk of CSF leakage during anterior cervical removal of OPLL because it is technically challenging to separate OPLL from ossified dura mater. To prevent leakage of CSF both during and after surgery, the surgeon can select a surgical strategy such as the floating method, prepare abdominal fat tissue for grafting onto the dura mater, or initiate lumbar drainage immediately after surgery. If indirect decompression of the spinal cord from OPLL is performed by a posterior decompression surgery such as laminoplasty, the surgeon should be aware of postoperative dynamic change in OPLL. Chiba et al. [7] and Hirabayashi et al. [8] reported that progression of OPLL was more likely to occur in the early phase after laminoplasty and was less likely to occur in the late phase. Moreover, some researchers have suggested that dynamic factors stimulate the progression of OPLL, and stabilization of the range of motion can reduce the progression of OPLL [54-59]. There are options for posterior decompression and fixation if it is difficult to directly remove OPLL by the anterior approach for any reason, especially when combined with kyphosis.
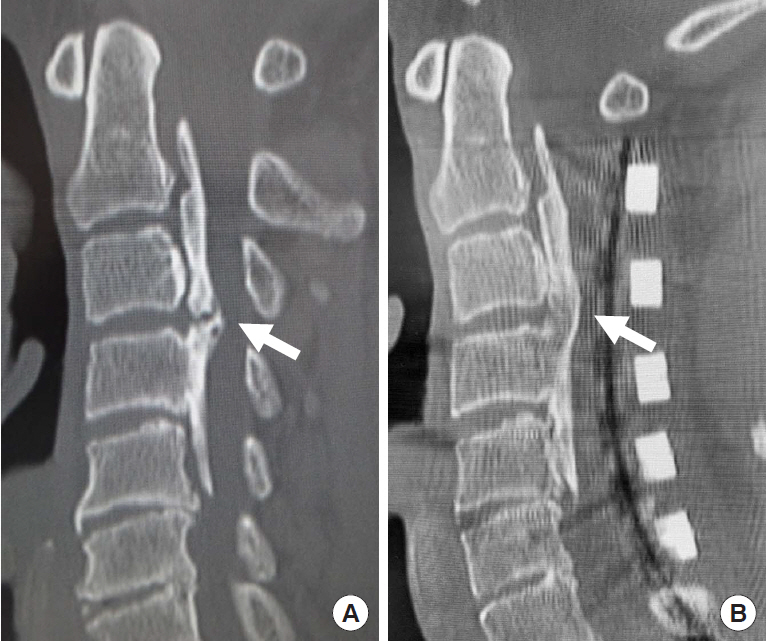
Katsumi et al. [59] evaluated whether laminoplasty with instrumented fusion (posterior decompression and fusion [PDF] group) suppresses the progression of OPLL in comparison with stand-alone laminoplasty (LP group), using their novel OPLL volume analysis with 3-dimensional CT. They reported that the mean annual rate of lesion increase was 2.0┬▒1.7%/yr (range, ŌĆō3.0% to 5.3%) in the PDF group and 7.5┬▒5.6%/yr (range, 1.0%ŌĆō19.2%) in the LP group, and there were significant differences in the annual rate of increase between the 2 groups (p<0.001). They also found that regarding change of OPLL type in the PDF group, those with mixed-type OPLL more often changed to continuous type, and the segmental-type OPLL more often changed to mixed type, compared with the LP group. Sequential morphological change in OPLL itself can be recognized after posterior fusion surgery (Fig. 12). Depending on the patientŌĆÖs condition, a 2-stage surgery might be another option; e.g., initial posterior indirect decompression of spinal cord followed by direct anterior removal of OPLL if the patientŌĆÖs symptoms develop. It is noteworthy that OPLL may undergo dynamic change over a long time and influence the patientŌĆÖs activities of daily life. We also emphasize the importance of long-term follow-up of OPLL patients.
To achieve a satisfactory outcome, various points need to be discussed carefully before surgery for cervical OPLL. In this article, the authors focused on the radiological evaluation process. However, the surgical indication should be made based on not only the results of radiological evaluation but also the patientŌĆÖs age, preoperative neurological findings, social background, activities of daily life, and the presence or absence of comorbid diseases. The authors sincerely anticipate that future clinical research will be more helpful for surgeons to determine the optimal surgical strategy for cervical OPLL.
Fig.┬Ā1.
Surgical techniques for cervical ossification of the posterior longitudinal ligament (OPLL). Pre- and postsurgical images are shown in the left- and right-hand columns, respectively. (A, B) Anterior resection of OPLL by anterolateral oblique corpectomy. (C, D) Posterior decompression and fusion. (E, F) Anterior and posterior segmental decompression and fusion.
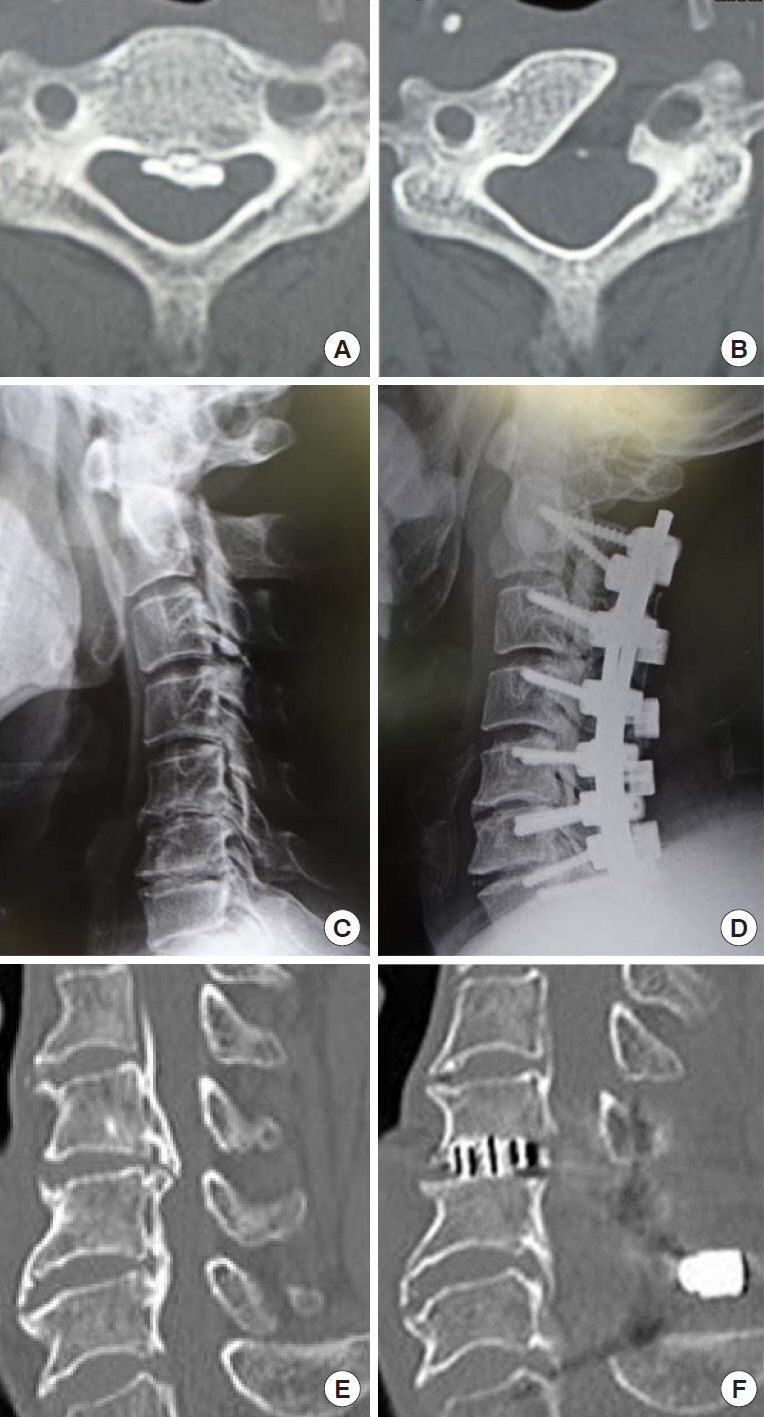
Fig.┬Ā2.
Schematic drawing of the cervical radiological parameters. The K-line is a straight line connecting the midpoints of the spinal canal at C2 and C7 on a neutral cervical lateral radiograph. The maximum occupying ratio of ossification of the posterior longitudinal ligament (OPLL) is calculated from a sagittal computed tomography image of the cervical spine.
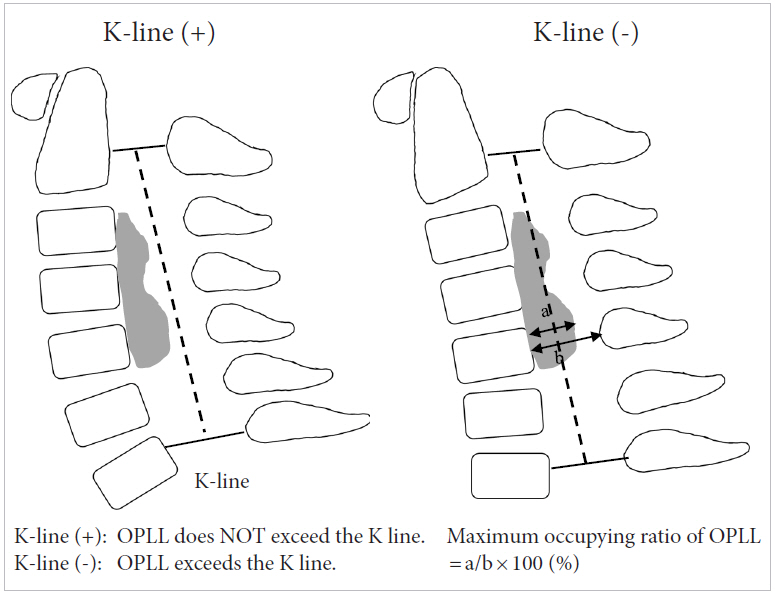
Fig.┬Ā3.
Images showing each of type of radiological classification for cervical ossification of the posterior longitudinal ligament in the system proposed by the Investigation Committee for Ossification of the Spinal Ligaments (part of the Japanese Ministry of Health, Labor and Welfare). (A) Segmental type. (B) Continuous type. (C) Mixed type. (D) Localized type.
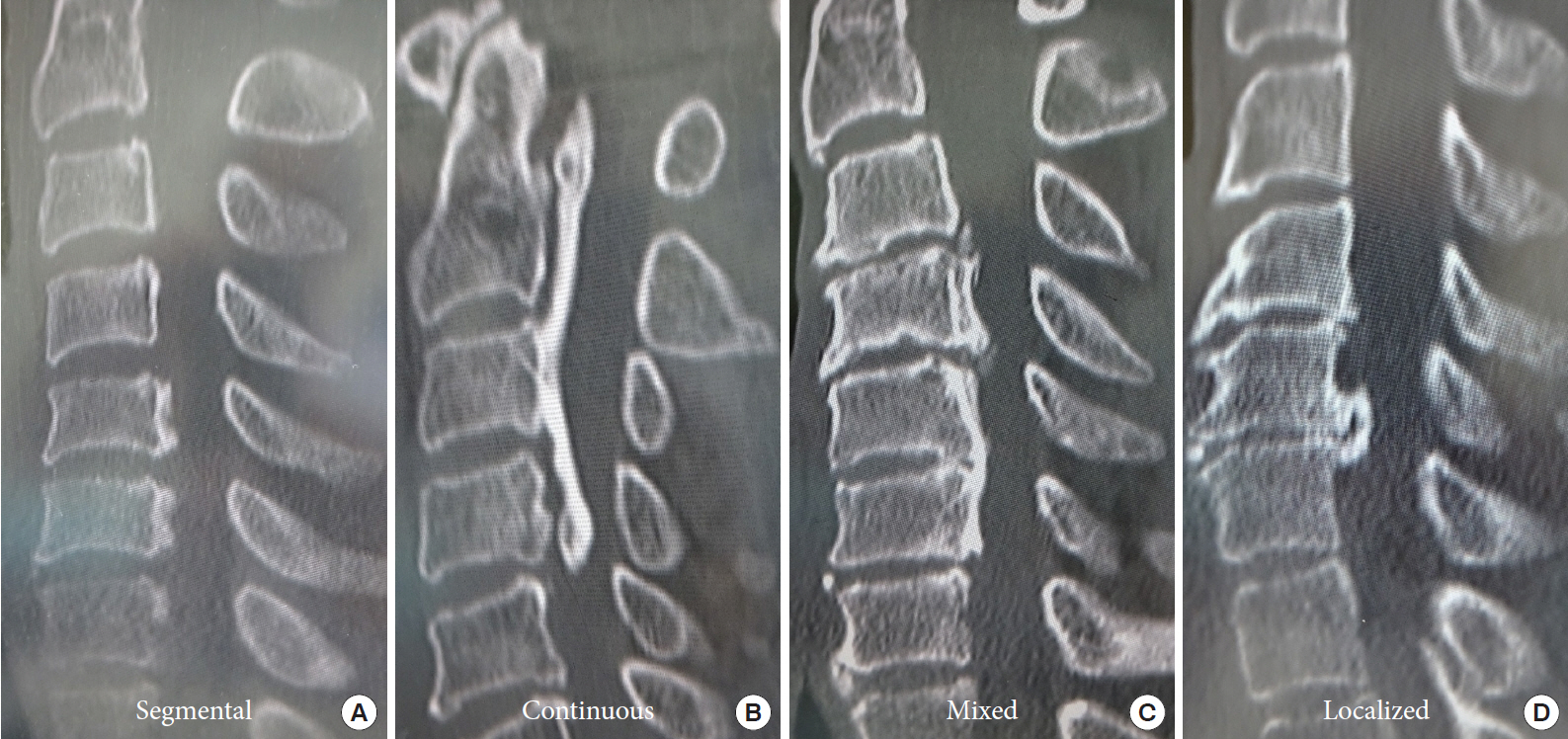
Fig.┬Ā4.
Lateral radiographs of the cervical spine showing the outline of ossification of the posterior longitudinal ligament. (A) Plateau shape. (B) Hill shape.
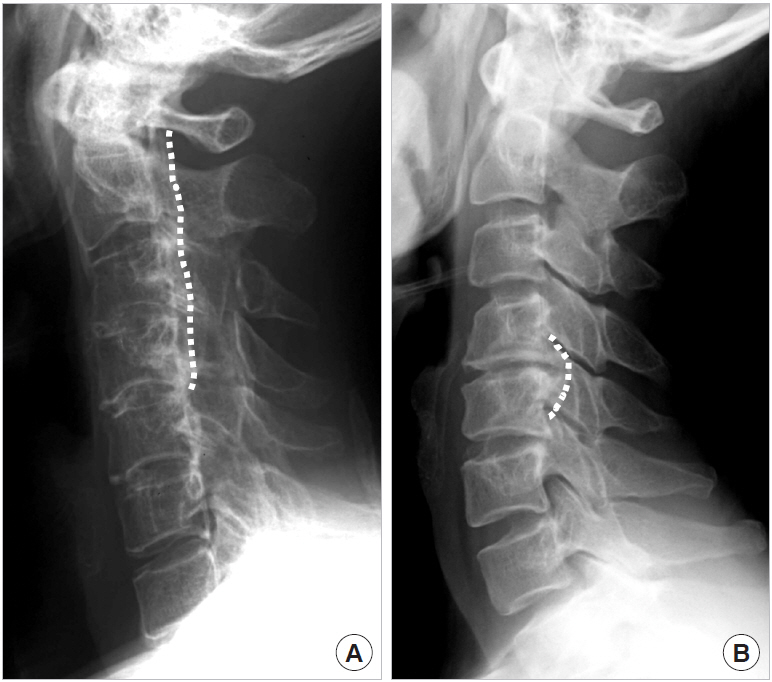
Fig.┬Ā5.
Anatomical configuration of spinal cord compression caused by cervical ossification of the posterior longitudinal ligament. (A) Boomerang type. (B) Teardrop type. (C) Triangular type.
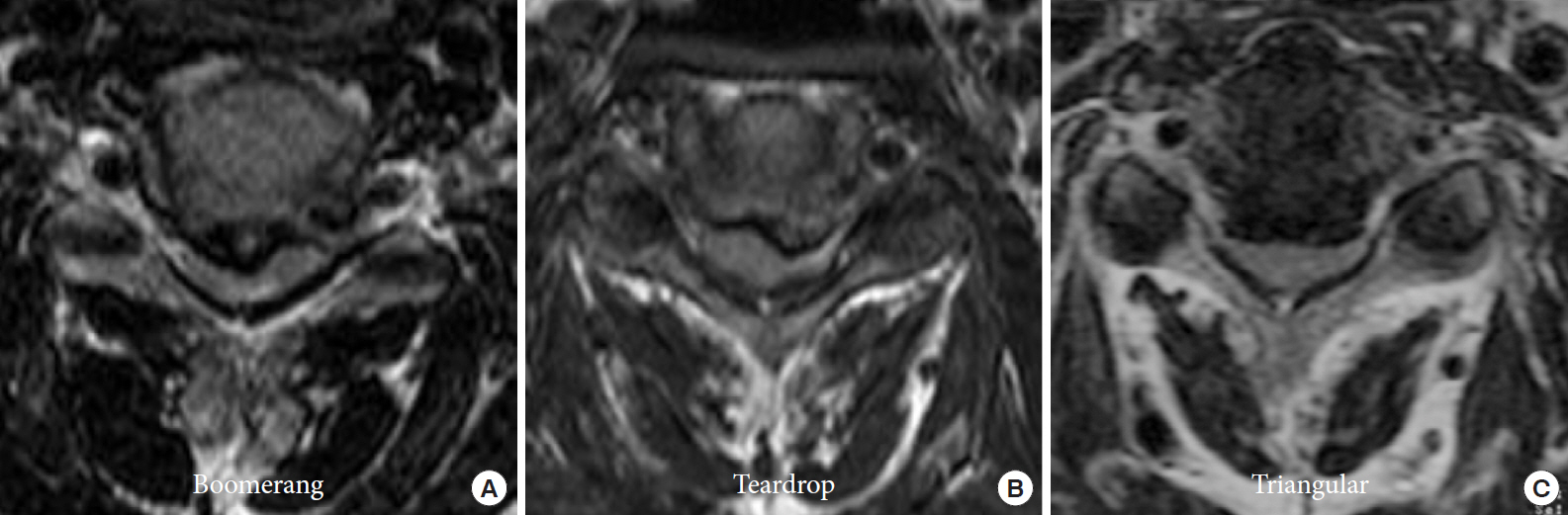
Fig.┬Ā6.
Dural ossification associated with ossification of the posterior longitudinal ligament. (A) Isolated type. (B) Double-layer type. (C) En bloc type.
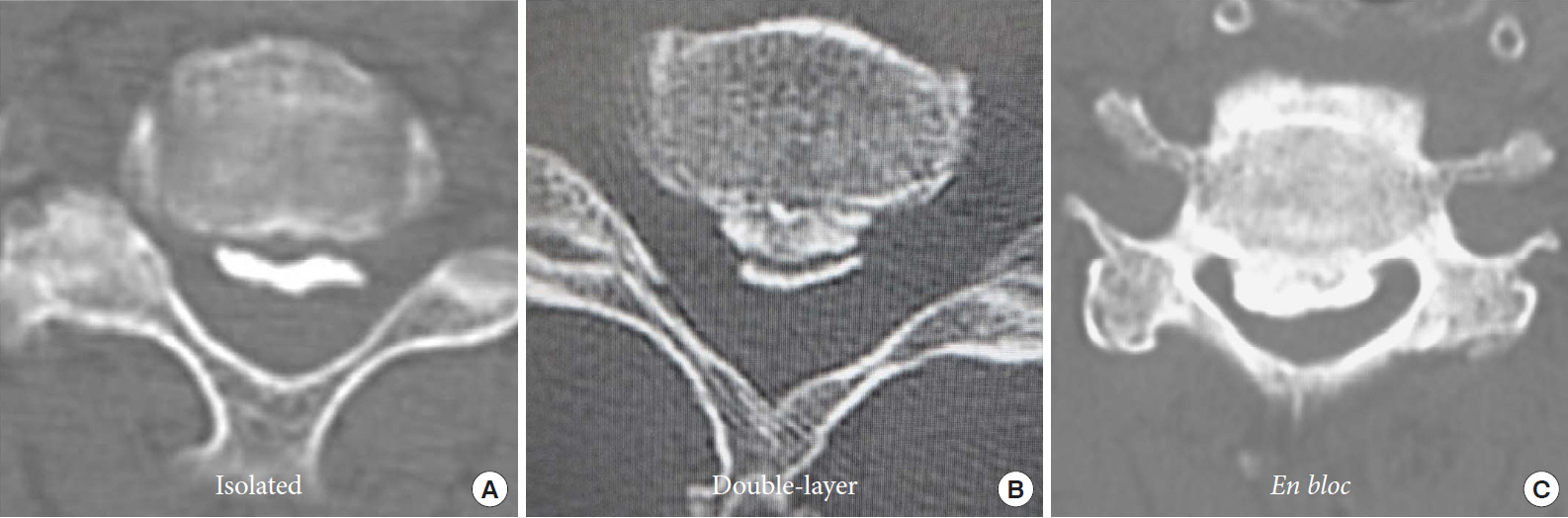
Fig.┬Ā7.
(A) Sagittal computed tomography (CT) scan. (B) Axial CT scan. C sign of ossification of the posterior longitudinal ligament (OPLL). Lateral, curved, and irregular OPLL masses showing the hook-like configuration (arrows).
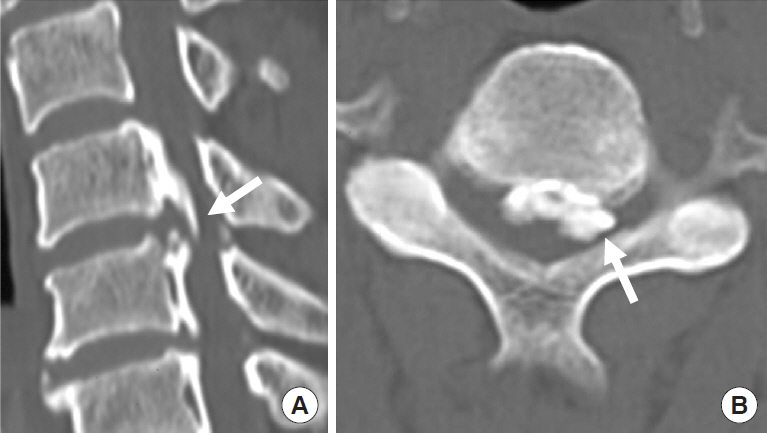
Fig.┬Ā8.
Rule of Nine. Axial computed tomography showing the Rule of Nine for evaluating surgical safety before resectioning of cervical ossification of the posterior longitudinal ligament. Please note the danger line and danger zone.
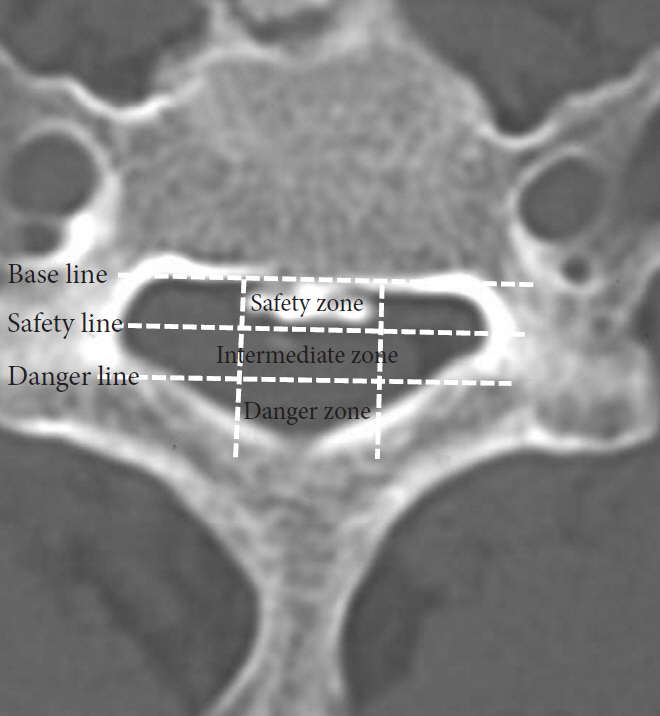
Fig.┬Ā9.
Broken sign. Sagittal (A) and axial (B) computed tomography images showing the Broken sign, visible as a crack in ossification of the posterior longitudinal ligament plaque. The arrows indicate the crack.
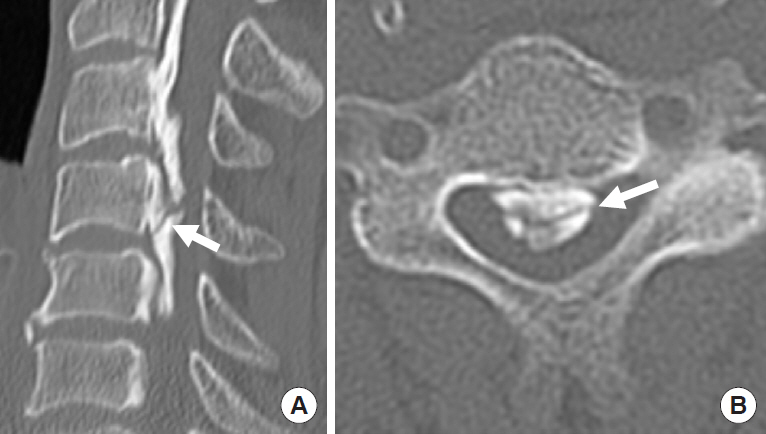
Fig.┬Ā10.
Modified K-line. T1-weighted midsagittal slice of preoperative magnetic resonance imaging (A) showing the placement of the C2ŌĆō7 line and enlarged image (B). The arrow indicates the minimum interval at the peak level of spinal cord compression.
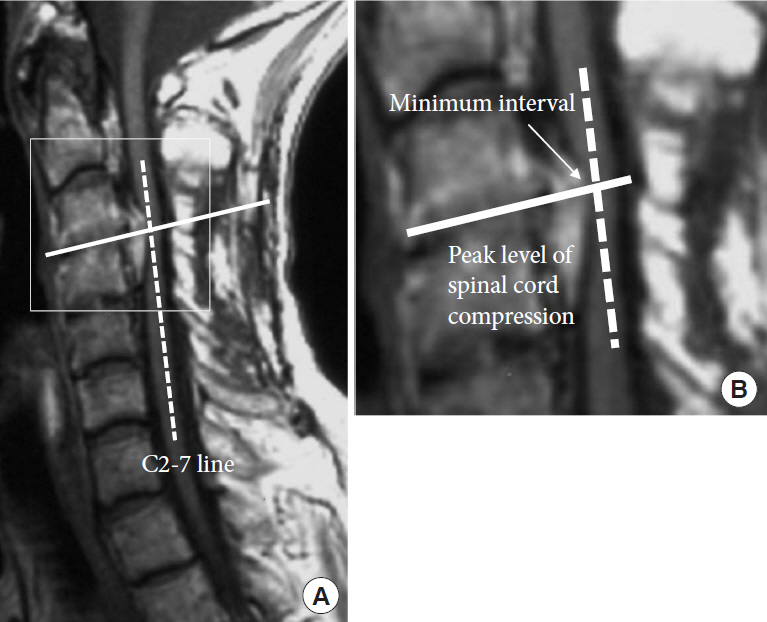
Fig.┬Ā11.
Radiological parameters of cervical sagittal balance. Cervical tilt angle, C2ŌĆō7 angle, C2ŌĆō7 sagittal vertical axis, T1 slope, neck tilt angle, and thoracic inlet angle.
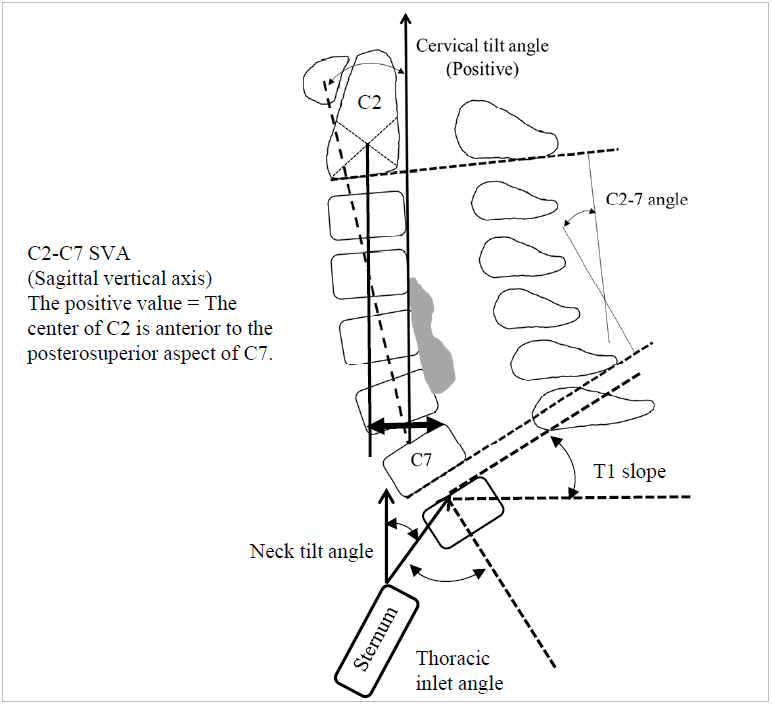
Fig.┬Ā12.
A representative case of sequential morphological change of ossification of the posterior longitudinal ligament (OPLL) after posterior fusion surgery. (A) Preoperative sagittal computed tomography (CT) demonstrates hill-shape type (beak type) OPLL at the C3/4 level (arrow). (B) Postoperative CT obtained 2 years after posterior decompression and fusion demonstrates the change from beak type to plateau type (arrow).
Table┬Ā1.
Three types of the double layer sign of dural ossification associated with cervical OPLL
OPLL, ossification of the posterior longitudinal ligament; CSF, cerebrospinal fluid.
Proposed by Yang et al. [20]
REFERENCES
1. Matsunaga S, Sakou T, Hayashi K, et al. Trauma-induced myelopathy in patients with ossification of the posterior longitudinal ligament. J Neurosurg 2002 97(2 Suppl):172-5.


2. Abe H, Tsuru M, Ito T, et al. Anterior decompression for ossification of the posterior longitudinal ligament of the cervical spine. J Neurosurg 1981 55:108-16.


3. Kojima T, Waga S, Kubo Y, et al. Anterior cervical vertebrectomy and interbody fusion for multi-level spondylosis and ossification of the posterior longitudinal ligament. Neurosurgery 1989 24:864-72.


4. Iwasaki M, Kawaguchi Y, Kimura T, et al. Long-term results of expansive laminoplasty for ossification of the posterior longitudinal ligament of the cervical spine: more than 10 years follow up. J Neurosurg 2002 96(2 Suppl):180-9.


5. Iwasaki M, Okuda S, Miyauchi A, et al. Surgical strategy for cervical myelopathy due to ossification of the posterior longitudinal ligament: Part 1: Clinical results and limitations of laminoplasty. Spine (Phila Pa 1976) 2007 32:647-53.


6. Tani T, Ushida T, Ishida K, et al. Relative safety of anterior microsurgical decompression versus laminoplasty for cervical myelopathy with a massive ossified posterior longitudinal ligament. Spine (Phila Pa 1976) 2002 27:2491-8.


7. Chiba K, Yamamoto I, Hirabayashi H, et al. Multicenter study investigating the postoperative progression of ossification of the posterior longitudinal ligament in the cervical spine: a new computer-assisted measurement. J Neurosurg Spine 2005 3:17-23.


8. Hirabayashi K, Miyakawa J, Satomi K, et al. Operative results and postoperative progression of ossification among patients with ossification of cervical posterior longitudinal ligament. Spine (Phila Pa 1976) 1981 6:354-64.


9. Lee SH, Kim KT, Lee JH, et al. 540┬░ Cervical realignment procedure for extensive cervical OPLL with kyphotic deformity. Spine (Phila Pa 1976) 2016 41:1876-83.


10. Arima H, Naito K, Yamagata T, et al. Anterior and posterior segmental decompression and fusion for severely localized ossification of the posterior longitudinal ligament of the cervical spine: technical note. Neurol Med Chir (Tokyo) 2019 59:238-45.



11. Tsuyama N. Ossification of the posterior longitudinal ligament of the spine. Clin Orthop Relat Res 1984 (184):71-84.

12. Tetreault L, Nakashima H, Kato S, et al. A systematic review of classification systems for cervical ossification of the posterior longitudinal ligament. Global Spine J 2019 9:85-103.


13. Fujiyoshi T, Yamazaki M, Kawabe J, et al. A new concept for making decisions regarding the surgical approach for cervical ossification of the posterior longitudinal ligament: the K-line. Spine (Phila Pa 1976) 2008 33:E990-3.


14. Nagashima H, Nanjo Y, Tanida A, et al. Influence of spinous process spacers on surgical outcome of laminoplasty for OPLL. Orthopedics 2013 36:e494-500.


15. Ito K, Yukawa Y, Ito K, et al. Dynamic changes in the spinal cord cross-sectional area in patients with myelopathy due to cervical ossification of posterior longitudinal ligament. Spine J 2015 15:461-6.


16. Matsuyama Y, Kawakami N, Yanase M, et al. Cervical myelopathy due to OPLL: clinical evaluation by MRI and intraoperative spinal sonography. J Spinal Disord Tech 2004 17:401-4.


17. Epstein NE, Hollingsworth R. Anterior cervical micro-dural repair of cerebrospinal fluid fistula after surgery for ossification of the posterior longitudinal ligament. Technical note. Surg Neurol 1999 52:511-4.


18. Hida K, Iwasaki Y, Koyanagi I, et al. Bone window computed tomography for detection of dural defect associated with cervical ossified posterior longitudinal ligament. Neurol Med Chir (Tokyo) 1997 37:173-5.


19. Mizuno J, Nakagawa H, Matsuo N, et al. Dural ossification associated with cervical ossification of the posterior longitudinal ligament: frequency of dural ossification and comparison of neuroimaging modalities in ability to identify the disease. J Neurosurg Spine 2005 2:425-30.


20. Yang H, Yang L, Chen D, et al. Implications of different patterns of ŌĆ£double-layer signŌĆØ in cervical ossification of the posterior longitudinal ligament. Eur Spine J 2015 24:1631-9.



21. Epstein NE. Identification of ossification of the posterior longitudinal ligament extending through the dura on preoperative computed tomographic examinations of the cervical spine. Spine (Phila Pa 1976) 2001 26:182-6.


22. Yang H, Lu X, Wang X, et al. A new method to determine whether ossified posterior longitudinal ligament can be resected completely and safely: spinal canal ŌĆ£Rule of NineŌĆØ on axial computed tomography. Eur Spine J 2015 24:1673-80.



23. Liu H, Li Y, Chen Y, et al. Cervical curvature, spinal cord MRIT2 signal, and occupying ratio impact surgical approach selection in patients with ossification of the posterior longitudinal ligament. Eur Spine J 2013 22:1480-8.




24. Sumi M, Osamu K, Satoru S, et al. Radiological analysis of cervical spondylotic myelopathy. Rinsho Seikei Geka 1998 33:1277. -86. (Japanese).
25. Ito K, Yukawa Y, Machino M, et al. Spinal cord cross-sectional area during flexion and extension in the patients with cervical ossification of posterior longitudinal ligament. Eur Spine J 2013 22:2564-8.




26. Chen J, Song D, Wang X, et al. Is ossification of posterior longitudinal ligament an enthesopathy? Int Orthop 2011 35:1511-6.



27. Kawaguchi Y, Seki S, Hori T, et al. Characteristics of ossified lesions in the upper cervical spine associated with ossification of the posterior longitudinal ligament in the lower cervical spine. J Bone Joint Surg Am 2008 90:748-53.


28. Hirai T, Kawabata S, Enomoto M, et al. Presence of anterior compression of the spinal cord after laminoplasty inhibits upper extremity motor recovery in patients with cervical spondylotic myelopathy. Spine (Phila Pa 1976) 2012 37:377-84.


29. Hirai T, Okawa A, Arai Y, et al. Middle-term results of a prospective comparative study of anterior decompression with fusion and posterior decompression with laminoplasty for the treatment of cervical spondylotic myelopathy. Spine (Phila Pa 1976) 2011 36:1940-7.


30. Subramaniam V, Chamberlain RH, Theodore N, et al. Biomechanical effects of laminoplasty versus laminectomy: stenosis and stability. Spine (Phila Pa 1976) 2009 34:E573-8.


31. Taniyama T, Hirai T, Yamada T, et al. Modified K-line in magnetic resonance imaging predicts insufficient decompression of cervical laminoplasty. Spine (Phila Pa 1976) 2013 38:496-501.


32. Taniyama T, Hirai T, Yoshii T, et al. Modified K-line in magnetic resonance imaging predicts clinical outcome in patients with nonlordotic alignment after laminoplasty for cervical spondylotic myelopathy. Spine (Phila Pa 1976) 2014 39:E1261-8.


33. Vialle R, Levassor N, Rillardon L, et al. Radiographic analysis of the sagittal alignment and balance of the spine in asymptomatic subjects. J Bone Joint Surg Am 2005 87:260-7.


34. Smith JS, Singh M, Klineberg E, et al. Surgical treatment of pathological loss of lumbar lordosis (flatback) in patients with normal sagittal vertical axis achieves similar clinical improvement as surgical treatment of elevated sagittal vertical axis: clinical article. J Neurosurg Spine 2014 21:160-70.


35. Van Royen BJ, Toussaint HM, Kingma I, et al. Accuracy of the sagittal vertical axis in a standing lateral radiograph as a measurement of balance in spinal deformities. Eur Spine J 1998 7:408-12.




36. Hyun SJ, Kim KJ, Jahng TA, et al. Relationship between T1 slope and cervical alignment following multilevel posterior cervical fusion surgery: impact of T1 slope minus cervical lordosis. Spine (Phila Pa 1976) 2016 41:E396-402.


37. Glassman SD, Berven S, Bridwell K, et al. Correlation of radiographic parameters and clinical symptoms in adult scoliosis. Spine (Phila Pa 1976) 2005 30:682-8.


38. Glassman SD, Bridwell K, Dimar JR, et al. The impact of positive sagittal balance in adult spinal deformity. Spine (Phila Pa 1976) 2005 30:2024-9.


39. Kim YJ, Bridwell KH, Lenke LG, et al. Results of lumbar pedicle subtraction osteotomies for fixed sagittal imbalance: a minimum 5-year follow-up study. Spine (Phila Pa 1976) 2007 32:2189-97.


40. Lafage V, Ames C, Schwab F, et al. Changes in thoracic kyphosis negatively impact sagittal alignment after lumbar pedicle subtraction osteotomy: a comprehensive radiographic analysis. Spine (Phila Pa 1976) 2012 37:E180-7.


41. Lowe T, Berven SH, Schwab FJ, et al. The SRS classification for adult spinal deformity: building on the King/Moe and Lenke classification systems. Spine (Phila Pa 1976) 2006 31(19 Suppl):S119-25.


42. Smith JS, Bess S, Shaffrey CI, et al. Dynamic changes of the pelvis and spine are key to predicting postoperative sagittal alignment after pedicle subtraction osteotomy: a critical analysis of preoperative planning techniques. Spine (Phila Pa 1976) 2012 37:845-53.


43. Smith JS, Klineberg E, Schwab F, et al. Change in classification grade by the SRS-Schwab Adult Spinal Deformity Classification predicts impact on health-related quality of life measures: prospective analysis of operative and nonoperative treatment. Spine (Phila Pa 1976) 2013 38:1663-71.


44. Terran J, Schwab F, Shaffrey CI, et al. The SRS-Schwab adult spinal deformity classification: assessment and clinical correlations based on a prospective operative and nonoperative cohort. Neurosurgery 2013 73:559-68.



45. Oshima Y, Takeshita K, Taniguchi Y, et al. Effect of preoperative sagittal balance on cervical laminoplasty outcomes. Spine (Phila Pa 1976) 2016 41:E1265-70.


46. Tang JA, Scheer JK, Smith JS, et al. The impact of standing regional cervical sagittal alignment on outcomes in posterior cervical fusion surgery. Neurosurgery 2012 71:662-9.



47. Weng C, Wang J, Tuchman A, et al. Influence of T1 slope on the cervical sagittal balance in degenerative cervical spine: an analysis using kinematic MRI. Spine (Phila Pa 1976) 2016 41:185-90.


48. Lee SH, Kim KT, Seo EM, et al. The influence of thoracic inlet alignment on the craniocervical sagittal balance in asymptomatic adults. J Spinal Disord Tech 2012 25:E41-7.


49. Lee SH, Son ES, Seo EM, et al. Factors determining cervical spine sagittal balance in asymptomatic adults: correlation with spinopelvic balance and thoracic inlet alignment. Spine J 2015 15:705-12.


50. Knott PT, Mardjetko SM, Techy F. The use of the T1 sagittal angle in predicting overall sagittal balance of the spine. Spine J 2010 10:994-8.


51. Dalbayrak S, Yaman O, Firidin MN, et al. The contribution of cervical dynamic magnetic resonance imaging to the surgical treatment of cervical spondylotic myelopathy. Turk Neurosurg 2015 25:36-42.

52. Gotkine M, Abraham A, Drory VE, et al. Dynamic MRI testing of the cervical spine has prognostic significance in patients with progressive upper-limb distal weakness and atrophy. J Neurol Sci 2014 345:168-71.


53. Paholpak P, Tamai K, Shoell K, et al. Can multi-positional magnetic resonance imaging be used to evaluate angular parameters in cervical spine? A comparison of multi-positional MRI to dynamic plain radiograph. Eur Spine J 2018 27:1021-7.



54. Kawaguchi Y, Kanamori M, Ishihara H, et al. Progression of ossification of the posterior longitudinal ligament following en bloc cervical laminoplasty. J Bone Joint Surg Am 2001 83:1798-802.


55. Fargen KM, Cox JB, Hoh DJ. Does ossification of the posterior longitudinal ligament progress after laminoplasty? Radiographic and clinical evidence of ossification of the posterior longitudinal ligament lesion growth and the risk factors for late neurologic deterioration. J Neurosurg Spine 2012 17:512-24.


56. Fujimori T, Iwasaki M, Nagamoto Y, et al. Three-dimensional measurement of growth of ossification of the posterior longitudinal ligament. J Neurosurg Spine 2012 16:289-95.


57. Fujiyoshi T, Yamazaki M, Okawa A, et al. Static versus dynamic factors for the development of myelopathy in patients with cervical ossification of the posterior longitudinal ligament. J Clin Neurosci 2010 17:320-4.






























