Biomaterials in Spinal Implants: A Review
Article information
Abstract
The aim to find the perfect biomaterial for spinal implant has been the focus of spinal research since the 1800s. Spinal surgery and the devices used therein have undergone a constant evolution in order to meet the needs of surgeons who have continued to further understand the biomechanical principles of spinal stability and have improved as new technologies and materials are available for production use. The perfect biomaterial would be one that is biologically inert/compatible, has a Young’s modulus similar to that of the bone where it is implanted, high tensile strength, stiffness, fatigue strength, and low artifacts on imaging. Today, the materials that have been most commonly used include stainless steel, titanium, cobalt chrome, nitinol (a nickel titanium alloy), tantalum, and polyetheretherketone in rods, screws, cages, and plates. Current advancements such as 3-dimensional printing, the ProDisc-L and ProDisc-C, the ApiFix, and the Mobi-C which all aim to improve range of motion, reduce pain, and improve patient satisfaction. Spine surgeons should remain vigilant regarding the current literature and technological advancements in spinal materials and procedures. The progression of spinal implant materials for cages, rods, screws, and plates with advantages and disadvantages for each material will be discussed.
INTRODUCTION
Spinal surgery has existed for since Jules Gerin first attempted surgical scoliosis correction in 1839 [1]. In the late 1800’s Dr. Berthold Earnest Hadra created a fixation device for C6–7 dislocations using silver wire that he later applied to the treatment of Potts disease [2]. His methods gained the attention of orthopedists across Europe who modified it to stabilize the spinous processes, laminae, and pedicles for the cervical spine using wiring, lateral mass screws, plates, and rods, and pedicle screws [3-5]. Since its inception, spinal surgery and the devices used therein have undergone a constant evolution in order to (1) meet the needs of surgeons who have continued to further understand the biomechanical principles of spinal stability and (2) have improved as new technologies and materials are available for production use. Spinal implants need to demonstrate biostability (ability to resist the effects of micro-oganisms) and biocompatibility (not harmful to living tissue) and the difficulty in finding an optimal material lies in determining a material with an appropriate Young’s modulus (also known as Elastic Modulus, the stress to strain ratio which demonstrates increased stiffness and brittleness with higher values), stiffness, and fatigue [6]. The materials that have been most commonly used include stainless steel (SS), titanium, cobalt chrome, nitinol (a nickel titanium alloy), tantalum, and polyetheretherketone (PEEK). The perfect biomaterial would be one that is biologically inert/compatible, has a Young’s modulus similar to that of the bone where it is implanted, high tensile strength, stiffness, fatigue strength, and low artifacts on imaging. This review article will discuss the uses of these various biomaterials and their application in spinal implants. The progression of spinal implant materials for cages, rods, screws, and plates will be discussed with advantages and disadvantages for each material as have been demonstrated in multiple clinical studies.
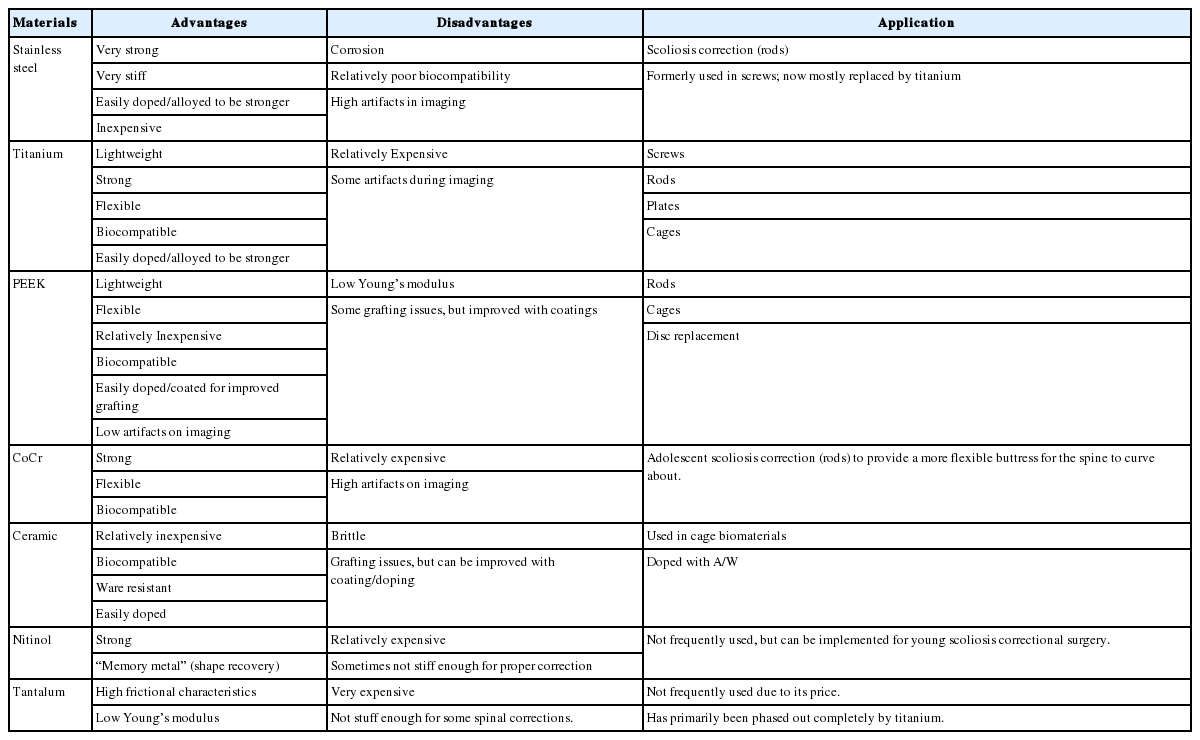
BIOMATERIALS IN VARIOUS TYPES OF SPINAL IMPLANTS
1. Cages
Cages are spinal implants that act as a stabilizer for force distribution between vertebral bodies and to restore the height of the intervertebral and foramina space [7]. They provide a graft for vertebrae to refuse and heal when an intervertebral disc has failed [7]. Originally, autologous bone grafts were used; however, donor site morbidity combined with high failure rates resulting from collapse, subsidence, retropulsion, or resorption of the graft with prolonged healing time was frequently seen [8].
Cages are typically made of metal – ranging from pure titanium (PTi), titanium composite/alloy, ceramic – usually silicon nitride, or plastic – usually PEEK or another bioinert plastic such as acrylic by itself or coated in another material (such as hydroxyapatite [HA] or titanium) [9-14]. These cages are porous as to allow the bones to grow through them and stabilize [15].
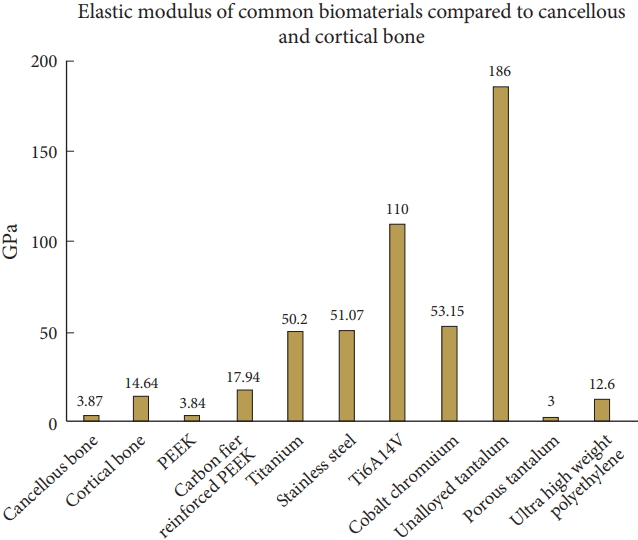
The most popular materials used today are titanium (titanium-aluminum-vanadium, Ti6Al4V) and PEEK (Tables 1, 2) [16]. PTi and titanium alloys are the preferred metal in orthopaedic implants due to their high fracture resistance and biocompatibility [17]. Titanium has a superior biocompatibility, corrosion resistance, and Young’s modulus compared with SS and cobalt counterparts (Fig. 1) [18]. The major issue with polished or abraded titanium is its lack of bone-bonding ability, hence increased research has been done to coat/dope titanium alloys to improve bone-bonding.
PEEK has a similar Young’s modulus compared to bone, without sufficient doping such as titanium plasma spray and vapor deposition, has weak surface interfaces that can fracture upon cage implantation [19]. PEEK has radiographic properties that allow surgeons to monitor possible migration and success of the implant. However, the primary issue with PEEK is that it is hydrophobic and unable to bond to bone to achieve a solid fusion [20]. Research has shown that there may be associations with cage migration and pseudarthrosis [21]. It has also been shown that PEEK has a significantly lower stress compression strength compared to titanium (2.5 times weaker) [22]. However, for anterior cervical discectomy and fusion (ACDF) surgeries, it has been shown that PEEK cages have a significantly lower loss of Cobb angles and lower cage subsidence rate compared to titanium (34.5% and 5.4% respectively) [23].
Increased research for advancements in disc separation has been made to find more biocompatible materials than titanium and PEEK to improve bone grafting. Experimentation with silicon nitride (Si3N4) showed no significant different compared to PEEK [24]. Other studies have tried an Apatite-Wollastonite (A/W) ceramic cage to replace the PEEK cages. However, like the silicon nitride cage materials, there was no significant advantage of the A/W cage compared to the porous PEEK cages [25]. An additional study performed stress/strain tests on porous biodegradable materials such as poly(ε-caprolactone) combined with HA. Computational and in vitro modeling confirmed the mechanical integrity of the new biodegradable material within the human spine [26]. Many of these studies have demonstrated that there are many theoretical models that seem to have promising results; however, no clinical data have been published in human trails to date for these materials.
2. Rods
Spinal rods are used in conjunction with other spinal implants to add stability to spinal implant structure. Rods are contoured to a specific patient to fit the spine. In 1962, Dr. Harrington introduced the “Harrington Rod” – an SS rod – for surgical treatment of scoliosis [27]. However, rod contouring (such as French Benders) imparts marks and weaknesses onto the rod that compromise its durability [28].
Rods were initially made of SS to provide sufficient stability and stiffness for proper spine alignment [29]. Today, rods are typically made of titanium or PEEK. The shift from SS to titanium resulted from titanium’s improved biomechanical properties to bone (similar modulus elasticity of cortical bone), improved biocompatibility, and improvements in stress shielding of lumbar pedicle instrumentation [30,31].
The metal rods used today fall into one of 3 alloy families: iron (Fe)-chromium (Cr)-nickel (Ni) alloys and austenitic SS, titanium and its alloys (PTi and Ti6Al4V alloy), and cobalt-chromium alloys (CoCr). These alloys were chosen because they are relatively biocompatible and are protected from corrosion by the presence of a stable oxide layer; however, SS is the least corrosion resistant [32]. Of these metals, titanium has gained popularity due to its biomechanical properties as well as its significantly few artifacts on imaging [33].
These other metal rods are stiffer than titanium rods. CoCr rods have gained recent popularity with adolescent idiopathic scoliosis with promising frontal correction rate in all-screw constructs due to their increased rigidity compared to titanium, closer reflecting SS [34]. CoCr provided fewer complications in human trials and allowed for better thoracic sagittal realignment compared to titanium (p= 0.01) [35]. The primary drawback of CoCr compared to titanium is cost and increased artifacts on MRI; however, a study has shown that the spinal canal and neural element analysis was not impinged by the CoCr artifacts [36].
Nitinol (50% Ni and 50% Ti) rods—also called memory rods —are not as common as Ti since the metal is expensive and notch-sensitive [37]. Nitinol is unique compared to SS or Ti because it can be placed in the spinal canal with no inflammatory response from the lymph nodes or other organs [38]. Nitinol shape recovery force have been used to achieve scoliosis correction. However, Nitinol is not frequently used due to its higher costs and lower Young’s modulus compared to Ti or SS rods [39].
Besides the rigid metal rods, there are semirigid rods. One of the most popular is the PEEK rods. It has been shown that PEEK provides comparable stability compared to titanium rods of similar size [40]. Some advantages of PEEK compared to titanium are load sharing; PEEK rods allow for greater contact between the end plate and bone graft due to its improved biological compliance and elasticity. PEEK is a radiolucent material, providing an advantage over other metallic devices since it results in only minor artifact on postoperative computed tomography and magnetic resonance imaging; however, it is more difficult for clinicians to identify faults and breaks within the PEEK rods, thus some PEEK rods use a contrast agent to improve imaging [41]. Some possible disadvantages to PEEK rods range from higher failure rates with early reoperations and insufficient capabilities of PEEK to form a stable union with the screw interface, leading to increased rates of pseudarthrosis [42].
Many biomechanical studies have shown that when rods are bent to fit a patient using a French bender, significant surface defects and weakness are introduced [43]. Rod fatigue, fractures, and significant deformation almost always occur at these notch points and at the screw-rod junction [44]. One study showed that notching and bending significantly reduces the mechanical stability of titanium alloy rods, but not SS rods (p< 0.05) [28]. Clinical studies have found that waved rods have significantly improved fusion rates compared to rigid pedicle screws (45/50 compared to 36/46, p= 0.039) [45].
New research is being conducted to create more efficient and more failure-resistant materials. Beta-type titanium-molybdenum and oxygen-modified beta-type titanium-chronium alloys have demonstrated promising Young’s Moduli, high bending strength, and high tensile strength [44,46]. A study has shown that polycarbonate-urethane rods demonstrated more comparable intradiscal pressure compared to titanium rods consistently through 3,000 loading cycles (p< 0.05) [47]. Another study tested the feasibility of biodegradable rods for posterior lumbar fusions in vitro which demonstrated that the biodegradable rods—like the standard titanium rods—could withstand 5,000,000 dynamic compression cycles under a 145 N axial load. However, the biodegradable rods had a 20% and 80% decrease in Young’s modulus after 6 months and 12 months [48]. Furthermore, both of these studies were done not done in human trials or under true biological conditions, thus not all the effects of wear and tear could be measured and tested.
3. Pedicle Screws
Pedicle screws are frequently used in spinal surgeries. Pedicle screw and rod posterior spinal fusion has become the clinical standard for the treatment of scoliosis since the pedicle screws can redirect force through the powerful vertebral bodies [49]. To withstand such great force, pedicle screws need to be made of a material that is strong, but also bioinert, thus, many today are made of Ti6Al4V [50].
While pedicle screws provide some of the best correctional results in spinal surgery, some of the most common complications of pedicle screw placement include loosening, pullout, and screw breakage that can severely affect the bone healing process [51]. Various studies have tried coating and doping the pedicle screws with different materials ranging from HA, calcium phosphate (CaP), polymethymethacrylate bone cement (PMMA-BC), extracellular matrix (ECM), tantalum, and titanium plasma spray. It was found that coating the Ti6Al4V screws with HA can improve resistance against pullout force compared to uncoated (165.6± 26.5N vs. 103.1± 30.2 N, p< 0.001) [52] and had an improved bone-to-implant contact (64%± 31% vs. 9%± 13%) [53] respectively
A study that compared HA, CaP, and PMMA-BC showed that there was no significant difference between the controls versus HA or CaP (pHA = 0.6 and pCaP= 0.234); however, the pullout strength differences between PMMA-BC and control were significantly different (p= 0.026) [54]. Although PMMC-BC has a significantly greater pullout strength compared to the control, it is a far more rigid and solidified cement structure and cannot be modified easily postoperationally compared to HA or CaP. Another study coated the Ti6Al4V pedicle screws with tantalum and analyzed numerous effects a thin tantalum coating had on the performance of the Ti6Al4V screws. The tantalum screws had improved bone osteoclast proliferation in vitro, strong integration capacity of trabecular bone surrounding the screws, and greater pullout strength 12 weeks after fixation compared to the conventional Ti6Al4V screws in vivo. [55] Coating the pedicle screws with ECM also seemed to improve pullout strength when combined with HA compared to the uncoated screws. There was no significant difference in the pullout strength between the screws coated in just ECM vs screws just coated in HA, but there was a significant difference in pullout strength between screws coated in HA+ECM vs. HA or ECM alone [56].
New developments in pedicle screws have not focused much on the Ti6Al4V screw itself, but rather novel coatings and/or new cement on the Ti6Al4V screw to improve fixation and pullout strength. Lotz et al. [57] performed cadaveric lumbar tests with uncoated pedicle screws and pedicle screws in conjunction with the carbonated apatite cancellous bone cement demonstrated an average 68% (p< 0.001) greater pullout strength compared to the control screws.
In addition to pedicle screw biomaterial properties, technological advancements in screw design have recently occurred. The first pedicle screws were monoaxial and although provided good vertebral stabilization and correction, had difficulty adequately seating the rod in the screw [58]. This deficiency could result in movement of the screw within the vertebral body—causing loosening of the screw—and/or inadequate closure of the screw-rod connection [59]. Polyaxial pedicle screws used in unilateral or bilateral fixation are made out of titanium [60]. Polyaxial screws have been subsequently implemented and designed to provide increased freedom on the screw-to-rod connection to facilitate easier rod seating into the screw head saddle. It has been shown that adjacent segment degeneration is less likely to be observed in polyaxial pedicle screw fixation than in monoaxial fixation due to lower von Mises stress in the polyaxial pedicle screws compared to the monoaxial pedicle screws [61]. A compromise between monoaxial and polyaxial screws was the uniplanar or uniaxial screw construct, designed to accommodate sagittal angle variation of the pedicle screws with respect to rods. Uniplanar screws showed less residual apical vertebral rotation after a 1-year postoperative evaluation (p< 0.001) [62].
Problems associated with pedicle screws include loss of fixation, improper placement, fatigue and bending failure, dural tears, cerebral spinal fluid leaks, nerve root injury, and infection [63-65]. Bending or breakage is the most common type of pedicle screw failure [66]. Screw diameter, screw length, insertional depth, orientation, and cross-link conduits all affect bending and breakage [67]. Biomechanical studies show that osteoporosis, cortical fixation, pedicle morphology, screw orientation, bone density, screw thread area, and screw orientation all affect pedicle screw pullout [68,69].
4. Plates
Spinal plates are a pinnacle implant to stabilize and restore normal alignment to the spine. Biomechanical experiments for stress-strain analyses performed from T12–L4 and suggested that a titanium-plate fixation and laminectomy was more stable than just laminectomy or just a plate (p< 0.05) [70]. Many plates are traditionally made of PTi or Ti6Al4V [71]. Some plates have been developed to incorporate additional elements such as polyethylene ring utilized as a screw locking mechanism within the plate [72]. New developments in spinal plate technology include development of biodegradable plates that have been developed and tested in vitro compared to the standard titanium plate. The strong biodegradable plate and bone block (PLA-4G) have similar range of motion, torque strength, and von Mises stress results as titanium, and has a lower Young’s modulus compared to titanium (4,000 N/mm2 vs. 10,000 N/mm2) [73]. The biodegradable plate is not paramagnetic, reducing artifacts on future imaging studies.
Mini-plate systems are extensively applied to cervical laminoplasty for patients with multilevel cervical compressive myelopathy and to secure the posterior elements in the open position after an expansive open-door laminoplasty [74]. These miniplates are typically made of titanium, but research into developing a cheaper model for mini-plates is ongoing [75].
NEW DEVELOPMENTS
1. Lumbar Spine Disc Replacement
Total disc replacement (TDR) has yielded comparable or superior outcomes compared to lumbar fusion over a 2-year time period, preserving functional movement unlike with spinal fusion [76]. TDR had a relatively low rate of complications after a 5-year meta-analysis for lumbar TDR (0%–16.7%) and cervical (0%–4.0%) [77]. The first lumbar disc replacement was a steel ball implanted in 1960, which ultimately led to multiple postoperative complications [78]. In the 1980s, the implants shifted from a SS ball to 2 plates made of steel or titanium with a polyethylene sliding core in between them [79]. There was an additional implant called the SB Chartie prothesis which consisted of 2 chromium-cobalt plates and a mobile polyethylene core [80]. In 1989, the ProDisk-L was developed which had plates with a central titanium stem [81]. Ceramics were considered since they are more resistant to wear, but are significantly more fragile due to their low ductility. Surface coatings and anchorage to promote osseointegration ranged from HA, tricalcium phosphate, porous titanium or chromium-cobalt.
The ProDisc-L implants are made of a cobalt-chromium-molybdenum with an ultra-high molecular weight polyethylene combined with a rough titanium surface coating to promote bone growth alloy [82]. ProDisc-L showed significant improvement in fusion and Oswestry Disability Index (p< 0.001) compared to circumferential arthrodesis [82].
The ApiFix system is a new, less-invasive fusionless scoliosis correction system that connects 2 periapical pedicle screws through polyaxial mobile ball-and-socket joints with a rod [83]. It was demonstrated that this implant halved the range of motion of flexion-extension (-40% in humans), Lateral bending was partially affected (-18.2% in humans), axial rotation was not affected.
2. Cervical Spine Disc Replacement
The ProDisc-C implants are made of a cobalt-chromium-molybdenum with an ultra-high molecular weight polyethylene combined with a rough titanium surface coating to promote bone growth alloy [82]. ProDisc-C has been found to restore range of motion of the entire neck back to preoperative state in late phase postoperative surgery. It was also suggested that segmental degenerative kyphosisc was significantly corrected in those who underwent ProDisc-C replacement [84].
Another recent development is the Mobi-C cervical disc prothesis. The Mobi-C disc has 3 parts: 2 metal plates (typically made out of cobalt, chromium and molybdenum) covered with a HA coating (to improve bone grafting) and a plastic plate (made from polyethylene) at the center [85]. Mobi-C has demonstrated equal or increased improvement in range of motion, pain and short-form scores compared to the standard ACDF [86]. It has been shown that Mobi-C artificial discs resulted in an improved neck disability index score, patient satisfaction and reduced surgical intervention compared to ACDF [85]. Additional conclusive long-term clinical data is needed.
THREE-DIMENSIONAL PRINTING, AND ADVANCES IN SPINAL IMPLANTS
Three-dimensional (3D) printing is the most frequently utilized in preoperative planning stages, printing templates out of plastic—including but not limited to acrylonitrile butadiene styrene, acrylate resin, acrylate resin, polyamide photosensitive resin, titanium, and polylactic acid [87]. Pedicle screw templates have been printed with a 94.60% acceptable rate with cervical spine surgery [88]. These models have been proven to reduce the perioperative blood loss during complicated spinal fusion surgeries as well as reduction in fluoroscopy time, improved communication with the surgical team, and lower rates of screw misplacements [89]. Models have also shown to reduce operation time by 15%–20% through a more involved pathology (location and surgical approach) as well as facilitation of preoperative instrumentation decisions [89]. These models cost an additional $300 to over $1,000 and take between 5 hours and 2 days to create [90]. However, limitations with preoperative modeling exist ranging from lack of surgically useful information such as joint instability and a sense of real-time information as provided with imaging combined with a significant learning curve for the software and hardware required to create the models [87,88,91].
Arguably the most exciting application of 3D printing is the ability of surgeons to create specifically-designed implants for each patient. So far, a majority of the implants have been made out of titanium (TiV6Al4) due to its biocompatibility for applications ranging from C1/2 posterior fixation devices to Sacrum replacements [92-94]. However, there is an increased research effort towards 3D printing biodegradable scaffolds using a degradable polyurethane to mimic the elasticity of the intervertebral disc [95]. Tissue and bone engineering is being explored as possible alternatives to biodegradable plastics for intervertebral discs and other spinal implants [96].
CONCLUSION
Spinal implants have come a long way from the original 1890s silver wire fusion methods. Now, the standard material is either PEEK or titanium. New research is being conducted to find materials with increased bone grafting properties (by doping existing materials or developing new materials), improving strength/ Young’s modulus, and developing novel ideas to prevent further postoperative complications by improving range of motion, decreasing pain, distributing anatomic forces to decrease adjacent segment disease, and minimizing the necessity for additional spinal surgery. New developments in biomaterials for spinal implants and the advent of new technologies, like 3D printed patient-specific implants, have made incredible progress in biocompatibility of spinal tools. Spine surgeons should remain vigilant regarding the current literature and technological advancements in spinal materials and procedures.
Notes
The authors have nothing to disclose.