Internet of Things, Digital Biomarker, and Artificial Intelligence in Spine: Current and Future Perspectives
Article information
Abstract
Recent interest in medical artificial intelligence (AI) has increased with onset of the fourth industrial revolution. Real-time monitoring of patients is an important research area of medical AI. The medical AI is very closely related to the Internet of Things (IoT), a core element of the fourth industrial revolution. Attempts to diagnose and treat patients using IoT have been already applied to patients with chronic disease such as hypertension and arrhythmia. However, in the spine, research on IoT and digital biomarkers are still in the early stages. The digital biomarker obtained by IoT devices is objective and could represent real-time, real-world, and abundant data. Based on its characteristics, IoT and digital biomarkers can also be useful in the spine. Currently, research on real-time monitoring of physical activity or spinal posture is ongoing. Therefore, the authors introduce the basic concepts of IoT and digital biomarkers, their relationship to AI, and recent trends. Current and future perspectives of IoT and digital biomarker in spine are also discussed. In the future, it is expected that IoT, digital biomarkers, and AI will lead to a paradigm shift in the diagnosis and treatment of spinal diseases.
INTRODUCTION
Recent interest in the fourth industrial revolution, represented by artificial intelligence (AI), big data, the Internet of Things (IoT), and robotics, is higher than ever [1,2]. In particular, studies are increasingly exploring the medical application of AI, which is the key element of the fourth industrial revolution [3-5]. Medical AI can be used in 3 different ways: First, AI is used to diagnose the disease or predict the treatment outcome with clinical data or genomic data [6-8]. Second, AI automatically reads medical images and diagnoses diseases instead of doctors, and third, AI can be used to prevent or predict disease by monitoring biomedical signals in real time [9,10].
In the field of spine, AI has been used to predict the outcome after surgery or survival of patients with spinal metastasis [6,11]. The focus of AI in medical imaging has been on segmentation of spinal structures, classification of disc degeneration, measurement of various spinal curve parameters, and diagnosis of fractures and spinal tumors [5,12]. Research into AI to predict and prevent disease by monitoring biomedical signals in real time is particularly active in internal medicine [13]. In particular, devices to monitor blood glucose or electrocardiogram in real time have been already commercialized [13,14]. Despite such high interest in real-time monitoring of patients, AI and real-time monitoring in spine, are still in early stages of research [5]. AI related to real-time monitoring of patients is closely related to IoT and digital biomarkers [15]. Here, authors introduce the basic concept of IoT, digital biomarker, and their relationships with AI. In addition, current and future perspectives of IoT and digital biomarker in spine are discussed.
CONCEPTS AND RELATIONSHIPS OF INTERNET OF THINGS, DIGITAL BIOMARKER, AND ARTIFICIAL INTELLIGENCE
The AI and machine learning have been actively studied even in the spine recently, and a few spine surgeons understand the basic concept of AI [5]. Moreover, most of spine surgeons are not familiar with IoT and digital biomarker.
IoT is the network of physical objects that contain embedded technology to communicate and sense or interact with their internal states or the external environment [16]. The “thing” in IoT can be anything in the world, not just mobile or wearable devices. Thus, IoT has recently been referred to as the Internet of Everything [17]. Previously, IoT and AI were defined separately. In recent years, IoT has evolved as a technology including AI.
IoT is generally composed of devices with embedded sensors, gateway, cloud, analytics, and user interface [18,19]. Devices with embedded sensor continuously collect information from the environment and deliver it to the next levels via wireless network. Low-power networks such as Wi-Fi, Bluetooth, and Long Range are commonly used [20]. Recently, 5G network has been regarded as an optimized technology of IoT with ultralow delay and ultraconnectivity [21]. Gateway plays a key role of receiving information from the sensors of various devices and delivering it to a cloud. Cloud provides a tool to collect, analyze, and store an abundance of data produced from devices in real time [19]. In analytics, AI derives meaningful patterns in unstructured data and provides feedback to the user interface. The user interface facilitates visualization of feedback for patients or medical staff. The concept of IoT system is very similar to that of human nervous system. Various sensory organs such as skin, eyes, ears, nose, and organs of equilibrium correspond to digital devices equipped with sensors. The spinal cord and nerves correspond to the wireless network, and the human brain corresponds to cloud and AI. Actuator or feedback service is similar to response of human body such as muscle action to sensory information (Fig. 1). The actuator coupled to the IoT system becomes a moving robot. Robot is a complex of various IoT systems [22].
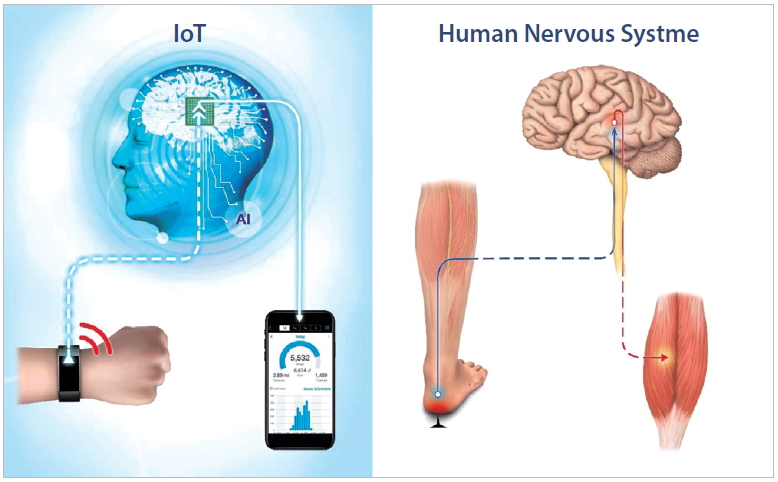
The concept of Internet of Things (IoT) system is very similar to that of human nervous system. AI, artificial intelligence.
In terms of IoT, the digital biomarker represents digitized data acquired from patients via IoT devices. The biomarker is defined as an objective parameter and indicator of normal biological, and pathological processes, or represents pharmacological response to therapeutic intervention [23]. Therefore, the digital biomarker can be defined as a biomarker that is objectively and quantitatively measured using digital devices and be used to explain or predict health-related outcomes [24]. Digital biomarker is measured using the digital tools that include portable, wearable, implantable or digestible devices, and exclude data obtained via patient-reported measurements or traditional devices and equipment. The most representative examples of a digital biomarker includes heart rate, physical activity and steps measured using a smart band or smart watch [25]. In a broad sense, digital biomarker include all human data that can be measured using digital tool. Therefore, biomedical signals that directly or indirectly reflect health state such as physical activity, skin conductivity, body temperature, electrocardiogram, heart rate, blood sugar, oxygen saturation, electroencephalography, and electromyography (EMG) are potential be digital biomarkers [26].
To summarize the relationship between the IoT, digital biomarker, and AI, the biomedical signal is acquired from a sensor emitted by digital device, such as a wearable device. Basically, the signal is digital biomarker. AI analyzes digital signals in real time, processes them into meaningful data, and automatically provides a feedback service to patients or medical staff. The application of IoT in the medical field is closely linked to AI and digital biomarkers. Thus, AI and digital biomarker are components that are closely connected with medical IoT. AI can be used to learn and analyze vast amounts of data generated by real-time monitoring. In addition, AI facilitates diagnosis and prediction of disease progression that can be not recognized by humans.
THE EMERGENCE OF MEDICAL INTERNET OF THINGS AND DIGITAL BIOMARKERS
Recently, the application of IoT technology in healthcare has been in the spotlight [27]. According to the report, the global IoT in health care market size is expected to reach USD 534.3 billion by 2025 [28]. The term Internet of Medical Things is used to represent the medical application of IoT [29]. A large portion of medical IoT is used to monitor patients in real time using implanted medical devices or wearable external medical devices. Real-time monitoring can facilitate patients’ diagnosis and prompt treatment, contributing to lower medical costs [30]. Currently, IoT technology in healthcare has been developed for real-time monitoring of blood glucose in diabetes mellitus, and auto-regulation of blood glucose by controlling the insulin pump [13,14]. In addition, the IoT has been developed to monitor chronic diseases such as hypertension, arrhythmia, and chronic obstructive pulmonary disease [31]. A recent area of interest is to monitor patients with Parkinson and Alzheimer disease in real time to provide medical staff with information about progression and treatment response [32].
The advantage of the medical IoT is to reduce unnecessary hospital visits and hospitalization by monitoring patients remotely and in real time [31]. The IoT can be used to continuously monitor patients and provide real-time data to medical staff to facilitate prompt and timely intervention. In addition, accurate data collection, automated systems and data-driven decisions can reduce time and system costs. New drug development and clinical trials are an expensive area in healthcare industry and the use of IoT processes and devices is a cost-effective strategy [33,34].
A digital biomarker associated with IoT is primarily characterized by objectivity [26,32,35]. Patient reported outcome (PRO) using Questionnaires, a traditional method for evaluation of patients is subjective data because it is influenced by patient’s ability to recall and emotional involvement at the time of examination. Digital biomarkers are very objective data with limited subjective influence of patients and doctors [36]. Whereas traditional assessment methods are measured only during hospital visits, the digital biomarker is measured in real world including home and provides accurate information about patient’ health state and performance in real life. The digital biomarker is measured continuously to obtain an abundance of data from a single patient. The objective and abundant digital biomarker is excellent data for AI and machine learning.
Because of the advantages, they are used in medical IoT, such as real-time monitoring of Parkinson disease or Alzheimer disease [32]. Also in clinical trials of new drug, compared with the traditional clinical trials, the effect of new drug can be measured more objectively with a digital biomarker in fewer patients. Therefore, pharmaceutical companies and researchers are highly interested in digital biomarkers [34]. In particular, real-time monitoring of digital biomarker using wearable device and smartphone has been already used in clinical trials of new drug in Parkinson disease. U.S. Food and Drug Administration-approved devices and software programs have been used in clinical practice [37]. Recently, the area of digital biomarkers has been expanded and the studies for monitoring mental health and illness by based on individual internet searches data or social media posts as digital biomarkers have also attracted attention [26].
INTERNET OF THINGS AND DIGITAL BIOMARKERS ASSOCIATED WITH SPINE
To date, AI researches in the field of spine have focused on predicting outcome after spinal surgery with preoperative data and diagnosing spinal images [5]. Research about IoT-based monitoring and digital biomarker in the spine is still in early stages. Currently, the focus of interest in IoT and digital biomarkers in the spine is on the assessment of surgical outcomes by objectively analyzing the functional status of patients with degenerative spinal disease [35,38].
Before exploring the role of IoT and AI in the spine, it is important to understand the digital biomarkers of the spine. The severity of spinal disease is measured in term of pain intensity and functional state. The patient’s functional state is best represented by daily physical activity. Pain due to spinal disease eventually results in a decrease in daily physical activity and poor quality of life. Several items in Oswestry Disability Index (ODI) and 36-Item Short Form Survey, which are widely used to evaluate patients with lumbar disease, are related to daily physical activity [39]. A representative sensor to measure the physical activity is an accelerometer. Physical activity measured with a wearable device equipped with an accelerometer is the most typical digital biomarker in spine [35]. Walking is the basic element of physical activity, which is measured as the number of steps or walking distance per day. A smart band or watch is a typical IoT device with built-in accelerometer [31]. Recent studies to objectively measure physical activity of spinal patients have used various sensors and devices, which were validated via comparison with PRO [38]. Kim et al. [35] monitored the daily physical activity of patients undergoing lumbar surgery for 7 days using a smart band. In the study, the physical activity was significantly correlated with daily visual analogue scale (VAS) and ODI one month after surgery [31]. Other objective outcome measurement could be a digital biomarker in degenerative disease of lumbar spine such as Timed Up and Go test, Five-Repetition Sit-To-Stand test and smartphone-based 6-minute walking test [40].
Spinal posture measured using a wearable device can also be digital biomarker in spine [41]. By monitoring spinal posture in real time, feedback can be provided in case of persistent bad posture. The feedback can be used to take steps to prevent spinal disease caused by bad posture. Indeed, various IoT devices used for real-time monitoring of spinal posture are already available commercially [41,42].
Human gait can also serve as a digital biomarker in spine [38]. In Parkinson disease, gait pattern has been monitored via sensor built-in shoes or wearable devices [43]. Gait disturbance is an important sign of compressive myelopathy [44,45]. If gait is monitored in real time by sensors mounted on shoes or wearable devices, it is possible to promptly diagnose the progression of myelopathy in patients with cervical stenosis or ossification of posterior longitudinal ligament. Hand dexterity can be a digital biomarker in patients with cervical myelopathy. It can be measured by alternately pressing 2 touchscreens on the apps of smartphone and smart pad and analyzing the speed of touch [46,47].
EMG has been widely used to evaluate spine patients. EMG signal might be a digital biomarker. Needle EMG has been performed only in hospital environment. According as the accuracy of surface EMG has been improved, it is possible to monitor muscle condition in real time by connecting the communication module to surface EMG [48].
Pain is the most common symptom of spinal disease and an important indicator in treatment decisions and follow-up. VAS or numeric rating scale is the most common parameter used to measure pain assessment in spinal patients [49]. Such pain assessments are highly subjective because they are measured by the patient’s response to the questionnaire or the examiner’s queries during the hospital visit. The subjective scales are also influenced by several factors, including mood at the time of visit and adaptation by repeated measures. To date, there is no definitive biomarker that directly reflects pain. Even a digital biomarker that reflects pain indirectly is effective in many ways if measured objectively, in real time, and in real world with a digital device. In that sense, the physical activity that can be measured with an accelerometer can also serve as an indirect digital biomarker of pain [35]. Recently, there is a great interest in the development of a digital pill that monitors drug usage by attaching sensor to the drugs [50]. Depending on the severity of pain, the doses of pain killer will increase. Therefore, pain can be objectively measured by monitoring usage of digital painkiller in real time.
Thus, various digital biomarkers are available in spine and medical IoT using these digital biomarkers can facilitate diagnosis and treatment of spinal diseases The IoT system can be used to monitor drug effects in clinical trials, or outcomes after spinal surgery. It is possible to develop an IoT system in which AI can be used to detect paralysis or deterioration of myelopathy early in patients with spinal diseases. Wearable devices that manage spinal posture are already commercially available, and research involving monitoring spinal posture is continuously conducted using IoT of daily objects such as chairs and glasses [42].
FUTURE OF INTERNET OF THINGS AND DIGITAL BIOMARKER IN SPINE
IoT in spine can be used to not only monitor and analyze the digital biomarkers in real time via wireless network, but also to provide useful feedback service or solution.
The IoT system, which monitors the electrocardiogram of patients with arrhythmia, monitors the abnormality in real time and delivers it to patients and medical staff [51]. Thus, it is valuable for prevention of acute cardiac arrest or improvement of survival rate in patients requiring urgent treatment. However, development of digital biomarker and IoT systems in spine is still in early stages of research. Further studies are needed to determine the type of feedback service for the diagnosis and treatment of spinal diseases. The economic impact of IoT system on the treatment of spinal diseases is still unknown [33].
The disadvantage of wearable devices is poor patient’ compliance. 46 After all, unless patients wear or use a digital device, the benefit of IoT and AI cannot be optimized. The fundamental solution to low compliance is to insert an implantable device with a sensor into the human body [52]. In spine surgeries, implants such as pedicle screw, cages, and artificial disc are inserted into the patient. In the future, it may be possible to insert an instrument with sensor into a body to ensure real-time monitoring of patients. Of course, these IoT implants are still in the early stages of research and many challenges remain to be addressed, including safety [52]. However, implantable devices in spine are an attractive area to resolve low user compliance. Digital devices connected to the IoT are not limited to wearable devices or implants. As mentioned earlier, anything can serve as an IoT device. Surgical instruments and devices, such as endoscope and drill, can also be connected to wireless network to collect information [53]. Moreover, AI can provide meaningful services during surgical intervention. Thus, all object related to the spine can be spinal IoT devices (Fig. 2).
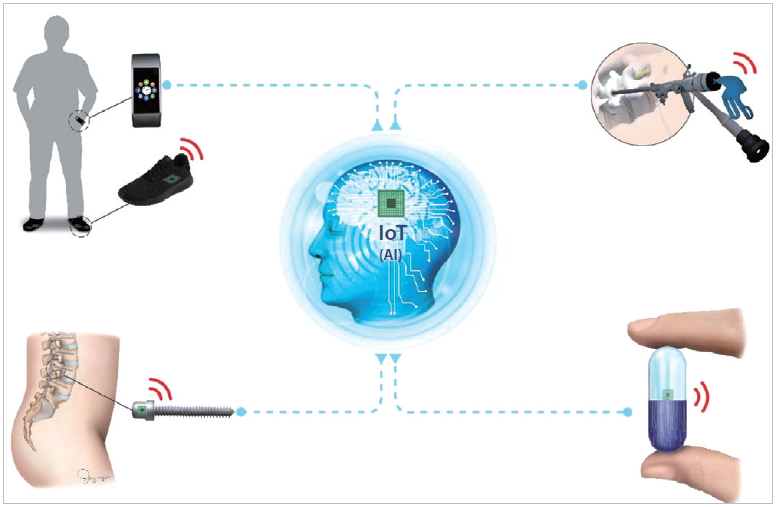
All object including smart watch, shoes, screws, surgical instruments, and pill can be a spinal Internet of Things (IoT) device. AI, artificial intelligence.
Despite the high potential for applications in future, there are many challenges that need to be addressed before the medical IoT can be used for diagnosis and treatment of patients. The accuracy of digital biomarkers and IoT device should be validated. Since the digital biomarker is a new concept in spine, it must be analyzed via comparison with traditional evaluation methods. Standardization of such digital biomarkers and IoT devices is essential [26].
The most important issue in limiting the application if IoT to patients is privacy [54]. Ethical issues may arise following leakage of personal health data. Also, in countries where the distribution of personal information is strictly restricted, the actual clinical application of such medical IoT may be difficult.
Diagnosis, treatment, and monitoring of patients using medical IoT have already entered the clinical phase despite the low interest of many clinicians. With the development of networks, sensors, and AI technology, the diversity and accuracy of medical IoT are expected to improve. The inherent value of medical IoT can be transformed into medical and economic usefulness based on ideas and creativity of the clinician who treats the patients.
AI research involving medical IoT and digital biomarkers in the spine stand at the starting point. Therefore, if spine surgeons understand the basic concepts and possibilities of IoT, digital biomarkers, and AI play an active role in research efforts, it may lead to paradigm shift in diagnosis and treatment of spinal diseases in future.
Notes
The authors have nothing to disclose.
Acknowledgements
This study was supported by the Technology Innovation Program (No. 20000515) funded by the Ministry of Trade, Industry & Energy (MOTIE, Korea).
