Transforaminal Endoscopic Surgery: Outside-In Technique
Article information
Abstract
Transforaminal endoscopic lumbar discectomy (TELD) with the outside-in technique can be applied to nearly all cases of lumbar disc herniation (LDH), and transpedicular endoscopic lumbar discectomy can be used to treat highly migrated LDHs. The purpose of this study was to outline these 2 outside-in surgical techniques and to present their clinical outcomes. Between January 2018 and January 2019, a total of 137 patients underwent either transforaminal or transpedicular endoscopic lumbar discectomy. We performed TELD in 124 patients and transpedicular endoscopic lumbar discectomy in 13 cases. All surgical procedures were performed under conscious sedation. The patients’ mean age was 51.3 years; 51 were women and 86 were men. The overall disc recurrence rate was 5.12%. Visual analogue scale scores decreased significantly in both groups. According to the MacNab criteria, good and excellent results were obtained in 92.74% of patients after transforaminal and in 92.30% of patients after transpedicular endoscopic LDH treatment. The results suggest that TELD with the outside-in technique can be effective for the treatment of most cases of LDH. Transpedicular endoscopic lumbar discectomy can be considered as an alternative treatment for highly migrated LDH.
INTRODUCTION
Transforaminal endoscopic lumbar discectomy (TELD) has been developed into a standard technique for minimally invasive spine surgery.
The pioneers of endoscopic spine surgery, such as Hijikata et al. [1] and Kambin and Gellman [2], began with percutaneous endoscopic spinal surgery. Yeung and Yeung [3] developed the Yeung endoscopic spine surgery technique, based on the inside-out approach, for the treatment of lumbar disc herniations (LDHs).
Hoogland et al. [4] and Schubert and Hoogland [5] introduced the outside-in technique using the transforaminal approach. In this technique, reamers (and later drills) are used to enlarge the neuroforamen, making it possible to approach the spinal canal through the neuroforamen and to remove sequestered disc herniations and fragments from the anterior epidural space.
Inside-out TELD is defined as primarily introducing a cannula into the disc (Fig. 1A), followed by removal of the sequestered nucleus pulposus [3,6-8].

Spine model. (A) Intradiscal access with a cannula (inside-out technique). (B) Extradiscal access with a cannula in the caudal neuroforamen (outside-in technique).
The access angle in Yeung’s technique is between 25° and 35°: therefore, the indications for his technique are restricted [3,7-9]. Yeung’s technique enables successful decompression of contained disc herniation, but movement of the cannula is limited outside of the disc.
Another inside-out TELD technique was described by Ruetten et al. [10]. This technique uses extreme lateral access at an angle of 10°, with the cannula touching the dorsal annulus and the posterior longitudinal ligament. It allows intradiscal and extradiscal decompression, but is not suitable for L5–S1 or the upper levels L1–2 and L2–3. To extend the endoscopic indications, Ruetten et al. [11,12] and Choi et al. [13] developed interlaminar endoscopic discectomy.
In outside-in TELD, the cannula is placed on the disc surface and in the neuroforamen (Fig. 1B). Some parts of the superior articular process (SAP), cranial parts of the caudal pedicle, and in some cases osteophytes of the lower vertebral body can be removed by drilling or shaving to decompress nerve roots [4-7,9].
For highly inferiorly and superiorly migrated disc herniation, this technique is challenging because of its extreme oblique approach. Hu et al. [14] described a modification of the Hoogland technique involving partial transpedicular endoscopy. They used a reamer technique with an extreme oblique access.
Krzok et al. [15-17] described transpedicular endoscopic surgery for highly superiorly and inferiorly migrated disc herniation and facet cysts [18]. Further case reports of Uniyal et al. [19] and Quillo-Olvera et al. [20] confirmed the successful use of this burr hole technique, which allows direct access to the sequestration behind the pedicle.
The objective of the present study is to prove the results, for a wide range of indications, of outside-in TELD and transpedicular lumbar endoscopic discectomy.
MATERIALS AND METHODS
Between January 2018 and January 2019, a total of 137 patients underwent outside-in transforaminal and transpedicular endoscopic lumbar discectomy. We performed TELD in 124 patients and transpedicular endoscopic lumbar discectomy in 13 cases.
All surgical procedures were performed by a single surgeon with long experience in endoscopic spinal surgery.
1. Inclusion and Exclusion Criteria
The inclusion criteria were: (1) radicular pain after at least 6 weeks of unsuccessful conservative treatment (medication, physical therapy, spinal injections with nerve blocks under a C-arm), (2) neurological deficit, (3) positive straight leg raising test, and (4) magnetic resonance imaging (MRI) confirmation of disc herniation (Fig. 2).

Magnetic resonance images of a 26-year-old woman with central disc herniation at L5–S1 (red arrow). T2-weighted sagittal (A) and axial images (B).
The transpedicular approach was used only in cases of highly superiorly or inferiorly migrated LDH.
The exclusion criteria were: (1) spondylolisthesis or other segmental instability, (2) central stenosis, (3) spinal tumor, or (4) level L5–S1 disease with an extremely high iliac crest and an access angle over 50°.
2. Surgical Procedures
All surgical procedures were performed under conscious sedation (remifentanil, propofol, and midazolam) supplemented with local anesthesia (2% ropivacaine). Patients were placed in the lateral position, lying down on the opposite site. Knees and hips were flexed to reduce lumbar lordosis. The patients were covered by a sterile drape after skin disinfection.
Following recommendations, the spinous process (the dorsal midline) and iliac crest were marked. The entry point depended on the disc level, but also on the size of the patient; on average, it was 12 cm from the midline for access to L4–5 and L5–S1, 10 cm for access to L3–4 and L2–3, and 8 cm for L1–2. The assumed direction to the sequestration was determined with forceps under C-arm control.
After local anesthesia, a spine needle with 10-G (2.6 mm) diameter was introduced until it reached the border between the cranial part of the caudal pedicle and the SAP. This large needle was stiff, which prevented bending and wrong positioning. A guidewire with a diameter of 2 mm and a sharp tip was introduced through the spine needle and was fixed to the bone by a gentle tap on the top of the mallet.
The spine needle was replaced by a Jamshidi needle. By tapping the mallet, the Jamshidi needle was advanced under imaging guidance until the medial pedicle line (Fig. 3).
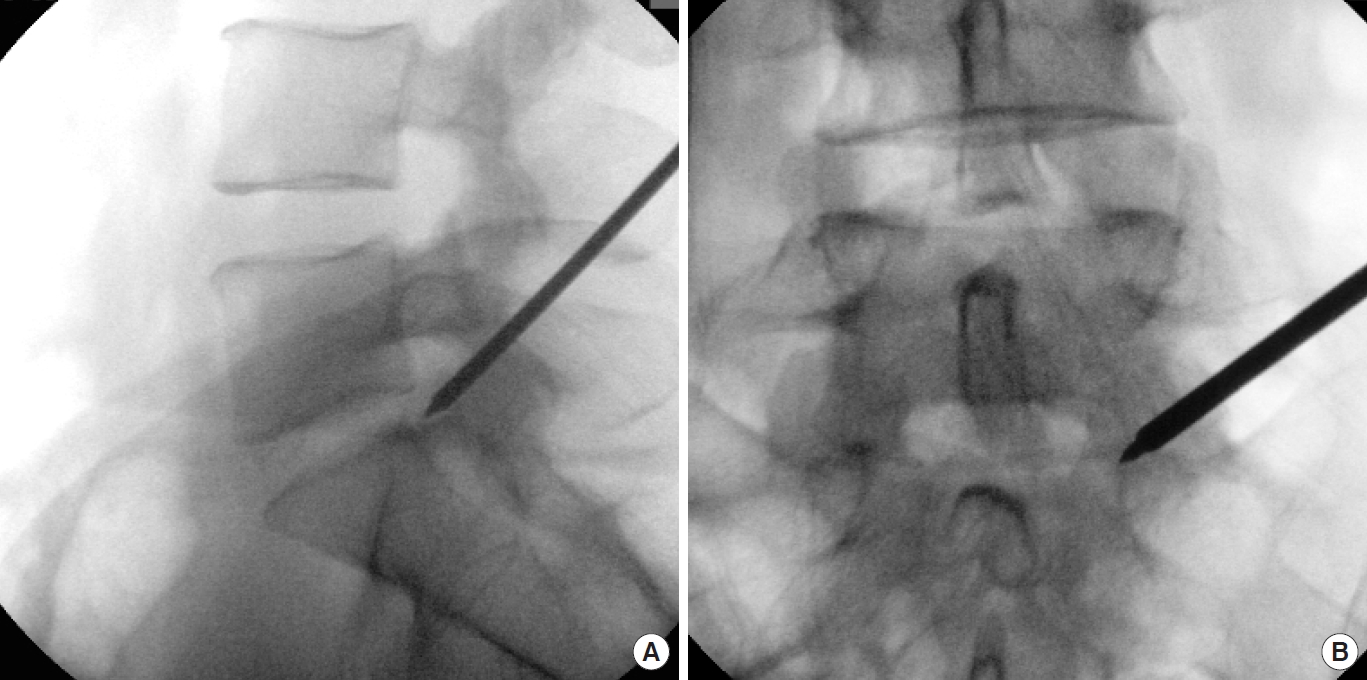
C-arm image of a Jamshidi needle passing through the superior articular process. (A) Lateral view: the needle direction to the endplate of S1. (B) Anteroposterior view: the tip of the needle touching the medial pedicle line.
Next, the soft tissue was dilated and manual drilling was performed with stepwise enlarging of the lower neuroforamen until 8 mm (Fig. 4A, B). The drill was advanced through the protective tube to avoid soft tissue trauma.

C-arm image of an 8-mm manual drill. (A) Lateral view of the drill used to increase the size of the lower foramen. (B) Anteroposterior view of the drill touching the cranial part of the S1 pedicle.
The working tube was inserted over the guiding rod (Fig. 5A, B). Discography was carried out with an 18-G needle through the working tube (Fig. 5A), using a mixture of 0.1 mL of toluidine blue dye and 4.9 mL of x-ray contrast medium (Ultravist 300, Bayer Pharma AG, Berlin, Germany).
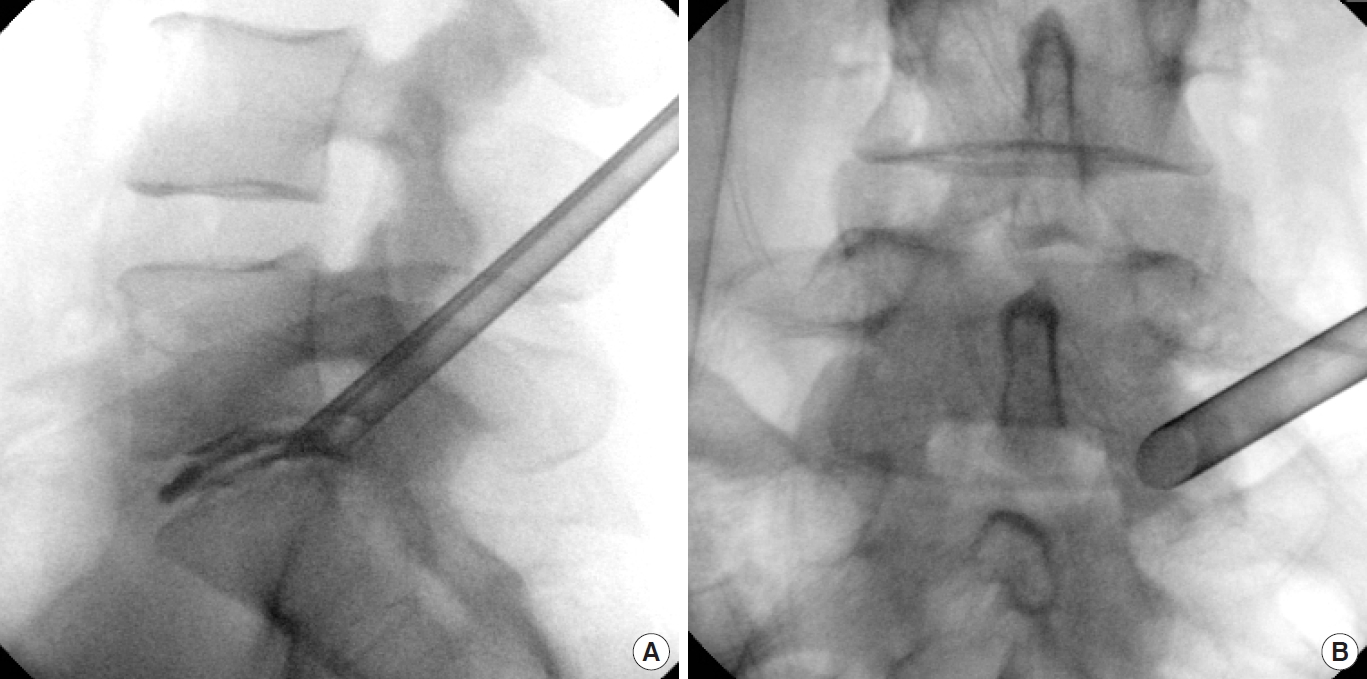
C-arm image of a working tube. (A) Lateral view of the working tube with discography. (B) Anteroposterior view with the working tube inside the lower neuroforamen.
A 25° spinal endoscope with a working channel of 4.1 mm (RIWOspine, Richard Wolf GmbH, Knittlingen, Germany) was inserted through the working tube. Under an endoscopic view, the anatomical landmarks were identified. If the patient was in the side position and the working tube was facing the window to the spinal canal, the resected bone of the SAP was found at 12 o’clock and the disc space was located in the 6 o’clock direction (Fig. 6A).
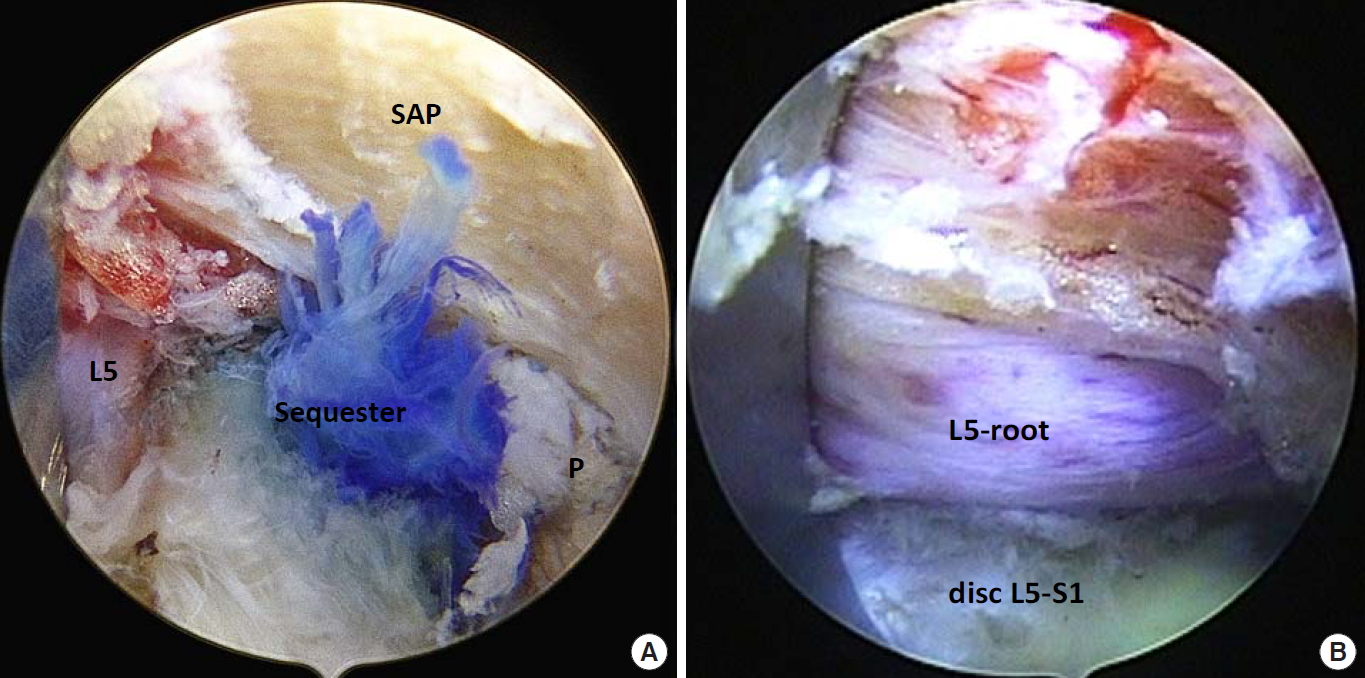
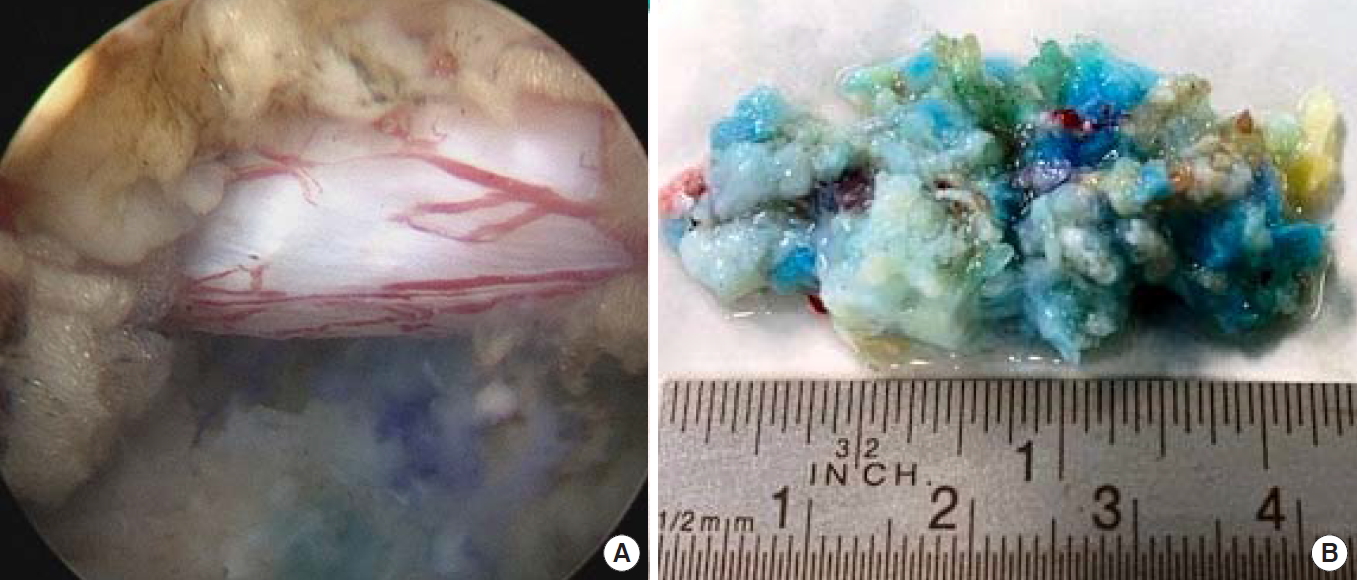
Endoscopic view of L5–S1. (A) The blue-colored disc herniation, resected parts of the superior articular process (SAP), caudal pedicle, and cranial L5 root. (B) The L5 exiting root after turning the working tube to the cranial direction.
The sequestration of the nucleus pulposus was blue-dyed and could be easily identified (Fig. 6A). The caudally located working tube provided a view of the lower part of the neuroforamen with the SAP, and the cranial border of the lower pedicle and the medial part of the lower vertebral body could be found. By turning the working tube to the cranial direction and advancing to the ground plate of the upper vertebra, the exiting nerve root could be clearly distinguished (Fig. 6B).
The intraspinal position of the forceps should be controlled by C-arm imaging (Fig. 7A, B). Depending on the pathology, decompression of the roots and the epidural space (Fig. 8A) can be performed by removing the disc sequestration (Fig. 8B). Additional use of high-speed burrs (Powerspeed AS1, Richard Wolf GmbH, Knittlingen, Germany) to remove the bone spurs improves the endoscopic decompression.
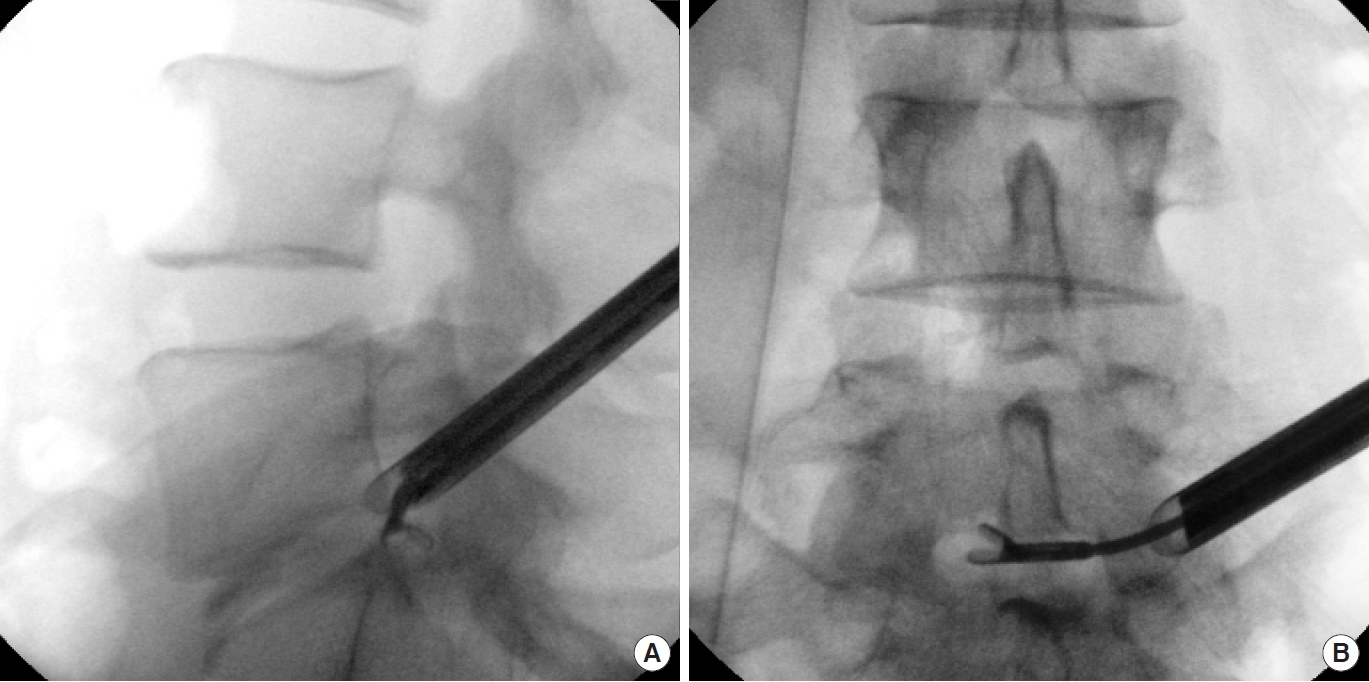
C-arm image of working tube and forceps. (A) Lateral view showing the caudal sequestration being caught. (B) Anteroposterior view with forceps anterior to the dural sac.
The surgery was finished when the exiting root was free and the epidural space moved in a pulsating manner.
3. Transpedicular Endoscopic Surgery
Highly inferiorly or superiorly migrated (Fig. 9) disc herniations were treated through postero-lateral transpedicular access. The entry point was parallel to the pedicle or slightly oblique from the cranial with the same distance as in the transforaminal outside-in technique.
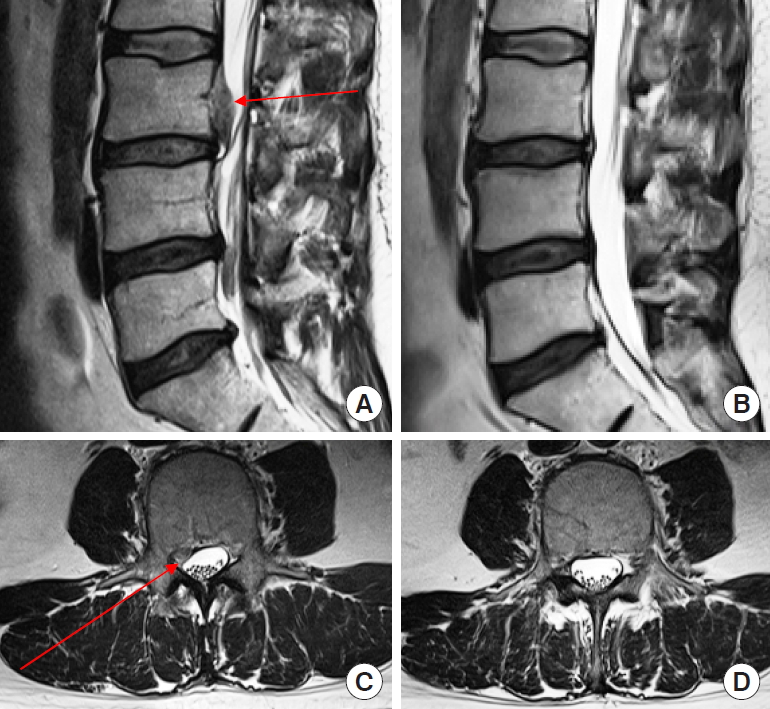
Magnetic resonance images of a 43-year-old man with cranial sequestration at L3–4 on the right side (red arrows). (A) T2-weighted sagittal view with a drop-shaped sequestration and (B) complete removal of the sequestration 3 weeks after surgery. (C) Axial images with intraspinal compression. (D) Decompression of the spinal canal 3 weeks after surgery.
The Jamshidi needle was advanced to the middle of the pedicle parallel to the transverse process until the medial border of the pedicle (Fig. 10). In contrast to the first description of this technique by Krzok et al. [15-17], access posterior to the transverse process was preferred, as reported by Quillo-Olvera et al. [20].

C-arm image of a Jamshidi needle being advanced in the anteroposterior view (A, B) and in the lateral view (C, D). Start of the pedicle puncture parallel to the transverse process in the middle of the pedicle (A, C), stopping at the medial wall of the pedicle behind the vertebral body (B, D).
By careful mallet blows, and lastly by manually advancing the Jamshidi needle (Fig. 10B, D), the medial border of the pedicle was passed. The surgeon felt this moment by loss of resistance, and patients sometimes experienced leg pain.
After stepwise manually drilling until 8 mm (Fig. 11A, B), the working tube was inserted into the burr hole.

C-arm image of the 8-mm manual drill passing through the pedicle in lateral (A) and anteroposterior views (B).
Discography was carried out over an extra puncture of the disc (Fig. 12A, B). The removal of the sequestration should be controlled by C-arm imaging (Fig. 13A, B).
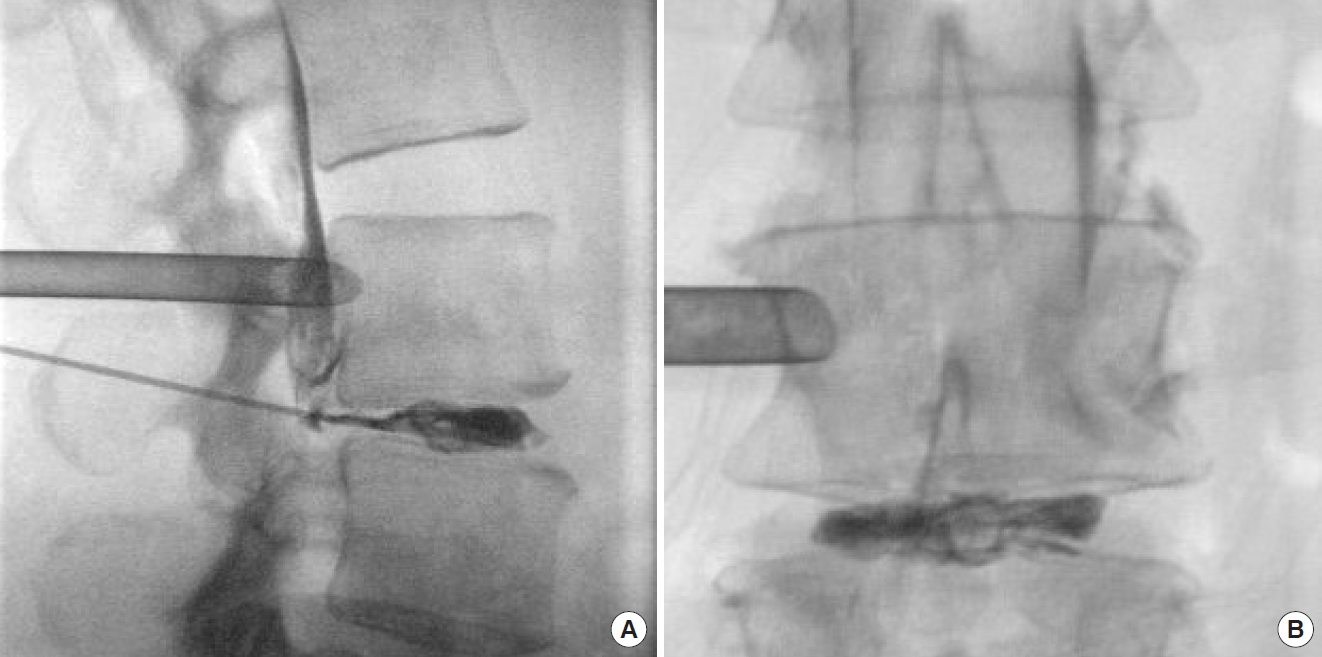
C-arm image after insertion of the working tube shows discography with a stained cranial sequestration in lateral (A) and anteroposterior views (B).

C-arm image with transpedicular removal of a highly superiorly migrated sequestration with the forceps in lateral (A) and anteroposterior views (B) in the cranial-caudal direction.
Endoscopic exploration confirmed highly migrated sequestration, which was partially or totally blue-dyed behind the pedicle (Fig. 14A) and the compressed traversing nerve root.
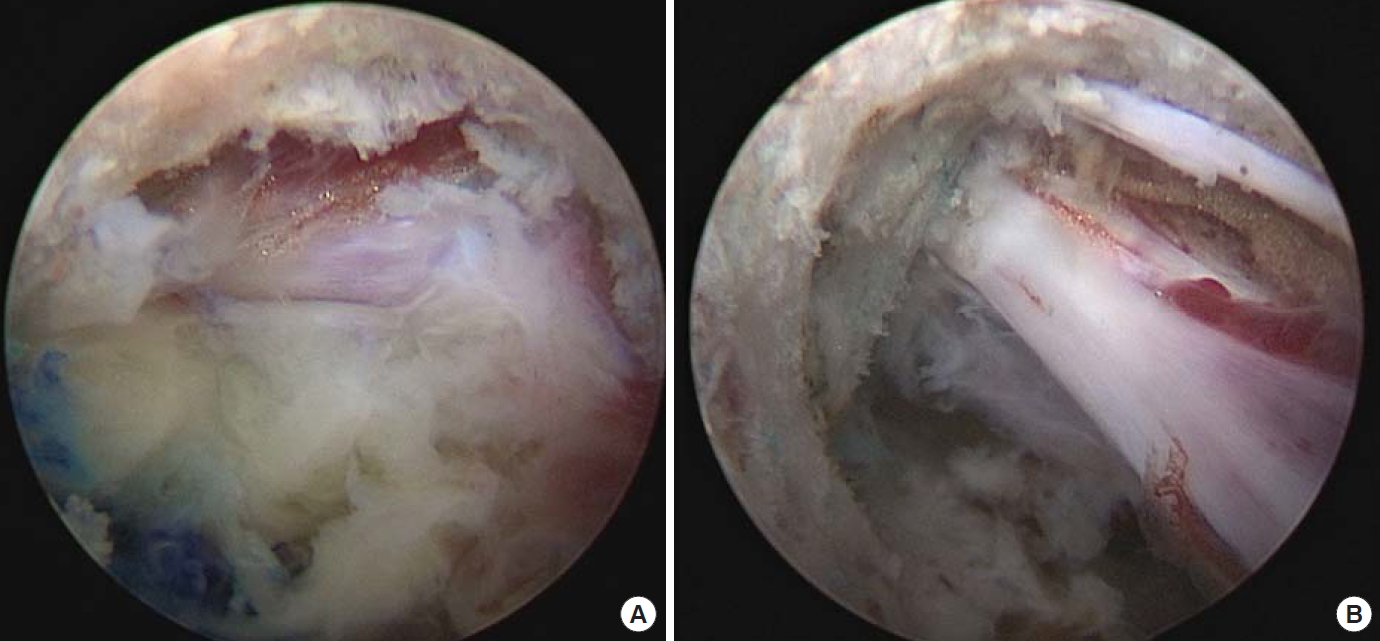
Endoscopic view through the burr hole of the pedicle. (A) The cranial sequestration with compression of the traversing L3 nerve root. (B) Decompressed L3 nerve root.
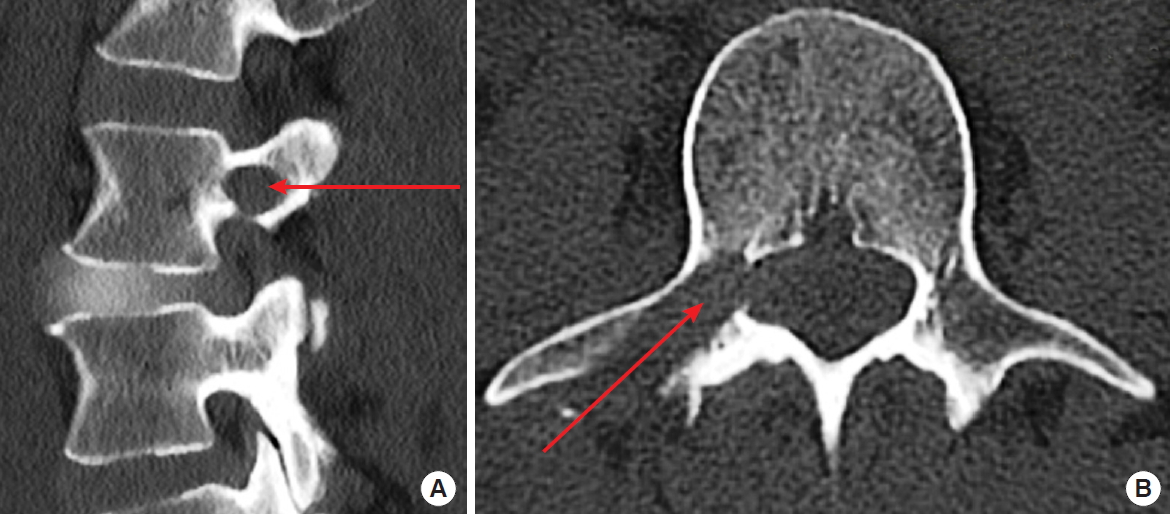
Computed tomography was performed in every case after transpedicular endoscopic surgery to exclude pedicle fracture, at 1 day (Fig. 15) and 6 months postoperatively. MRI was done in some special cases, but not routinely.
RESULTS
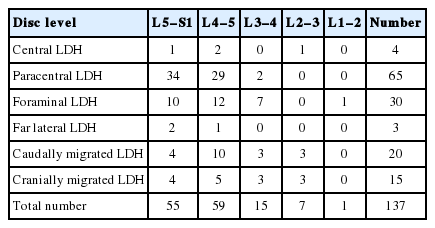
The patients included 51 women and 86 men. The patients’ age varied from 22 to 82 years (average age, 51.3 years). The LDHs were mostly located in the lower lumbar spine with 55 cases (40.15%) at the L5–S1 level and 59 cases (43.06%) at the L4–5 level, followed with 15 cases (10.95%) at the L3–4 level, 7 cases (5.11%) at the L2–3 level, and 1 case (0.73%) at the L1–2 level (Table 1). The average duration of surgery was 54 minutes. The mean blood loss was 12 mL. Only 2 cases needed Redon’s suction drainage for 1 night, with blood loss of 60 and 80 mL, respectively. No infection occurred; especially, no discitis was observed. Three patients experienced a headache on the first day after surgery, but without signs of liquor loss. No intraoperative dura leakage was observed. LDH recurrence took place in 7 cases (5.12%), of which 3 were repeated recurrences.
In 2 cases, we performed surgery at 2 levels. The most commonly treated disc levels were in the lower lumbar spine (levels L4–5 and L5–S1) (Table 1). In terms of localization, most of the LDHs were paracentral (n = 65), followed by foraminal (n = 39), caudally migrated (n = 20), cranially migrated (n = 15), central (n = 4), and far lateral (n = 3).
The VAS score for leg pain improved from 7.72 to 2.12 after TELD (Table 2). According to the MacNab criteria (Table 3), 52 patients (41.94%) had excellent results. Good results were seen in 63 patients (50.81%), fair in 8 patients (6.45%), and poor in 1 patient (0.81%). More than 92% of the patients had rapid relief of leg pain and showed improvement of motor function.

Visual analogue scale (VAS) scores for leg pain after transforaminal endoscopic lumbar discectomy (n=124)

MacNab criteria after transforaminal endoscopic lumbar discectomy up to 12 months postoperatively (n=124)
TELD was performed in 19 cases for recurrent disc herniation, including 8 cases after interlaminar microscopic discectomy and 11 cases after transforaminal endoscopic discectomy.
Therefore, excellent and good results were obtained in 16 cases. In 2 cases, leg pain did not change postoperatively after primary microscopic interlaminar discectomy due to scar compression of the root, and in 1 case, leg pain did not improve due to a facet fracture .
In an uncommon case in a 40-year-old woman, we found a huge recurrent herniation at L5–S1 (Fig. 16A), compressing the S-root (Fig. 16B). She underwent the first surgery at L5–S1 by microscopic interlaminar discectomy at age of 30. Because of recurrent herniation at L5–S1, transforaminal endoscopic revision surgery was performed 3 times, at 3, 8, and 9 years after the first procedure. She always recovered very early and was satisfied with the endoscopic surgeries.

Magnetic resonance images of a 40-year-old woman after 1 interlaminar and 3 transforaminal revision surgeries at L5–S1. T2-weighted sagittal (A) and axial images (C) of huge paracentral recurrent disc herniation. T2-weighted sagittal (B) and axial images (D) after decompression in the fifth transforaminal endoscopic lumbar discectomy 6 months postoperatively.
Before the fifth surgery, she suffered from severe leg pain, numbness, and weakness of foot extension on the right side. MRI showed a huge recurrent herniation (Fig. 16A, C) with S1 compression on the right side. These findings were an indication for fusion of L5–S1 and L4–5, but the patient refused open disc surgery and fusion, which was why endoscopic surgery for LDH was performed for the fifth time.
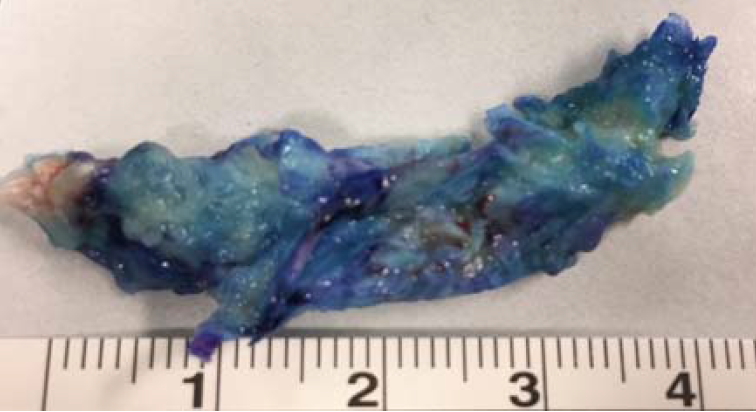
The decompression of the neuroforamen was very challenging in this case because of scar formation. Chromo-discography with toluidine blue was very helpful (Fig. 17). The blue color dyed only the nucleus pulposus and made the endoscopic orientation more straightforward (Fig. 18). At last, a huge sequestration (Fig. 19) was removed. A clinical examination 6 months later with MRI imaging showed sufficient decompression (Fig. 16B, D). The patient recovered quickly, with no leg pain or any signs of nerve root compression during follow-up, but she reported low back pain after long periods of standing or sitting. Her outcome was good according to the MacNab criteria.
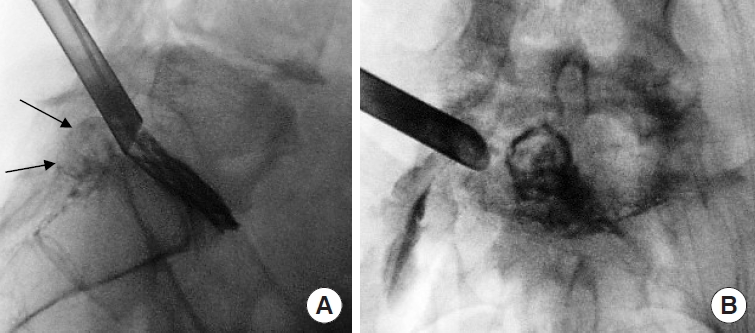
C-arm image after discography. (A) Sagittal view of discography through the working tube with a huge stained caudal sequestration (arrows). (B) Anteroposterior view of the working tube used for intraspinal access.
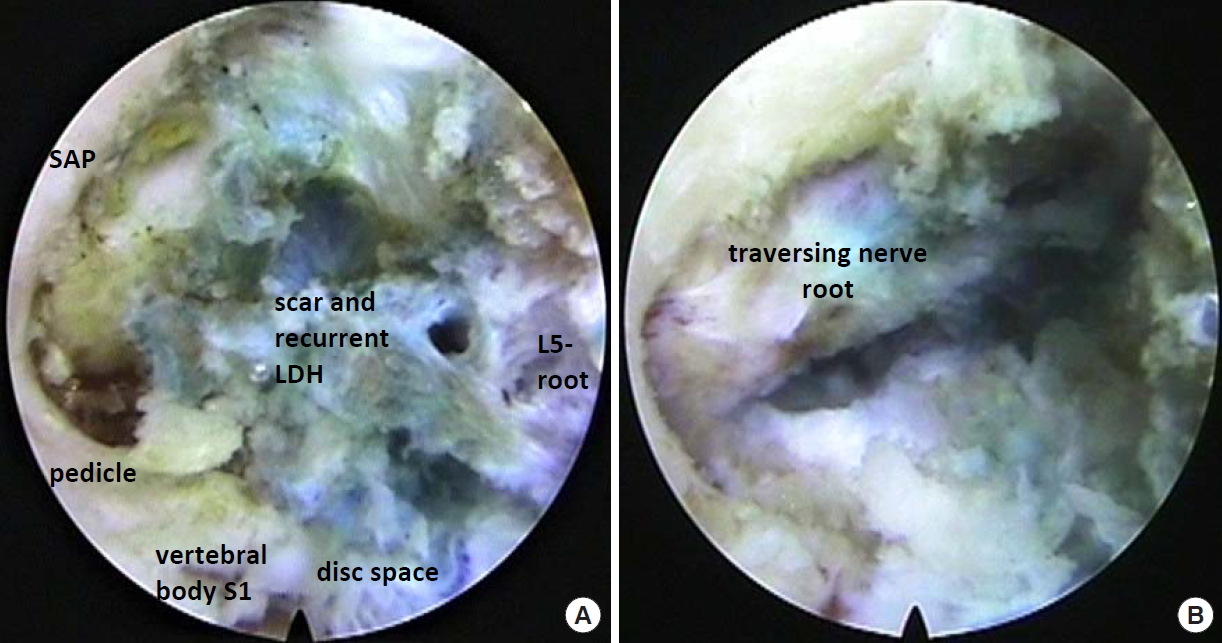
(A) Endoscopic view of the neuroforamen of L5–S1, filled with recurrent disc herniation and scar tissue. (B) After decompression of the traversing nerve root. SAP, superior articular process; LDH, lumbar disc herniation.
One complication occurred after revision surgery in a 55-year-old woman after recurrence of a foraminal herniation at L5–S1 on the left side. One day after surgery, the patient developed drop foot on the left side (M1–2). An MRI examination showed a foraminal compression at L5–S1 on the left side due to a bone fragment. Urgent transforaminal revision surgery was performed 1 day after surgery and the bone fragment of the facet joint was removed from the L5 root (Fig. 20). The patient recovered within 1 week with better muscle function of foot extension (M4), but after 6 months she underwent fusion surgery at L5–S1 because of low back pain and weakness.

Endoscopic view of the neuroforamen of L5–S1 with L5 root compression by a bone fragment. SAP, superior articular process.
1. Results After Transpedicular Endoscopic Discectomy
The 13 patients who underwent transpedicular lumbar discectomy (Table 4) included 4 women and 9 women, with a mean age of 59.23 years. This technique was only used in cases of highly inferiorly migrated LDH (7 cases) and highly superiorly migrated LDH (6 cases).

Localization of lumbar disc herniation (LDH) in patients who underwent transpedicular endoscopic lumbar discectomy
All patients with highly migrated disc herniation suffered from severe leg pain (VAS score, 8.19) before surgery and showed a rapid relief of pain after transpedicular surgery (VAS score 3.07 after 2 weeks and 1.88 after 12 months) (Table 5). Two patients needed additional transforaminal endoscopic surgery in the same procedure. In these cases, the sequestrations were cranially and paracentrally located.

Visual analogue scale (VAS) scores for leg pain after transpedicular endoscopic lumbar discectomy (n=13)
One case with a highly superiorly migrated sequestration required transforaminal endoscopic revision surgery because of foraminal recurrence 4 months later. Neurological failure occurred in 5 cases preoperatively, with weakness of foot and big toe extensor paresis in 4 cases and one with quadriceps paresis; these patients recovered within the 6-month period. No pedicle fractures were observed.
The MacNab criteria showed excellent results in 8 patients (61.54%), good results in 4 patients (30.77%), and fair results in 1 patient (7.69%) in this group (Table 6).
DISCUSSION
Transforaminal endoscopic surgery with the outside-in technique offers more possibilities to treat disc herniation of the lumbar and lower thoracic spine. In contrast to the inside-out technique [1-3], the access to the neuroforamen is not made primarily through the disc. Stepwise increasing of the size of the neuroforamen by using manual drills or reamers allows an endoscopic view of the spinal canal with the neurological structures and disc herniation.
When primarily accessing the disc, the movement of the endoscope is limited. For successful endoscopic decompression of roots and epidural space, an unlimited ability to move the cannula is necessary.
Increasing the size of the lower part of the neuroforamen was achieved by removing parts of the SAP and the superior margin of the lower pedicle by manual drilling. Additional endoscopic decompression with removal of bone spurs can be done by high-speed burring.
The working tube should be placed through the drill hole at the endplate of the lower vertebra (Fig. 5A, B) or behind the lower vertebral body. The original outside-in technique of Hoogland et al. [4] and Schubert and Hoogland [5] described first puncturing the disc, with optional injection of contrast medium. The modification of his technique consisted of primarily increasing the size of the lower neuroforamen and inserting the working tube there. Advancing a spine needle through the working tube into the disc and injecting contrast medium prevented the cannula from being inserted into the disc in the wrong direction (wrong inside-out technique).
The advantage of discography with toluidine blue was that it improved endoscopic orientation in patients with severe degenerative disc disease or recurrent LDH (Fig. 18A, B). The dye distribution in the LDHs in the present study was nearly equal between the L5–S1 and L4–5 levels.
The previous studies of Kim et al. [9,23] reported a higher frequency of percutaneous endoscopic transforaminal lumbar discectomy at the L4–5 levels. Access to the L5–S1 level was challenging, but possible in most cases, with the exception of those with extremely high iliac crests.
The technique described herein is demanding and the learning curve is steep. The key point of this technique is precise needle placement on the target of the SAP or pedicle and careful manual drilling under C-arm guidance. The needle and the cannula should not enter the medial pedicular line.
The results after 1 year of follow-up showed good and excellent results according to the MacNab criteria in 92.74% of the 124 patients who underwent TELD with the outside-in technique.
One woman had a poor result of transforaminal repeated revision of foraminal disc recurrence at the L5–S1 level according to the MacNab criteria. The cause was a bone fragment of the facet joint that caused compression of the L5 nerve root (Fig. 19). Too extensive bone resection of the SAP by manual drilling and high-speed burring can lead to facet fracture. This complication is rare. The surgeons should also prevent access through the facet joint space.
Caudally and cranially migrated LDHs occurred in 35 cases. Low-grade disc migration was found in 22 cases and high-grade disc migration in 13. LDHs with low-grade migration were treated by TELD according to Ahn et al. [24] and highly migrated LDHs by transpedicular endoscopic lumbar discectomy [14,15].
In the small group of 13 patients who underwent transpedicular endoscopic surgery, we found 1 case with incomplete removal of the sequestration; this patient needed transforaminal endoscopic revision surgery. Most patients were satisfied with the surgical outcomes (92.31% according to the MacNab criteria). The preoperative VAS score in this group was higher, as was the postoperative decrease in pain. In 2 cases, it was necessary to combine the transforaminal and transpedicular techniques because of incomplete removal of the sequestration. Unlike the study of Ahn et al. [24], the results after transpedicular and transforaminal access were equivalent. The advantage of the technique of Ahn et al. [24] was combined transforaminal und interspinal decompression, while the disadvantage was the very oblique access with the danger of incomplete removal of the disc sequestration. The advantage of the transpedicular technique was direct and parallel access to the sequestration through the pedicle. However, it was disadvantageous in cases of combined paracentral and highly migrated LDH because additional transforaminal access was needed.
Krzok [15] reported 2 cases with a fracture of the transverse process, but without any pedicle fractures.
CONCLUSIONS
The results suggest that TELD with the outside-in technique can be effective for the treatment of most cases of LDH, with the exception of LDH at L5–S1 in patients with a high iliac crest and an extremely oblique access angle. Transpedicular endoscopic lumbar discectomy can be considered as an alternative treatment for highly migrated LDH.
Manual drilling with stepwise enlarging of the neuroforamen and additional use of the high-speed burr under endoscopic view improved the endoscopic transforaminal decompression.
Notes
The author has nothing to disclose.
SUPPLEMENTARY MATERIALS
Supplementary video clip 1 can be found via https://doi.org/10.14245/ns.2040128.064.v.1.