Unilateral Biportal Endoscopy for Decompression of Extraforaminal Stenosis at the Lumbosacral Junction: Surgical Techniques and Clinical Outcomes
Article information
Abstract
Objective
The aims of this study were to describe the unilateral biportal endoscopic (UBE) technique for decompression of extraforaminal stenosis at L5–S1 and evaluate 1-year clinical outcomes. Especially, we evaluated compression factors of extraforaminal stenosis at L5–S1 and described the surgical technique for decompression in detail.
Methods
Thirty-five patients who underwent UBE decompression for extraforaminal stenosis at L5–S1 between March 2018 and February 2019 were enrolled. Clinical results were analyzed using the MacNab criteria, the visual analogue scale (VAS) for back and leg pain, and the Oswestry Disability Index (ODI). Compression factors evaluated pseudoarthrosis within the transverse process of L5 and ala of sacrum, disc bulging with or without osteophytes, and the thickened lumbosacral and extraforaminal ligament.
Results
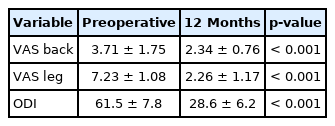
The mean back VAS was 3.7±1.8 before surgery, which dropped to 2.3±0.8 at 1-year postoperative follow-up (p<0.001). There was a significant drop in postoperative mean VAS for leg pain from 7.2±1.1 to 2.3±1.2 at 1 year (p<0.001). The ODI was 61.5 before surgery and 28.6 (p<0.001). Pseudoarthrosis between the transverse process and the ala was noted in all cases (35 of 35, 100%). Pure disc bulging was seen in 12 patients (34.3%), and disc bulging with osteophytes was demonstrated in 23 patients. The thickened lumbosacral and extraforaminal ligament were identified in 19 cases (51.4%). No complications occurred in any of the patients.
Conclusion
In the current study, good surgical outcomes without complications were achieved after UBE decompression for extraforaminal stenosis at L5–S1.
INTRODUCTION
Extraforaminal stenosis in L5–S1, or far-out syndrome (FOS), was initially reported by Wiltse et al. [1] in 1984, who demonstrated that an L5 nerve could be compressed by the transverse process (TP) of the L5 and the ala of the sacrum. A preoperative diagnosis of extraforaminal stenosis at L5–S1 is important because a lack of diagnosing extraforaminal stenosis at L5–S1 can result in failed back surgery syndrome [2,3]. In the past, using a paraspinal approach for microsurgical decompression of foraminal lesions had been considered the gold standard for surgical treatment of lumbar foraminal or extraforaminal stenosis [1,4,5]. But, access to the L5–S1 extraforaminal region is difficult for 2 reasons: (1) the distance between the L5 TP and sacral ala is short, and (2) the iliac crest generally makes the operative corridor very narrow [6-9]. Furthermore, significant manipulation of the dorsal root ganglion can result in postoperative leg pain or dysesthesia, and the surgery is technically challenging and more invasive due to the deep location. As a result, some studies report a high rate of chronic or recurrent radiculopathy following FOS microsurgical decompression [10,11].
Lately, due to advancements in endoscopic spine surgery, the unilateral biportal endoscopic (UBE) technique has been applied in the cervical, thoracic and lumbar spine [12-15]. For extraforaminal stenosis at L5–S1, the UBE technique, which can go further into the extraforaminal lesion less invasively, is becoming widespread and surpassing microscopic surgery in popularity. The purpose of this study is to present the UBE technique for complete decompression of extraforaminal stenosis at L5–S1 and evaluate 1-year clinical outcomes. Additionally, we evaluated the compression factors of extraforaminal stenosis at L5–S1 and described the surgical technique for complete decompression in detail compared to previous reports. Especially, we describe (1) a surgical corridor that can allow for effective removal of the thickened lumbosacral and extraforaminal ligament and (2) an anatomical landmark (thickened lumbosacral and extraforaminal ligament), one of the compression factors of extraforaminal stenosis at L5–S1 that shows how far to decompress laterally.
MATERIALS AND METHODS
1. Patient Population
After obtaining informed consent from Institutional Review Board approval (P01-202003-21-005), we retrospectively reviewed patients with extraforaminal stenosis at L5–S1 who underwent decompression using UBE between March 2018 and February 2019 at our institution. The indications for UBE decompression for extraforaminal stenosis at L5–S1 were the same as those for microsurgical decompression. The patients were eligible if they fulfilled the following inclusion criteria: (1) medically intractable unilateral L5 radiculopathy for at least 6 weeks; (2) L5–S1 extraforaminal stenosis with or without disc herniation confirmed on computed tomography and magnetic resonance imaging (MRI); (3) minimum follow-up for 1 year; and (4) no segmental instability. The exclusion criteria included multiple-level surgery, spondylolisthesis, or comorbid pathological conditions such as infection and compression fractures. The records of these patients were reviewed retrospectively and analyzed for demographic data, side of the surgery, duration of symptoms, follow-up period, operation time, hospital stay, and concomitant foraminal stenosis.
2. Clinical and Radiological Assessment
Three clinical and radiological subjects were evaluated: (1) clinical results, (2) compression factors of extraforaminal stenosis at L5–S1, and (3) surgical complications.
Clinical results were analyzed using a questionnaire for outcome scores (MacNab criteria, visual analogue scale [VAS] for back and leg pain, and Oswestry Disability Index [ODI]) preoperatively and 1 year postoperatively. Compression factors were analyzed using preoperative images as follows: (1) pseudoarthrosis between the TP of L5 and ala of sacrum, (2) ventral factor (disc bulging or disc bulging with osteophytes), and (3) the thickened lumbosacral and extraforaminal ligament. Pseudoarthrosis between the TP of L5 and ala was defined as contact between the TP of L5 and sacral ala on radiography (Fig. 1). Our study classified ventral factors into 2 subgroups: disc bulging with osteophytes and without osteophytes. Disc bulging with osteophytes was defined as calcification as seen in coronal CT. On the lumbar MRI, we checked the thickened lumbosacral and extraforaminal ligament at the intersection of the vertebral body, disc, TP of L5, and ala (Fig. 2). As a result of degeneration, the disc height is lowered, the gap between the TP of L5 and ala becomes narrower, and the lumbosacral and extraforaminal ligament buckles and becomes thicker. As a result, the hypertrophied lumbosacral and extraforaminal ligament creat a pathologic structure that compresses the L5 nerve root.
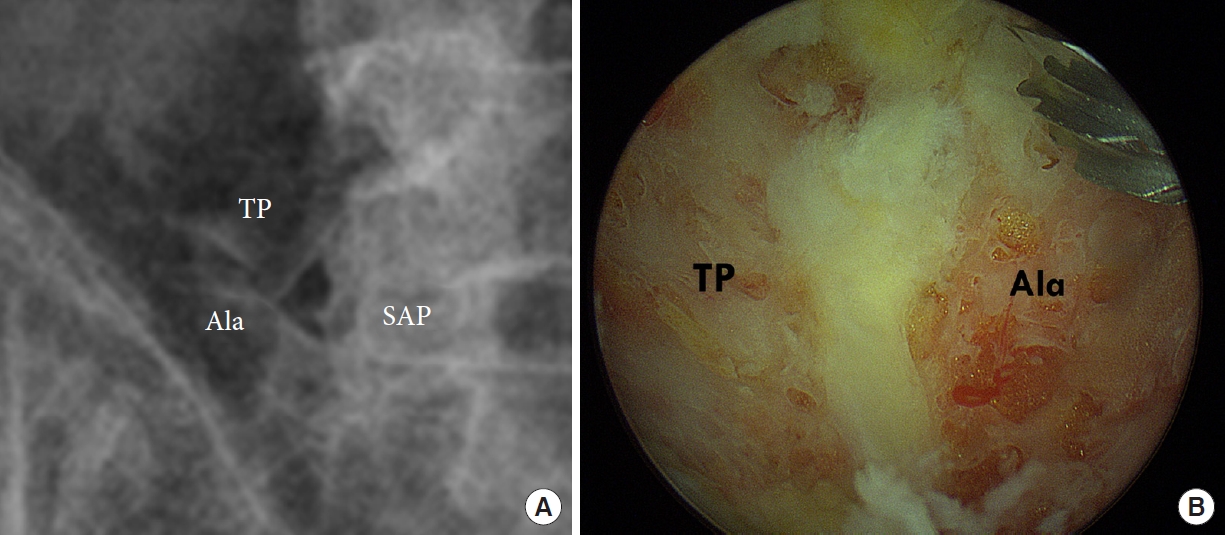
Preoperative lumbar simple x-ray (A) and intraoperative finding (B) showed contact within the transverse process of L5 and sacral ala. TP, transverse process; SAP, superior articular process.
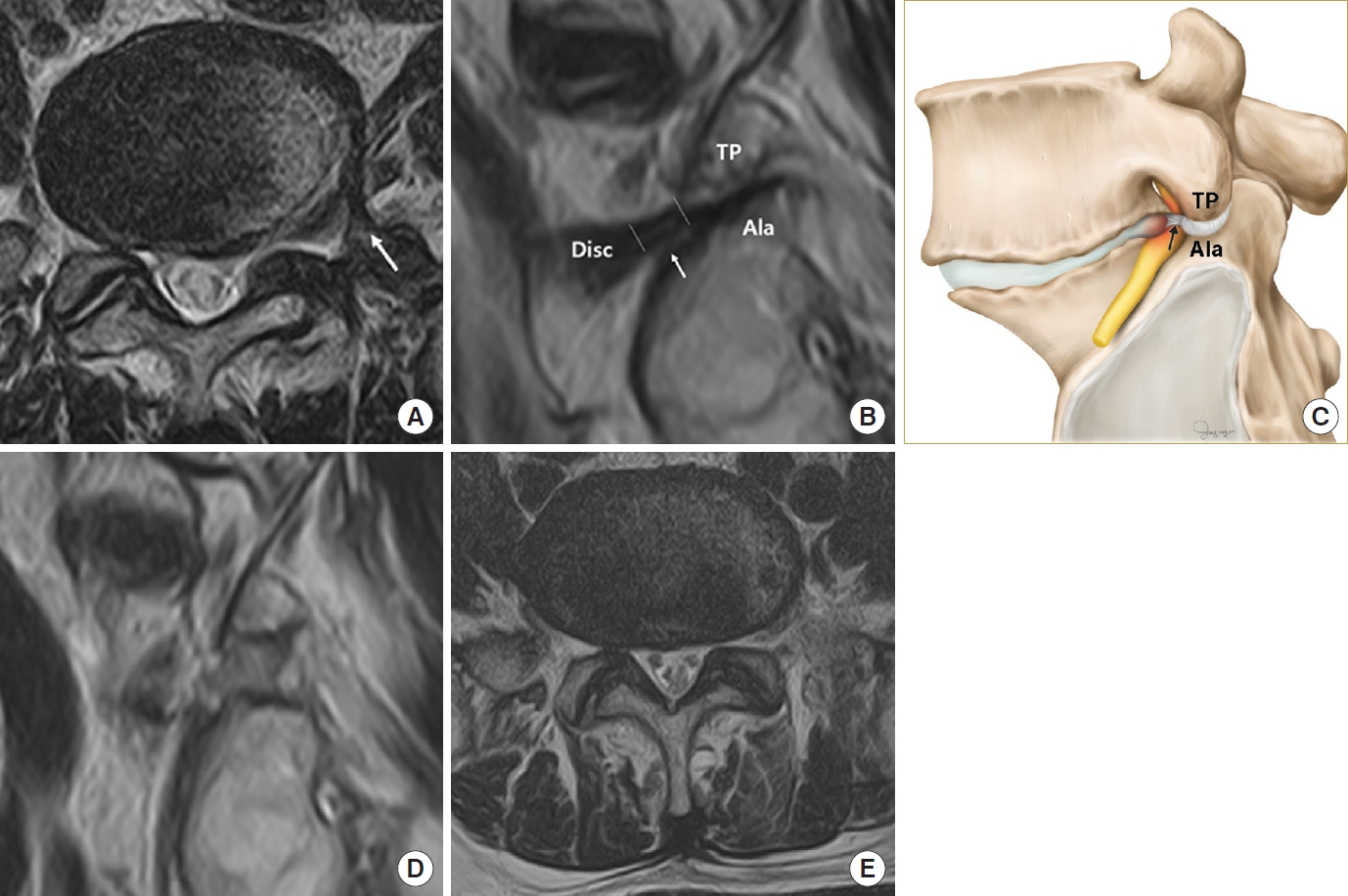
The magnetic resonance imaging (MRI) images and schematic representation of the thickened lumbosacral ligament and extraforaminal ligament. (A) MRI in the axial plane of the thickened lumbosacral and extraforaminal ligament (white arrow). (B) MRI in the sagittal plane of the thickened lumbosacral and extraforaminal ligament. The between white line indicates the thickened extraforaminal ligament. The white arrow indicates the root compressed by the thickened lumbosacral and extraforaminal ligament. (C) Schematic anatomy of the sagittal plane of the thickened lumbosacral and extraforaminal ligament (black arrow). Sagittal T2-weighted (D) and axial T2-weighted (E) postoperative MRI demonstrating removal of the thickened lumbosacral and extraforaminal ligament. TP, transverse process.
Surgical complications were analyzed according to the incidence of dural tear and nerve root injury, reoperation due to postoperative hematoma, and infection during a year.
3. Surgical Techniques
Under general anesthesia, the patient is prepared in the prone position over a radiolucent chest frame. The operator stands on the ipsilateral side of the pathology. An assistant is on the opposite side of the operator to hold the retractor (Fig. 3).

The operator is on the ipsilateral side of the pathology. An assistant is on the opposite side of the operator to hold the retractor.
1) Skin marking
The target is the superior sacral notch (the junction between the lateral aspect of the superior articular process [SAP] and sacral ala) on an anteroposterior view of the C-arm fluoroscopy. For right-handed surgeons, a left-side incision is made for a scopic portal for endoscopic viewing, and a right-side incision is made for a working portal to manipulate the surgical instruments. The incisions are approximately 3 cm apart, where the center of both incisions are placed 1 cm lateral to the target (Fig. 4). When the caudal incision has interference from the iliac crest, a caudal incision can be made medially to avoid the iliac crest.
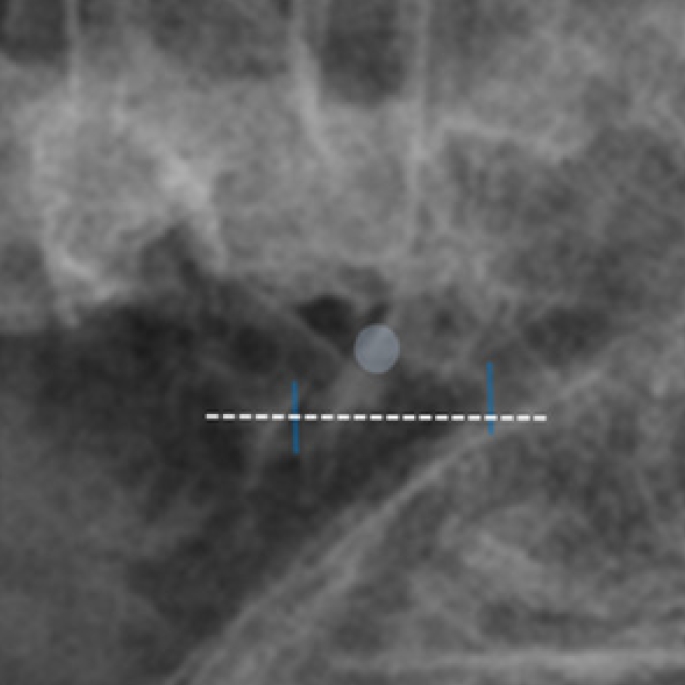
Skin incision and target point on the fluoroscopic anteroposterior view. The target (white circle) is the superior sacral notch. The 2 incisions (blue lines) are approximately 3 cm apart, where the center of the incision is made 1 cm lateral to the target (dotted line). A scope is placed through the left incision and a working tool is placed through the right incision.
2) Creating the portal and working space
Using fluoroscopic guidance, serial dilators are passed through the working portal and obturator, and a scopic sheath is advanced to the targeting point through the scopic portal. It is necessary to make a triangular position of the scopic sheath and serial dilator at a target point using the C-arm fluoroscopic view. Constant saline irrigation through 2 portals makes a clear surgical view during surgery. After confirmation of the correct positioning for both portals, a radiofrequency coagulator is coagulated to clean the soft tissues and muscles to make clear the anatomy. The surgical anatomy is first noticed as a landmark, including the inferior aspect of TP of L5, the supramedial part of the sacral ala, and the lateral aspect of the SAP of S1 (Fig. 5A).
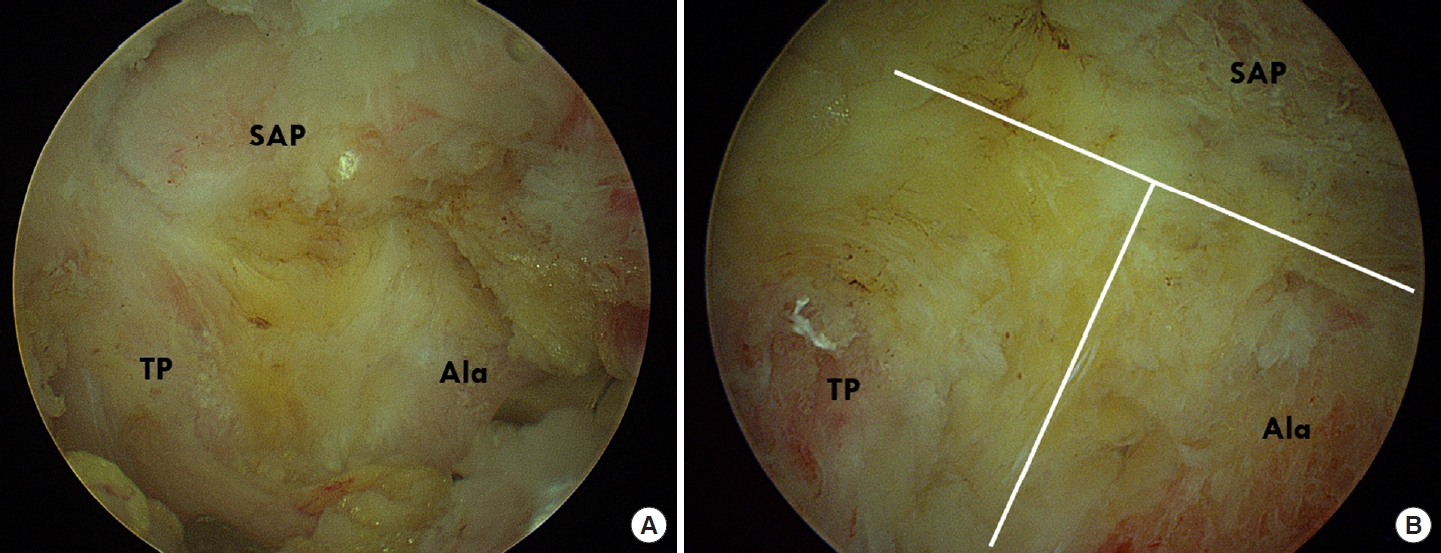
Intraoperative images showing the steps in order of exposure and decompression in a case of extraforaminal stenosis at L5–S1. (A) The surgical anatomy is first noticed as a landmark, including the inferior aspect of L5 TP, the supramedial part of the sacral ala, and the lateral aspect of the SAP of S1. (B) After drilling of these bony structures, a T-shape figure made of partially removed inferior aspect of TP of L5, the supramedial part of the sacral ala, and the lateral aspect of the SAP of S1 is identified as a landmark for decompression. TP, transverse process; SAP, superior articular process.
3) Bone working and ligament removal
The inferior aspect of TP of L5, the supramedial part of the sacral ala, and the lateral aspect of the SAP of S1 are removed with a high-speed drill and Kerrison rongeur. After drilling these bony structures, a T-shape figure made of a partially removed inferior aspect of TP of L5, the supramedial part of the sacral ala, and the lateral aspect of the SAP of S1 is identified as a landmark for decompression (Fig. 5B). This bony landmark is removed until the ligament is released from the bony structures. The lower part of the TP is removed from medial to lateral until the ligament is detached. After bone working is completed, the proximal portion of the ligament is detached from SAP using a freer elevator and Kerrison rongeur. After ligament removal, the exiting nerve root can be identified.
4) Removal of disc/osteophytes, thickened lumbosacral and extraforaminal ligament
After root confirmation, cautious and complete decompression of the root is made through the entire extraforaminal area. It is essential to completely remove the bulging disc and osteophytes that compress the exiting root ventrally. After flavectomy completion, the annulus is exposed right below the root. Then we use a radiofrequency probe to undego the annulotomy and insert straight or curved pituitary forceps to remove disc fragments or osteophytes through Kambin’s triangle (Fig. 6A). The authors propose a new surgically corridor called “Son’s corridor,” because removal of the thickened lumbosacral and extraforaminal ligament via the Kambin triangle needs much retraction of the nerve root and may result in incomplete decompression. The boundaries of Son’s corridor are the remaining TP of L5, the lumbosacral ligament, and the exiting nerve root, as depicted in Fig. 6B. Son’s corridor serves as a strategic site of the superiolateral area of the L5 exiting root, which allows access to the thickened lumbosacral and extraforaminal ligament for complete decompression of extraforaminal stenosis at L5–S1. After ventral lesions removal, decompression continues laterally towards the thickened lumbosacral and extraforaminal ligament. The L5 root is compressed by a thickened lumbosacral and extraforaminal ligament that is the lateral margin of decompression for extraforaminal stenosis at L5–S1 (Fig. 6C). In addition, Son’s corridor considers the surgical corridor for removal of osteophytes from the L5 vertebral body, with a particular focus on removing the thickened lumbosacral and extraforaminal ligament. The arthroscopy basket punch is useful when removing the thickened lumbosacral and extraforaminal ligament (Supplementary video clip 1). The endpoint of extraforaminal stenosis at L5–S1 decompression is removal of thickened lumbosacral and extraforaminal ligament compressed L5 root and free mobilization of the root, which endoscopic viewing can confirm (Fig. 6D).

Intraoperative images showing the sequential steps of removal of disc bulging/osteophytes, the thickened lumbosacral and extraforaminal ligament in a case of extraforaminal stenosis at L5–S1. (A) After completion of bone working and flavectomy, the annulus is exposed right under the root. (B) Illustrations on a extraforaminal area at L5–S1 demonstrating Son’s corridor (dotted triangle) and the thickened extraforaminal ligament (black arrow). (C) The Son’s corridor (white triangle) serves as a strategic site of superiolateral area of L5 exiting root, access to the thickened lumbosacral and extraforaminal ligament (white arrow) for complete decompression of extraforaminal stenosis at L5–S1. (D) The endpoint of extraforaminal stenosis at L5–S1 decompression is removal of the thickened lumbosacral and extraforaminal ligament compressed L5 root and free root mobilization, which can be demonstrated with endoscopic viewing. TP, transverse process; LSL, lumbosacral ligament.
4. Statistical Analysis
All statistical analyses were performed using IBM SPSS ver. 18.0 (IBM Co., Armonk, NY, USA). All data are reported as mean±standard deviation. Significant differences in preoperative and postoperative VAS scores were determined using the paired t-test. The threshold for statistical significance was set at p<0.05.
RESULTS
1. Demographic Results
Between March 2018 and February 2019, 72 patients with extraforaminal stenosis at L5–S1 were treated decompression using UBE. Thirty-seven patients were excluded because of multiple-level surgery, spondylolisthesis, or less than 1 year of follow-up. Thirty-five consecutive patients who met the inclusion criteria with extraforaminal stenosis at L5–S1 underwent decompression by UBE. The study group consisted of 16 men and 19 women with a mean age at surgery of 68.4±6.6 years. The mean duration of symptoms was 17.1 weeks. The mean follow-up was 14.9 months. The mean operative time was 63.5 minutes. The mean hospital stay was 6.9 days. Concomitant foraminal stenosis was found in 10 cases (28.6%) (Table 1).
2. Clinical Results
The MacNab criteria were “excellent” in 11 (31.4%), “good” in 17 (48.6%), “fair” in 5 (14.3%), and “poor” in 2 (5.7%) (Table 1). Out of 2 poor patients, one patient underwent lumbar fusion surgery 3 months after the operation and the other patient showed controlled symptoms with conservative treatment. The average VAS for back pain was 3.7±1.8 before surgery, and this value dropped to 2.3±0.8 at 1-year follow-up (p<0.001). There was a significant drop in average postoperative VAS for leg pain from 7.2±1.1 to 2.3±1.2 at 1-year follow-up (p<0.001). The ODI score was 61.5 points before surgery and 28.6 points at the 1-year follow-up and improved significantly after surgery (p<0.001) (Table 2).
3. Surgical Complications
No serious complications occurred for any patient, including dural tear and nerve root injury, reoperation due to postoperative hematoma, and infection within a year.
4. Compression Factors of Extraforaminal Stenosis at L5–S1
All patients had extraforaminal stenosis at L5–S1 with a decreased disc height. There were no extraforaminal lumbar disc herniations or far-lateral disc herniations at L5–S1 with a maintained disc height. Pseudoarthrosis between the TP of L5 and the sacral ala was noted in all cases (37 of 37, 100%). On preoperative imaging, pure disc bulging was noted in 12 patients (34.3%), and the presence of osteophytes with disc bulging was noted in 23 patients (65.7%). The thickened lumbosacral and extraforaminal ligament was found in 19 cases (51.4%) (Table 3).
DISCUSSION
Traditional microscopic foraminotomy via the Wiltse paraspinal approach has been seen as an effective surgical treatment in treating FOS, but the procedure has drawbacks. UBE purports to mitigate those drawbacks, as it can allow for reaching deeper into the foraminal region less invasively [6-9,16]. Our preliminary results on UBE for extraforaminal stenosis at L5–S1 showed that (1) extraforaminal stenosis at L5–S1 has 3 compression factors: (i) pseudoarthrosis between the TP of L5 and sacral ala, (ii) osteophytes and disc bulging, and (iii) the thickened lumbosacral and extraforaminal ligament; (2) UBE has good clinical outcomes, including lower postoperative VAS of back and leg and ODI scores; and (3) no patients in our preliminary study had complications arising from the technique.
As mentioned, surgical access to the L5–S1 extraforaminal area is difficult [7,16,17]. Further, a prominent iliac crest sometimes may prevent a surgical corridor through the paraspinal Wiltse approach [17,18]. Perhaps as a consequence, several researchers have reported poor outcomes after microscopic foraminotomy for extraforaminal stenosis at L5–S1 [10,11]. Many researchers have reported concomitant foraminal stenosis along with inadequate decompression as predictors of poor outcomes of microscopic foraminotomy for extraforaminal stenosis at L5–S1 [7,19]. Intra-and extraforaminal space decompression in such patients can only happen with sufficient removal of the facet joint with microscopic foraminotomy, which can result in further degeneration with secondary instability and chronic back pain [7].
UBE has several advantages over microscopic foraminotomy for extraforaminal stenosis at L5–S1. First, UBE enables the surgeon to reach a deep site quickly and provides a well-illuminated surgical field with proper magnification [12,20-22]. Second, the technique avoids unneeded invasion of surrounding tissues and preserves the facet joint, which consequently provides advantages for patients, such as reduced postoperative back pain and early resumption of routine activities [12]. Third, necessary bone removal can be assessed in cases of concomitant foraminal stenosis. Decompressing an intraforaminal lesion has been considered difficult with microscopic surgery in many cases [20]. In contrast, UBE allows for specific assessment of the needed degree of facet resection, which minimizes the risk for residual stenosis or instability [20].
We identified 3 compression factors in the present study: (1) pseudoarthrosis between the TP and ala, (2) osteophytes and disc bulging, and (3) the thickened lumbosacral and extraforaminal ligament. First, Wiltse et al. [1] demonstrated a extraforaminal stenosis at L5–S1 characterized by L5 nerve entrapment caused by a descending TP of L5 and the superior portion of the medial sacral ala. In our study, the affected roots were compressed within the TP and the sacral ala at the medial aspect of articulation in all cases. Second, another cause of extraforaminal stenosis at L5–S1 was osteophytes/disc bulging of the lateral margin of the vertebral body on the L5 and S1 vertebrae. Osteophytes/disc bulging were identified and seen to compress the L5 nerve root in recent studies [23,24]. We believe that removal of disc bulging or osteophytes is important in extraforaminal stenosis at L5–S1, because if these compression factors are not removed, the symptoms may remain or the root may be compressed over time. Third, Nathan et al. [24] showed in a cadaveric study that the lumbosacral ligament, where the L5 spinal nerve passes, could be entrapped. Furthermore, several authors have indicated that the transforaminal or extraforaminal ligament, which is attached between the facet joints or TP and the intervertebral disc, could compress the lumbar nerve root in the extraforaminal area [25,26]. Degenerative changes of intervertebral discs, lumbosacral ligament, and extraforaminal ligament make a pathologic structure, which could compress the L5 nerve root at the lumbosacral tunnel. To summarize, the lumbosacral and extraforaminal ligaments have no compression effect on the nerve root in normal circumstances, but with degeneration, the thickened lumbosacral and extraforaminal ligaments may play a role in causing radiculopathy. In our study, in cases where the thickened lumbosacral and extraforaminal ligaments were observed on MRI, it was shown that these pathologic bands compressed the superolateral aspect of the L5 nerve root under intraoperative endoscopic viewing (Fig. 6C). Earlier, we described the usefulness of “Son’s corridor” for complete removal of the thickened lumbosacral and extraforaminal ligaments. Detailed understanding of this strategic triangle can provide for precise and safe removal of the thickened lumbosacral and extraforaminal ligaments without much retraction of the nerve root.
Retroperitoneal fluid collection is one of the most fatal complications in the paraspinal approach using the UBE technique [12]. There is a possibility of fluid accumulating into the retroperitoneal space if fluid out is poor [12]. The barrier between the extraforaminal area and retroperitoneal area could be damaged when the thickened lumbosacral and extraforaminal ligaments are removed, which may result in retroperitoneal fluid collection [12]. Therefore, special care should be taken to not proceed ventrally farther than where the psoas muscle is. That is why we need to pay more attention to fluid coming out in the extraforaminal stenosis at L5–S1 using the UBE technique.
One limitation of the current study is that this was retrospective with a small sample size and the focus was on surgical techniques. Due its retrospective nature, there is a potential for selection bias within the patient cohorts. Detailed prospective trials using a larger cohort that compares UBE with other techniques, such as conventional surgery, are needed for a deeper analysis of this topic.
CONCLUSION
In the current study, good surgical outcomes after UBE decompression were achieved without complications for patients with extraforaminal stenosis at L5–S1. UBE decompression can avoid certain technical difficulties associated with conventional procedures for extraforaminal stenosis at L5–S1. It is necessary to achieve an accurate diagnosis based on MRI, and complete decompression based on analyzing the extraforaminal anatomy is necessary. It is possible that the UBE technique can replace conventional surgical procedures to become the standard operating procedure for extraforaminal stenosis at L5–S1.
Notes
The authors have nothing to disclose.
SUPPLEMENTARY MATERIALS
Supplementary video clip 1 can be found via https://doi.org/10.14245/ns.2142146.073.