Current Status of the Use of Salvaged Blood in Metastatic Spine Tumour Surgery
Article information
Abstract
To review the current status of salvaged blood transfusion (SBT) in metastatic spine tumour surgery (MSTS), with regard to its safety and efficacy, contraindications, and adverse effects. We also aimed to establish that the safety and adverse event profile of SBT is comparable and at least equal to that of allogeneic blood transfusion. MEDLINE and Scopus were used to search for relevant articles, based on keywords such as “cancer surgery,” “salvaged blood,” and “circulating tumor cells.” We found 159 articles, of which 55 were relevant; 20 of those were excluded because they used other blood conservation techniques in addition to cell salvage. Five articles were manually selected from reference lists. In total, 40 articles were reviewed. There is sufficient evidence of the clinical safety of using salvaged blood in oncological surgery. SBT decreases the risk of postoperative infections and tumour recurrence. However, there are some limitations regarding its clinical applications, as it cannot be employed in cases of sepsis. In this review, we established that earlier studies supported the use of salvaged blood from a cell saver in conjunction with a leukocyte depletion filter (LDF). Furthermore, we highlight the recent emergence of sufficient evidence supporting the use of intraoperative cell salvage without an LDF in MSTS.
INTRODUCTION
Cancer is one of the leading causes of morbidity in both developed and developing countries. Global cancer incidence was estimated to be 14.1 million in 2012 [1], and is expected to rise to 22 million per year, by 2030 [2]. Advancements in surgical techniques and adjuvant treatment for cancer have resulted in improved survival rates of cancer patients. This has led to an increasing prevalence of metastatic cancer. The most common sites for metastasis are the lung and liver, followed by the skeleton [3]. The spine is the most common site for skeletal metastases [4-6], with almost 70% of cancer patients developing spinal metastases; 10% of these patients present with cord compression [5].
Surgery is currently the mainstay of treatment for metastatic spine disease, when a patient’s life expectancy is more than 3 months [4,6,7]. Metastatic spine tumour surgery (MSTS) is indicated in patients experiencing intractable pain, diminished neurological status, or at risk for impending cord compression due to spinal instability [4]. The expected duration of survival is short (< 1–2 years) in many cases [4]; however, the decision for MSTS is made based on the patients’ quality of life [4]. Even terminally ill cancer patients can undergo MSTS for palliative reasons. Survival after MSTS is heavily dependent on the primary tumour type [8].
Although surgical techniques have advanced especially due to the advent of minimally invasive surgeries, significant blood loss during MSTS remains a major problem [9,10]. The average blood loss in open/conventional spine surgery for vertebral metastases was found to be 1,418 mL (713.3 to 3,120 mL), while that in minimally invasive spinal metastatic surgery was 745 mL (184 to 1,320 mL) [11].
The current mainstay of blood replenishment in MSTS is allogeneic blood transfusion (ABT) [12]. Despite the safety of allogeneic blood having improved, the immune-mediated complications of transfusion still exist, i.e., transfusion related acute lung injury [13,14]. Similarly, such immune-mediated phenomenon leads to ABT being an independent predictor of post-operative infections in spine surgery [8,15,16]. It was also pointed out that perioperative ABT together with subsequent postoperative infectious complications are associated with a poorer prognosis [8,17].
In order to reduce ABT and its associated risks, patient blood management (PBM) has progressed to secure a vital position in major surgeries [18]. PBM is an evidence-based patient-tailored approach aimed at reducing the need for ABT [18]. This is achieved by managing anaemia preoperatively, as well as by using perioperative blood conservation techniques such as administering agents which diminish blood loss or promote red blood cell production, reinfusing patients’ own blood (e.g., cell salvage), and surgical haemostasis [18]. Among these, intraoperative cell salvage (IOCS), also referred to as salvaged blood, has been used and studied extensively in the surgical setting. Salvaged blood transfusion (SBT) has been proven to reduce the need for ABT in nononcological elective surgeries by previous randomized [19,20], nonrandomized [21] and retrospective studies [22]. In spite of SBT being used routinely in trauma, cardiac, and major orthopedic surgeries [23], it has not found wide applications in oncological surgeries [23,24]. Concern exists that transfusion of blood collected by IOCS might reinfuse tumour cells and cause subsequent metastases [23-27].
We have previously reported on the safety of the blood salvaged during MSTS [23,28,29]. Our analysis supports the hypothesis that the number of tumour cells in salvaged blood, if any, is significantly less than those in the patients’ circulation. Furthermore, when these tumour cells are passed through the IOCS system, they are likely to lose their viability and hence will not promote subsequent dissemination [30]. Nevertheless, due to the lack of clinical data addressing the concerns of tumour dissemination, oncological surgeons remain reluctant to employ IOCS.
With this background, we reviewed the literature to scrutinize the current status of SBT in oncologic surgeries, with regard to its safety and efficacy, contraindications, and adverse effects. In this review, we also aim to establish that the safety and adverse event profile of SBT is comparable, if not superior to ABT.
MATERIALS AND METHODS
We used MEDLINE and Scopus to search for relevant publications over the past 22 years (January 1, 1986–May 31, 2018). We adopted a search strategy that commenced from the year 1986, since that was when salvaged blood in oncologic surgeries was strongly contraindicated in a report by American Medical Council [31]. We augmented the outcomes by searching for associated articles from the years preceding our selected study period. Pertinent articles were selected using combinations of the following search terms: “cancer surgery,” “metastatic cancer surgery,” “intraoperative cell salvage,” “autologous salvaged blood,” “circulating tumor cells,” and “metastatic spine tumour surgery.” We identified additional literature by manually searching reference lists of the identified studies. Only publications in the English language have been included in the review.
We directed our search towards articles which addressed the use of IOCS in oncologic surgeries. We omitted case reports and those studies where IOCS was used in benign oncologic surgeries. Our preliminary search revealed 159 articles; 104 articles were eliminated based on their titles and abstracts. From the 55 articles that were possibly relevant, 20 were excluded based on considerations such as the use of other blood conservation techniques in conjunction with cell salvage and very small case series with <5 patients. 5 other articles were identified by means of searching contacts and reference lists. Finally, a total of 40 articles were reviewed.
DISCUSSION
1. Salvaged Blood in Oncologic Surgical Procedures
The use of SBT in oncological surgeries has been debatable. This emanated from the previously mentioned American Medical Council report in 1986, which specified that autologous SBT is contraindicated in oncologic surgeries [31]. This concern surfaced after a single case report in 1975 where a 52-year-old patient who underwent a pneumonectomy was found to have malignant cells in salvaged blood investigated using the cell block technique [32]. The controversy regarding malignant cells being reinfused into patients during SBT arose due to the presumption that malignant cells are spilled into the operative field during manipulation and resection of the tumour [33]. However, it has since been demonstrated that oncological patients’ blood contains malignant cells which are shed from the tumour [34]. These are termed ‘circulating tumour cells’ (CTCs).
The concerns regarding reinfusion of malignant cells were readdressed using in vitro studies by Dale et al. [35] and Lane [36] in 1988 and 1989, respectively. They proposed that the apparatus used for autologous transfusion in the 1980s would preserve the tumour cells, resulting in reinfusion of malignant cells into patients. In 1995, Hansen et al. [37] conducted a study on 61 patients who underwent surgery for malignant tumours of urological, orthopedic, gynaecological, abdominal or head & neck origin. These 61 patients were compared with 15 other patients who were operated on for benign diseases. As a result of this comparative study, which had an in vitro experimental design, it was concluded that the CTCs shed during surgery were tumorigenic, i.e., they were capable of invasion and proliferation. Based on these observations, it was deduced that intraoperative SBT in oncological surgeries is contraindicated, as it would lead to dissemination of malignant cells, resulting in metastasis.
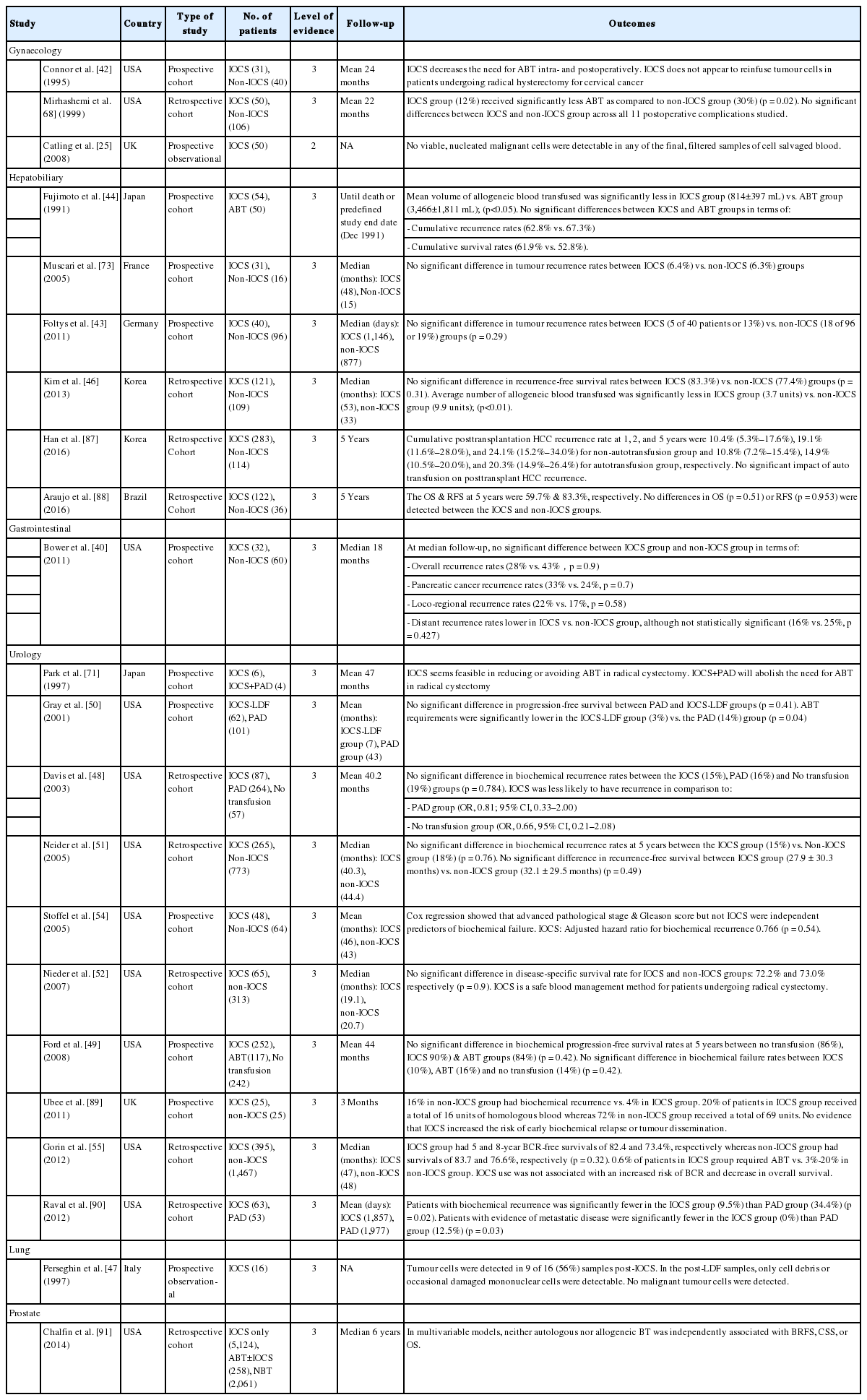
On the other hand, there were concurrent studies which reported that SBT did not apparently increase the risk of clinically detectable metastases [38,39]. Subsequently, the feasibility of using salvaged blood in surgery for malignancies of gastrointestinal [40], gynaecological [41,42], hepatobiliary [40,43-46], lung [47] and urological [39,48-55] origins, has been broadly studied (Table 1). These studies have successfully provided evidence on the safety of SBT with regards to disease progression in terms of increase in size of existing lesions and/or the appearance of new metastatic lesions. Even in patients with such disease progression, there is no evidence that it was due to tumour cells present in salvaged blood, since it could also be due to the CTCs already present in the patients’ blood [29,56].
2. CTCs in Metastatic Cancer Patients
The metastatic process heavily relies on CTCs, the concentration of which is largely influenced by tumor type and stage of disease [57]. Allan and Keeney [34] approximated that the peripheral blood of patients with metastatic cancer can contain 1 CTC per 105 to 107 mononuclear cells. Allard et al. [57] pointed out that the number of CTCs in metastatic cancer patients can be as high as 23,618 per 7.5 mL of blood. There is ample evidence to demonstrate that patients who have a CTC count of greater than 5 per 7.5 mL of blood, experience poor progression-free and overall survival compared to those who have less than 5 [57-62].
3. Fate of Tumour Cells in Circulation of Metastatic Cancer Patients
The CTCs in patients’ blood form the bridge between primary tumour and metastasis. CTCs are vulnerable to a variety of host defenses [63]. For instance, CTCs are actively removed from circulation by being trapped within the bone marrow or capillary bed, where some may enter into a phase of cellular apoptosis [64]. Terraube et al. [65] demonstrated that the Von Williebrand Factor present in the bloodstream can induce the tumour cells to enter apoptosis, thereby assuming a protective role in tumour metastases. Other CTCs may remain quiescent but later may evolve into clinically evident metastasis, although this is infrequent [64]. In addition, current literature indicates that the reticular endothelial system eliminates those CTCs which either do not complete the entire process of metastasis, or those which are removed from the circulation at the capillary bed [64]. Hence, only a mere minority of the CTCs successfully progress into metastatic lesions [66].
4. Fate of Tumour Cells in Salvaged Blood
During the cell salvage process, Catling et al. [25] observed that only fragmented cytoplasmic debris were detectable in pre- and post-filtration blood samples obtained from the reservoir. Similar results were observed in one of the recent studies by Kumar et al. [23], wherein the majority of cell blocks created from salvaged and filtered blood exhibited cytoplasmic debris which were devoid of any viable nuclei. Karczewski et al. [67] demonstrated that 62% of tumour cells in the blood underwent lethal trauma, while all the remaining tumour cells displayed morphological changes, after being processed with the IOCS device. In another study by Kumar et al. [30], it was established that the tumour cells which pass through the IOCS device, whether with or without leukocyte depletion filter (LDF), are morphologically altered and become nonviable, i.e., they lose the ability to form new metastatic deposits.
5. Salvaged Blood Is Appropriate for Oncologic Surgeries
There is ample evidence in literature about the clinical safety of salvaged blood used in oncological surgeries including gastrointestinal [40], gynaecological [41,42,68], hepatobiliary [40,44,46,69] and urological [48,49,51,52,55,70-72] surgeries (Table 1). Although patients who underwent SBT required significantly lesser amounts of allogeneic blood, their survival rates [44,51,55,70] and disease progression remained comparable to those who did not receive SBT [42,46,71,73]. Patients who underwent SBT had lower or similar rates of recurrence, as compared to the control cohort [48,51,55,72].
6. Advantages of IOCS
Autologous transfusion decreases the risk of postoperative infections and tumour recurrence as a result of transfusion-related immunomodulation [74]. In fact, SBT even reverses ‘the immunosuppression associated with surgical trauma and blood loss.’ [75,76] Furthermore, SBT reduces the amount of allogeneic blood required [19,21], thereby diminishing or possibly avoiding the risks associated with ABT [23,28]. Studies have also shown that RBCs in salvaged blood exhibit a higher mean viability [77] & deformability [78] than those in allogeneic blood, which translates to a longer survival of the transfused RBCs [77,79,80].
Although some studies have stated that oncologic surgery patients who received SBT, experienced a longer cancer-free survival as compared to those managed with ABT [43,69], these publications have a nonrandomized study design; hence the quality of evidence reported is debatable. There have been no randomized controlled trials, on the use of salvaged blood in oncologic surgery till date [24,81]. Large well-designed randomized controlled trials are required in order to alleviate confounding factors; however, such trials have practical and ethical constraints, especially for reinfusion studies in patients with malignancy [81]. Despite these restraints, there are ongoing attempts recently, to carry out welldesigned randomized control studies [82].
7. IOCS – Disadvantages & Contraindications
In spite of the above-mentioned benefits of IOCS, there remain some limitations regarding its clinical application. IOCS cannot be employed in patients with definite sepsis, with the exception of fatally hemorrhagic conditions where adequate quantities of allogeneic blood are unavailable [83]. Another contraindication for IOCS is the presence of contaminants and fluids (including sterile water, hydrogen peroxide, alcohol, or any hypotonic solution) which can possibly trigger lysis of RBCs [84,85]. The presence of pharmacologic agents in blood, which can potentially enhance clotting; or blood contaminated with urine, bone chips, methyl methacrylate, fat and amniotic fluid are also contraindications [56]. Nevertheless, washing and post-processing of the harvested blood can reduce such contaminants [56]. It is advised that the use of IOCS be assessed on a case-by-case basis, since there are no publications consolidating the various possible relative contraindications.
Complications of IOCS comprise of electrolyte and pH disturbances, air or fluid embolism, dilutional coagulopathy, and introduction of foreign bodies, microbes or tumour cells [74]. Multiorgan failure and consumption coagulopathy, collectively termed as “Salvaged blood syndrome,” has also been described [27,74].
8. Present Safety Standards of Salvaged Blood in MSTS
It is evident that SBT has been widely used in other oncological & metastatic surgeries [40-42,44,46,48,49,51,52,55,68-72], thereby re-enforcing its safety in metastatic disease of the spine (Table 1); however in MSTS, it is still in its infancy. The use of salvaged blood in MSTS has been shown to reduce complications that are usually associated with ABT [23,28,74] without increasing disease progression or reducing life expectancy [86]. This implies that SBT is safe in MSTS.
CONCLUSION
The evidence reveals that the current status of using salvaged blood in MSTS is gradually evolving. There is ample evidence in literature, to support the use of IOCS in oncologic surgeries, including metastatic skeletal tumour surgeries. It is established that the salvaged blood can be used from the cell saver in conjunction with LDF. There is also sufficient evidence forthcoming in this article supporting the use of IOCS without LDF in MSTS [30].
Notes
The authors have nothing to disclose.