Posterior Endoscopic Cervical Decompression: Review and Technical Note
Article information
Abstract
Endoscopic spine surgery for the treatment of degenerative spinal diseases from lumbar to cervical spine has accelerated over the past 2 decades. Posterior endoscopic cervical discectomy (PECD) has been described as a safe, effective, and minimally invasive procedure for cervical radiculopathy or even part of the myelopathy. This procedure also has been validated with comparable outcomes to open and microscopic surgery. Radiculopathy due to foraminal disc herniation or foraminal stenosis should be the optimum indications of this procedure. Intraoperative 3-dimensional navigation can help surgeons to get quick and great quality guidance for endoscopic surgeons. In this review, we will focus on the technical details and evidence-based results of PECD which is a promising procedure for cervical radiculopathy with the advantages of a minimally invasive method.
INTRODUCTION
Posterior cervical discectomy and foraminotomy have been used for many years and achieved good clinical results [1-3]. This technique can be used for nerve root decompression without segmental fixation, and then remain the mobility of the operative segment. However, due to the abundant paravertebral muscle tissue of the cervical spine, the exposure of the surgical field often needs a long surgical incision and extensive soft tissue stripping. Although the part of decompression is small, the long incision still brings great trauma and long postoperative recovery time. Therefore, this technology has not been widely used compare with anterior approach. Endoscopic surgery has been developed rapidly in the past 30 years. With its minimally invasive surgical approach and magnification and cold light source system, an extensive paravertebral soft tissue stripping in open surgery is not needed in the minimally invasive approach [4].
After the successfully applying in lumbar spine, the use of endoscopic techniques continued to expand to treat a wider range of pathology, such as the cervical spine [5,6]. Posterior endoscopic cervical discectomy (PECD) and foraminotomy for decompression of cervical nerve roots is a well-established minimally invasive technique for cervical radiculopathy with comparable outcomes to traditional open surgeries. Compared with open approach, this minimally invasive approach can decrease postoperative pain, blood loss, muscle spasm, and dysfunction [2,3,6]. The primary indications of this technique were posterolateral soft disc herniations or foraminal stenosis [7]. With the high definition endoscope and cold light source, a magnified view of surgery field can be achieved through a small working tube which usually set by a series of dilation procedure. The epidural bleeding can be well controlled by irrigation during the surgery [5]. Unlike the open surgery, the length of incision of PECD usually no more than 1 cm and without extensive stripping of the paraspinal musculature. Surgeons can establish the approach using a sequential dilation system onto the facet joint through the incision and a working tube is then placed on site. Then, a laminoforaminotomy can be applied by a high-speed bur under the monitor of endoscope. The decompression of nerve root is then completed under the endoscope. With the minimally invasive approach, the approach morbidity is reduced, recovery and hospital stay is shortened [5,6].
However, only a few studies of endoscopic procedures have been reported due to multiple anatomical factors and its “high-risk” profile of cervical spine in the past decade. Identification of anatomic structure is difficult by the limited visibility provided by an endoscope for surgeons. Moreover, sufficient decompression without excessive facet joint resection also a challenge for surgeons. The high-risk complication such as spinal cord injury is still the severe complication of PECD when compared with lumbar surgery.
Under the image guidance systems, surgeons can get greater accuracy and high efficiency in most of spine surgery [8,9]. However, for lower-level or obese patients, the x-ray cannot provide an available reference for the working tube placement. By the image-guided spinal surgery, surgical instruments can be tracked in 3-dimensional (3D) space which allows surgeons to navigate the spinal anatomy using a preoperative or intraoperative computer tomography (CT) scan data. By a navigation system, surgeons can get a 3D guidance of the spine structure and proximity of neurovascular structures in the surgery field. Previous studies have described successful navigation assisted decompression and fixation of the spine with the shorter operation time, high screw placement accuracy, and satisfactory decompression compare with traditional x-ray monitored surgery [8,10,11].
In this review, we will focus on the details of PECD technique. The primary goals are sufficient decompression of nerve root and minimal facet joint resection in the process.
The protocol follows the guidelines of Xinqiao Hospital’s human research ethics committee.
INDICATIONS AND CONTRAINDICATIONS
PECD is indicated in patients with unilateral radiculopathy of single level, often caused by foraminal soft disc herniation or bone spur. Other pathologies such as spinal canal due to ligament flavum hypertrophy or ossification, facet joint cyst [12], and ligament flavum cyst are also indications for this procedure [1,3,13].
The exclusion criteria are the presence of segmental instability, kyphotic deformity, and multiple-level pathology. Any associated infection, tumor, and fracture in the region of the cervical segment are also considered as the contraindications. In patients with myelopathy caused by anterior disc herniation and/or calcification is also the current contraindication of this technique since it can exacerbate these conditions, even more, has a disastrous consequences due to the risk of spinal cord injury. Although there were a few studies reported on outcomes following full-endoscopic decompression of patients with mild cervical myelopathy by modified posterior endoscopic techniques, it still needs a further prospective, randomized, and controlled study to confirm the efficacy and safety of the index procedure.
SURGICAL PROCEDURES
1. Preoperative Preparation
Before surgery, the authors perform anteroposterior, lateral, and flexion/extension imaging of cervical spine to confirm there is no dynamic instability or deformity. Magnetic resonance imaging should be carefully reviewed to assess for position and size of herniated disc, and degree of foraminal stenosis. CT scan is performed for the detailed analysis of foraminal zone such as the degree of stenosis, size, and location of the herniated disc as well as the amount of bony osteophytes. The contralateral facet joint should also be reviewed to exclude pre-existing pathology. This provides a reference of the size of bone resection of facet joint while preserving segmental stability.
2. Anesthesia and Skin Incision
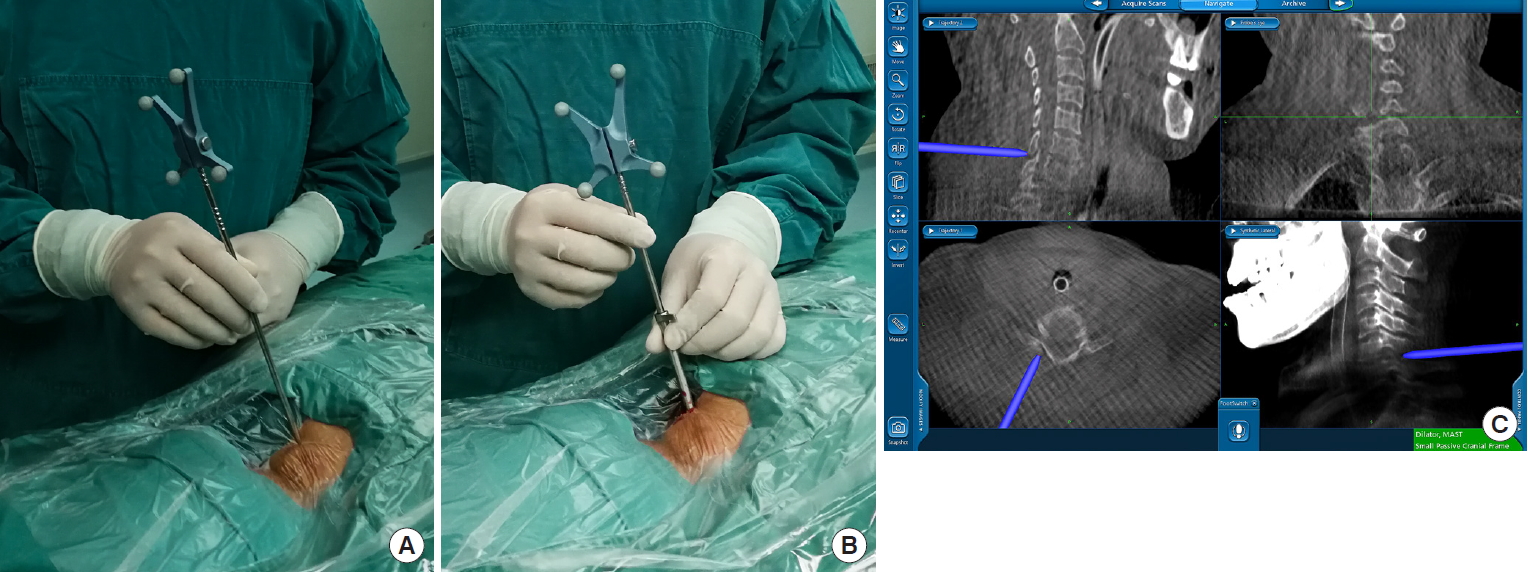
The procedure is performed under general anesthesia, a neuromonitoring should be prepared. The patient is placed in the prone position on a radiolucent operating table with the cervical spine delordosated and the head is fixed with Mayfield clamps. The arms are positioned toward caudal on the body and immobilized with adhesive tape. Somatosensory-evoked potentials and myotomal electromyography are monitored. In lower level or obese patients, the O-arm and computer-assisted navigation system (O-arm Surgical Imaging System and Stealth-Station S7, Medtronic, Minneapolis, MN, USA) are used. The reference frame is fixed on the extension of Mayfield head-rest system after preparation and draping of the surgical field. Then, the O-arm is used to take a 3D image with medium dose of irradiation. The reference frame can be fixed on the Mayfield system instead of the bone part such as skull or spinous process. Imaging reconstructions of the cervical spine are generated and ready for navigation. A single dose of antibiotics will be used before skin incision. Skin marking is drawn 1 cm away from midline of the cervical spine. A less than 1-cm skin incision is made vertically toward the “V” point of facet joint which is the intersection of inferior and superior lamina (Fig. 1A). The endoscopic procedure is performed with about 150 cmH2O of irrigation fluid pressure. The height of fluid should be adjusted to provide optimal clarity of endoscopic view but no more than 200 cmH2O. An irrigation pump also can be used to achieve this pressure.
3. Insertion of Endoscope
With a one-step dilatation, the working cannel, endoscope, and instruments are directly placed over the surface of the facet joint place through the skin incision. Blunt insertion of a dilator can be monitored by navigation system onto the facet joint (Fig. 1B, C). The 7.5-mm working tube was inserted via the dilator before the dilator was removed. A sharp guidewire should be avoided in this step for safety reason. The reduction of the incisions size and the direct placement of working cannel without any muscle dissection can result in a rapid postoperative recovery.
4. Surgical Procedure
After insertion of the 6.3-mm endoscope, further operation is performed under visual control and continuous irrigation. After cleaning the soft tissue on the joint by a bipolar radiofrequency and forceps, the margin of the superior laminar, inferior laminar and V-point (include inferior margin of the cephalic lamina, the medial junction of the inferior and superior facet joints, and the superior margin of the caudal lamina) are identified (Fig. 2A). A skill of this part is to feel the bone by working tube before endoscope insertion, which can help to shorten the time of soft tissue exposure and hemostasis control under the endoscope. A tender stripping of soft tissue over the facet joint by working tube also can be used. A mark can be made with the burr on the bone of the V-point. Then the 3-mm diamond burr is moved cranially first to remove the cephalic lamina which is on the top layer of joint. Then, a keyhole foraminotomy is performed start from the lamina-facet junction using 3-mm diamond burr (Fig. 2B). As the resection of facet joint is before decompress of nerve root, we use a 3.0-mm burr which helps in measurement of the size of facet joint resection which usually less the 10 mm in diameter. Depending on the size and location of herniation and surgical level, the hole of foraminotomy can be extended toward lateral or craniocaudal side. The facet joint will be dissected no more than 50% until the lateral margin of ligamentum flavum is showed. There are studies showed that removing more than 50% of the cervical facet joint can result in greater risk of further instability [14,15]. For safety, we usually leave a very thin or even semitransparent cortical bone of the lamina by diamond burr and then enlarge the hole by a endoscopic punch. Then, a very thin ligamentum flavum will be resected without violating the prominent venous plexus around the nerve root. Carefully identification of the lateral edge of dura and branching of the nerve root is important for spinal cord safety. Coagulation of venous plexus by bipolar radiofrequency should be performed for exposure of the desired nerve root and herniation (Fig. 3A). The ruptured fragments are removed by a 45° micropituitary forceps (Fig. 3B).
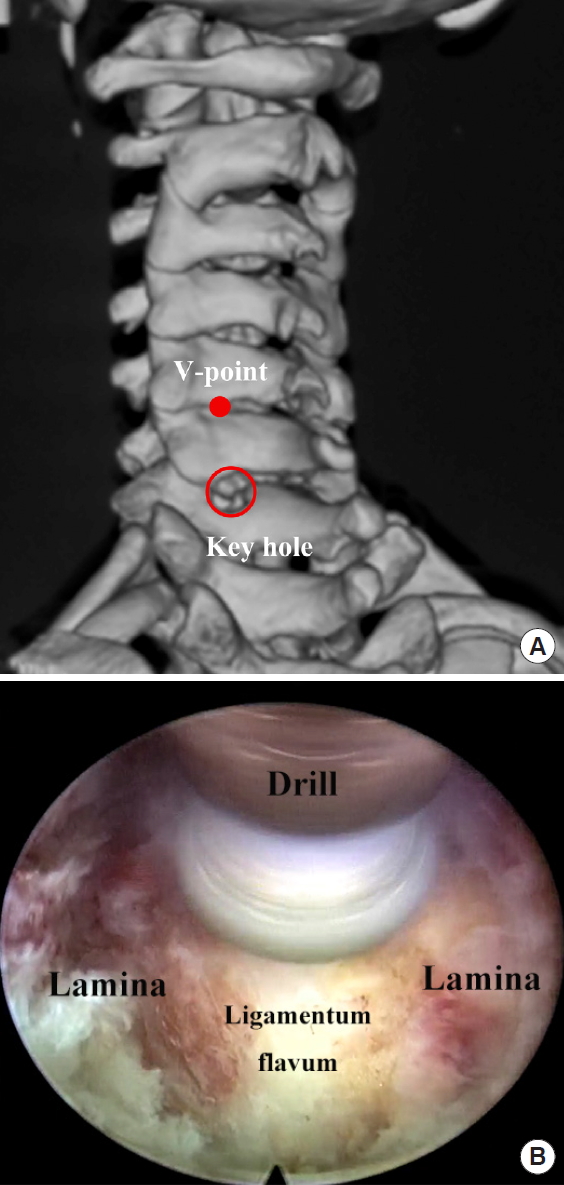
The demonstration of a keyhole procedure. (A) A keyhole foraminotomy is performed start from the lamina-facet junction using 3-mm diamond burr. (B) Postoperative view of a 3-dimensional reconstruction of the V-point and keyhole decompression field which preserved most of the facet joint.
5. Final Checking Point
Both the shoulder and axilla of nerve root should be visualized and palpated, the ventral side of both never root and spinal cord also should be decompressed well. If necessary, further discectomy or osteophyte resection can be performed with micropituitary forceps and high-speed bur. The incision of disc can be treated with radiofrequency coagulator shrinkage. There is no drain needed for this procedure. Skin is then closed.
DISCUSSION
Posterior cervical foraminotomy (PCF) is a well-established procedure for treating radiculopathy caused by foraminal soft disc herniation and/or foraminal stenosis. Compare with anterior cervical discectomy and fusion (ACDF), the gold standard surgery for cervical nerve compression, PCF for unilateral cervical radiculopathy has demonstrated equivalent clinical results based on improvements in radicular arm pain and neck pain [16]. Additional advantages of PCF include lower risk of the esophagus and laryngeal nerve injury, and motion preservation when compare with the ACDF [5]. However, there is significantly more muscle dissection for traditional open PCF due to the anatomic features.
Recently, PECD has been described in previous studies and has shown comparable outcomes to the open PCF approach with more rapid functional recovery, shorter inpatient stays, and earlier return to work. By a one-step blunt dilatation, the working tube, endoscope, and instruments were directly placed over the site of the lamina and facet joint through a < 10-mm skin incision. The reduction of the incisions and muscle dissection could result in reduction in blood loss, muscle disruption, and bony removal. Compared to traditional open surgery or microscopic surgery, the endoscopic technique can get a magnified view and limited bleeding, which under cold light system and continuous irrigation system [5]. Unlike the retractor or tubular assisted microscopic decompression, the endoscopic technique can get a clear vision and limited bleeding, due to a continuous fluid flow or an irrigation system. Moreover, by a radiofrequent electrode, the bleeding could be reduced efficiently. Bone resection is necessary in all cases, especially in foraminal stenosis. The range of bone resection always depended on the size and site of the pathology. The 3-mm-high speed drill can be an endoscopic marker for the lamina-window which usually less than 10 mm. With less exposure and more safety when compared to open approach, this minimally invasive approach is especially beneficial for obese and elderly patients.
The initial indication of PECD is limited to unilateral radiculopathy caused by foraminal soft disc herniation or bony stenosis. Through the experience gained in increasing cases, the use of endoscopic surgical techniques continues to expand to treat a wider range of compressive pathology in the cervical. Endoscopic laminoplasty has been reported with favorable outcomes with less perioperative complications for cervical myelopathy [17]. Through a more lateral and caudal posterior access which oriented from lateral mass of lower vertebra, with drilling of part bony portions of the pedicle and vertebral body, the endoscope can access the spinal canal and remove the ventral nucleus pulposus without stimulation of spinal cord [18,19]. Although a few new techniques have been reported, it is currently limited outcomes still regarding long-term follow-up and complication profiles. In particular, it is important that the continuous intraoperative electromyogram, somatosensory-evoked potentials, and intermittent transcranial motor-evoked potentials should be monitored for spinal cord safety.
A 3D finite analysis demonstrated that the postoperative stability of PECD is better than that of open ACDF. The results are particularly significant when performing rear protraction and lateral flexion [20]. Percutaneous endoscopic anterior cervical discectomy (PEACD) is also recognized as a minimally invasive procedure with minor disruption of diseased disc and no fixation needed when compared with traditional ACDF. However, study comparing PEACD with PECD showed that PEACD could make significant hypermobility and intradiscal pressure when compare to PECD. PECD is better method for the treatment of cervical diseases when compared with PEACD [21].
The main disadvantages of this procedure are steep learning curve and high risk of spinal cord injury. Firstly, most spine surgeons are not familiar with endoscopic system. Furthermore, under the endoscope, the verified local anatomy structure is hard with the narrow surgical field, especially for surgeons in the early phase of endoscopic practice. Endoscope combined with an efficient 3D navigation system can reduce the difficulty of surgery as well as increase accuracy and efficiency. In our institute, the intraoperative CT (O-ARM) combine with a navigation system, can provide intraoperative 3D imaging for most of endoscopic surgery [22]. With O-arm, a 13s or 26s scanning can accomplish with only part of radiation. Usually, few intraoperative radiographs are further needed. One of the advantages of the navigated approach is the visible trajectory for dilator and working cannel insertion to the site of surgical level, with the appropriate incision site. This technique can help surgeons place the instruments on patients with short and thick necks or lower level which is hard to be verified by x-ray image [23]. Therefore, the time of operation can be reduced. The keyhole size was assessed around the V-point in previous studies. However, we found that the V-point is not a constant structure due to the anatomical difference of individuals and the position setting of the patients. For the herniation decompression, the identification of the lateral edge of the dura and branch of the nerve root is the key anatomic localization. However, under the O-arm based navigation, a direct bone resection over the site of pathology can be achieved, especially for cervical foraminal stenosis.
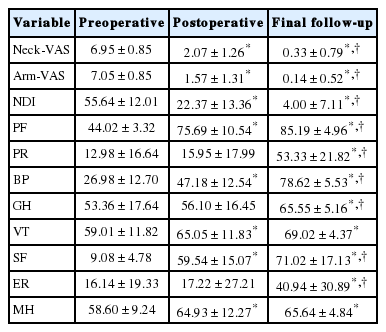
There is a significant learning curve that exists for surgeons performing PECD. Operative proficiency can be expected to occur about case 22 [24]. Despite longer operative times with earlier cases, PECD can safely and effectively be performed. In our previous study, the mean VAS of the neck and arm improved from 6.95 to 2.07 and 7.05 to 1.57, and Neck Disability Index improved from 55.6% to 22.30% immediately after the operation and increasingly improved in final follow-up [22] (Table 1). These data were in concordance with other’s results [5-7,23]. The PECD technique provides comparable outcomes, similar complication profiles, and index level reoperation rates but with less blood loss, fewer pain requirements, faster recovery, and shorter hospital stay over the open PCF technique. Also avoid the painful postoperative recovery, potential wound complications and risk of postoperative kyphosis caused by stripping of the posterior cervical musculature. The authors reported equivalent outcomes with no severe complications. Notably, and the 4.7% complication rate in our study was limited to transient hypesthesia [22]. Instead of increasing kyphosis or instability, the segmental Cobb angles were improved but not significantly changed [16].
CONCLUSION
PECD is a safe and efficient minimally invasive procedure for radiculopathy caused by soft cervical disc herniation and foraminal stenosis. Intraoperative 3D navigation is useful in guiding the extent of the laminoforaminatomy and reducing the difficulty of this procedure. Although PECD is a promising procedure for cervical disease treatment, the indications still should be considered cautiously due to the high-risk profile of cervical spine.
Notes
The authors have nothing to disclose
SUPPLEMENTARY MATERIALS
Supplementary video clips can be found via https://doi.org/10.14245/ns.2040166.083.v.1.