Frailty Status Is a More Robust Predictor Than Age of Spinal Tumor Surgery Outcomes: A NSQIP Analysis of 4,662 Patients
Article information
Abstract
Objective
The present study aimed to evaluate the effect of baseline frailty status (as measured by modified frailty index-5 [mFI-5]) versus age on postoperative outcomes of patients undergoing surgery for spinal tumors using data from a large national registry.
Methods
The National Surgical Quality Improvement Program database was used to collect spinal tumor resection patients’ data from 2015 to 2019 (n = 4,662). Univariate and multivariate analyses for age and mFI-5 were performed for the following outcomes: 30-day mortality, major complications, unplanned reoperation, unplanned readmission, hospital length of stay (LOS), and discharge to a nonhome destination. Receiver operating characteristic (ROC) curve analysis was used to evaluate the discriminative performance of age versus mFI-5.
Results
Both univariate and multivariate analyses demonstrated that mFI-5 was a more robust predictor of worse postoperative outcomes as compared to age. Furthermore, based on categorical analysis of frailty tiers, increasing frailty was significantly associated with increased risk of adverse outcomes. ‘Severely frail’ patients were found to have the highest risk, with odds ratio 16.4 (95% confidence interval [CI],11.21–35.44) for 30-day mortality, 3.02 (95% CI, 1.97–4.56) for major complications, and 2.94 (95% CI, 2.32–4.21) for LOS. In ROC curve analysis, mFI-5 score (area under the curve [AUC] = 0.743) achieved superior discrimination compared to age (AUC = 0.594) for mortality.
Conclusion
Increasing frailty, as measured by mFI-5, is a more robust predictor as compared to age, for poor postoperative outcomes in spinal tumor surgery patients. The mFI-5 may be clinically used for preoperative risk stratification of spinal tumor patients.
INTRODUCTION
Spinal tumors are much less frequent than intracranial tumors, with an estimated overall prevalence of one spinal tumor for every 4 intracranial lesions [1-4]. In the United States, the overall incidence of spinal tumors was found to be approximately 0.62 per 100,000 persons [1,3]. Metastatic spinal tumors account for the majority (up to 70%) of spinal tumors [5-7]. In a SEER (Surveillance, Epidemiology, and End Results) and National Program of Cancer Registries analysis, the majority of primary spinal tumors (78%) were benign [1]. Spinal tumors are further classified based on location into extradural (55%), intradural extramedullary (40%), and intramedullary (5%) [2,5,6]. Collectively, the management of spinal tumor patients is quite challenging [8]. The objectives of surgical treatment of primary and isolated metastatic spinal tumors include symptom palliation, prolonged survival, and curative therapy if possible [5,6,8]. Nonetheless, surgical intervention for spinal tumors may involve complex spinal reconstruction, which is associated with high peri-and postoperative complications rates [3,5,6,8-10]. To this end, preoperative risk stratification of spinal tumor patients is critical to optimize surgical outcomes [6,8].
The postoperative morbidity and surgical outcomes of spinal tumor resections represent a unique challenge with an urgent need for effective predictive tools for preoperative risk stratification in these patients [3,6,8,11]. Previously, advancing age has been identified as a poor prognostic factor for surgical outcomes of spinal tumors [5,12-18], nonetheless, the majority of this data is based on single-center, retrospective studies. Additionally, in recent years, clinicians have moved past age alone as a prognostic indicator given that frailty status (a measure of physiological reserve) has been identified as an independent and more robust predictor of outcomes after neurosurgical interventions as compared to chronological age [19,20]. Previously, frailty measures have been reported to exhibit a greater effect size and a better discriminative value to predict adverse events than chronological age alone in spine surgery patients [20].
Previous studies using data from large national databases have reported on preoperative risk stratification of spinal tumor patients [3,6,8,11]; however, to the best of our knowledge, none of these studies have directly compared frailty status at presentation and chronological age and their influence on surgical outcomes following resection of spinal tumors. Our study reports on a comparative analysis of the effects of baseline frailty status (as measured by modified frailty index-5 [mFI-5]) and age on outcomes after surgery of spinal tumors patients using data extracted from the American College of Surgeons (ACS) prospective registry, National Surgical Quality Improvement Program (NSQIP).
MATERIALS AND METHODS
1. Data Source
Patient data from 2015–2019 was obtained from the ACS database, NSQIP. The NSQIP database contains validated, multi-institutional (private and academic centers) data collected from institutions employing a uniform protocol by trained surgical reviewers across institutions [21]. NSQIP prospectively collects data from more than 700 participating sites on greater than 200 variables, including preoperative, intraoperative, 30-day postoperative variables, and all complications for patients who underwent surgery across all major surgical specialties. There is no administrative censoring or loss to follow-up of patients in the NSQIP database. The NSQIP data files are Health Insurance Portability and Accountability Act–compliant and do not identify patients, hospitals, or providers. The quality and reliability of the NSQIP data are ensured through rigorous training of data abstractors and interrater reliability audits of participating sites [22]. The NSQIP database has been employed previously to study outcomes in neurosurgical patients including spine surgery [6,8,20,23-27]. The present study was performed under the data user agreement of the ACS with University of New Mexico Hospital and was approved and considered exempt from continuing review by our Institutional Review Board (Study ID 21-315).
2. Patient Population and Baseline Characteristics
The current procedural terminology and International Classification of Diseases (ICD)-9 and ICD-10 codes were used to identify patients in the NSQIP data set (2015–2019) ages 18 years or older who, under general anesthesia, underwent resection of extradural, intradural extramedullary, and intramedullary primary or metastatic spinal tumors with a neurosurgeon or an orthopedic surgeon (Table 1) [6,8]. The baseline study population characteristics included age, sex, body mass index (BMI, kg/m2, calculated from weight and height), smoking status, and functional dependence (including both complete and partial dependence). The spinal cord tumor variables extracted included tumor location (extradural, intradural extramedullary, or intramedullary) and tumor type (primary, secondary/metastasis, or unknown). Operative time was also extracted. The comorbidities evaluated included diabetes mellitus (DM), chronic obstructive pulmonary disease (COPD), hypertension requiring medication, congestive heart failure (CHF), dyspnea, disseminated cancer (defined by the NSQIP as multiple metastases indicating that the cancer is widespread), open wound, steroid use, weight loss (substantial unintentional loss in body weight [ > 10%]), bleeding disorders (any chronic/persistent/active condition that places the patient at risk for excessive bleeding [e.g., vitamin K deficiency, hemophilia, thrombocytopenia, chronic anticoagulation therapy that has not been discontinued before surgery]), preoperative transfusion (preoperative blood loss or anemia necessitating transfusion of whole blood/packed red blood cells), and preoperative sepsis/septic shock/systemic inflammatory response syndrome (SIRS). Preoperative SIRS is defined by NSQIP as the presence of at least 2 of the following criteria: temperature > 38°C or < 36°C, heart rate > 90 beats per minute, respiratory rate > 20 breaths per minute or PaCO2 <32 mmHg, leukocytosis or leukopenia (white blood cell count > 12,000/mm3 and < 4,000/mm3, respectively) or > 10% immature (band) forms, or anion gap acidosis.
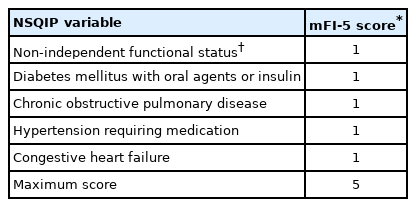
3. Modified Frailty Index-5
We used the mFI-5 as a measure of frailty. Previously, mFI-5 has been documented as an effective predictor of postoperative morbidity and mortality in neurosurgical patients including those undergoing spine surgery [20,28-30]. The mFI-5 categorical assessment was reported to be essentially equivalent to the 11-factor modified frailty index (mFI-11) score for spine surgery patients [20]. The mFI-5 score was calculated for each patient based on the presence of DM, hypertension, CHF, COPD, and dependent functional status (Table 2) [29]. The mFI-5 calculated using the 5 NSQIP variables resulted in an index ranging from 0 (least frail) to 5 (most frail), with a score of 1 as “prefrail,” 2 as “frail,” and 3 or more as “severely frail” as categorical variables, in accordance with the previously established standards [20,29].
4. Outcome Measures
Outcome measures included mortality, major complications, unplanned readmission, unplanned reoperation, hospital length of stay (LOS), and discharge to nonhome destination. Based on previous studies, patients who experienced one or more of the following postoperative adverse events were considered to have major complications: prolonged intubation of 48 hours or more, unplanned reintubation, sepsis/septic shock, deep vein thrombosis/thrombophlebitis, pulmonary embolism, coma, cerebrovascular accident/stroke with neurological deficit(s), myocardial infraction/cardiac arrest requiring cardiopulmonary resuscitation, surgical site infection (SSI, superficial/deep/organ space), wound disruption/dehiscence, acute renal failure, and pneumonia [6,8,31]. Further minor complications data extracted included perioperative blood transfusion, renal insufficiency, and urinary tract infection (UTI).
5. Statistical Analysis
All statistical analyses were performed employing IBM SPSS Statistics ver. 27.0 (IBM Co., Armonk, NY, USA) and GraphPad Prism v 9.0 (GraphPad Software Inc., La Jolla, CA, USA). Continuous variables with skewed data distribution are reported as median (and interquartile range, IQR). The D’Agostino-Pearson, Shapiro-Wilk, and Kolmogorov-Smirnov normality tests were used to determine whether the data were normally distributed or skewed. The incidence of mortality and major complication data in different age groups and frailty tiers are presented as percent incidence. The univariate analyses for age and mFI-5 were performed for the following outcomes: 30-day mortality, major complication, unplanned reoperation, unplanned readmission, hospital LOS, and discharge to a nonhome destination. Multivariable modeling of age and mFI-5, controlling for covariates, was done to define the discriminative ability of each measure. Effect sizes were summarized by odds ratio (OR) (dichotomous outcomes) or beta coefficients (continuous outcomes) and associated 95% confidence intervals (95% CIs). Receiver operating characteristic (ROC) curve analysis was performed to investigate the individual discrimination of age and frailty (by mFI-5) for mortality, and corresponding area under the curve (AUC) was depicted with 95% CI. For all purposes, p-value of < 0.05 was considered as statistically significant.
RESULTS
1. Study Population Characteristics
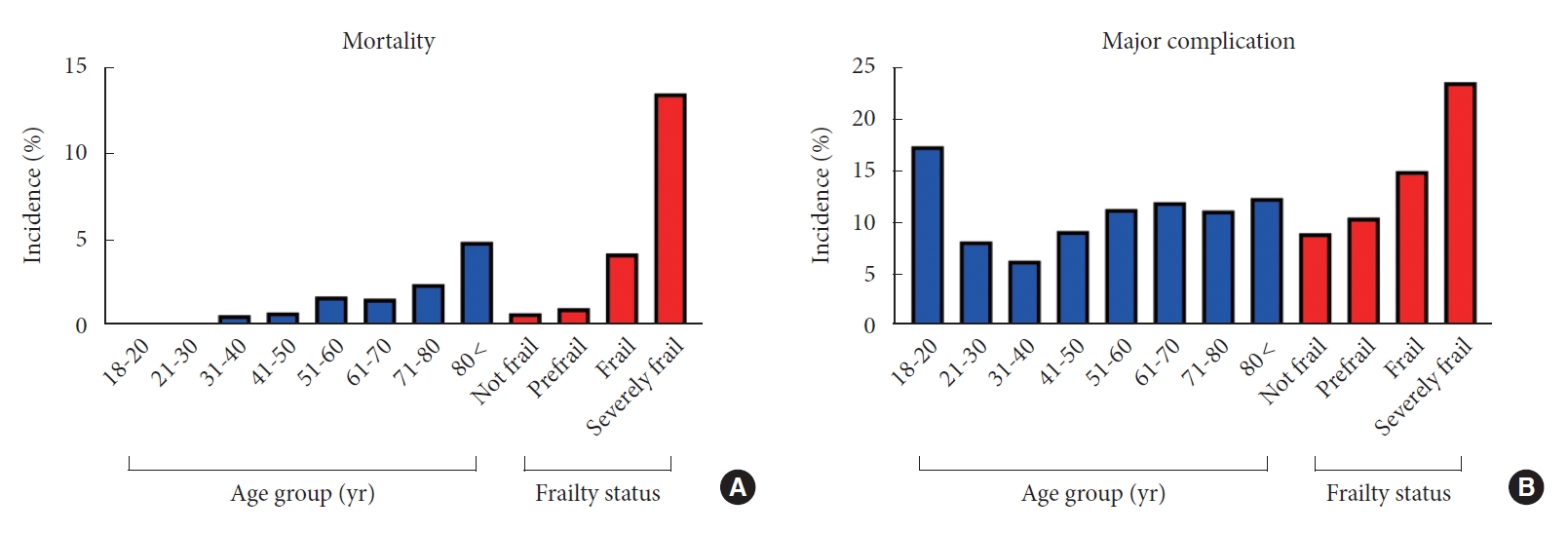
We identified and extracted data for a total of 4,662 spinal cord tumor patients who met our inclusion criteria. Median age of the study population was 59 years (IQR, 47–68 years); 53% were males with a median BMI of 19 kg/m2 (IQR, 16.3–22 kg/m2). The age distribution analysis showed the highest proportion of spinal tumor patients to be 61–70 years (25.7%), followed by 51–60 (23.4%) and 71–80 years (15.7%). Detailed study population characteristics are summarized in Table 3. Tumor location distribution was as follows: 46.7% extradural, 41.1% intradural extramedullary, and 12.2% intramedullary. Primary spinal cord tumors accounted for 37.6% whereas 43.3% were secondary/metastatic tumors; the tumor type was unknown in 19% patients. Hypertension was the most frequently observed comorbidity (41.7%), followed by disseminated cancer (28.8%) and DM (13.7%). Additionally, 7.3% of patients were functionally dependent (partially or completely) at initial presentation. The frailty distribution analysis showed that 50.4% were not frail, 34.8% were prefrail, 13.2% were frail, and 1.9% were severely frail. The median hospital LOS was 5 days (IQR, 3–9 days), and the median operative time was 187 minutes (IQR, 133–261 minutes). Postoperative 30-day mortality was 1.6%. Readmission occurred in 9.3% of patients, while 5.1% of patients required reoperation. Major and minor postoperative complications were observed in 10.6% and 15.6% of patients, respectively. The most common postoperative complication was perioperative blood transfusion (12.6%), followed by UTI (3.5%) and pneumonia (2.3%). Finally, 64.4% of patients were discharged home whereas the remaining were nonroutine discharges or mortality. The highest incidence of mortality and presence of major complication were noted in the “severely frail” group (Fig. 1; 13.5% and 23.6%, respectively).

Baseline demographic and clinical characteristics and outcomes of patients undergoing surgery for spinal tumors from the NSQIP database 2015–2019 (n=4,662)
2. Univariate Analysis of Age and Frailty Status on Surgical Outcomes
Univariate analysis demonstrated that frailty status (based on mFI-5 score) was a better predictor than age of 30-day mortality, presence of major complication, unplanned readmission, unplanned reoperation, hospital LOS, and discharge to nonhome destination (Table 4). Based on the analysis of frailty categories, increasing frailty was significantly associated with all outcome variables as evidenced by increasing effect size (Table 4).
3. Multivariate Analysis of Age and Frailty Status on Surgical Outcomes
Multivariable regression analysis (adjusting for sex, BMI, tumor location, tumor type, and operative time) confirmed that, with higher effect size, frailty status was a better predictor of adverse surgical outcomes (Table 5). Based on categorical analysis of frailty tiers, increasing frailty was significantly associated with increased risk of all adverse outcomes, with ‘severely frail’ patients demonstrating an OR of 16.4 (95% CI, 11.21–35.4) for 30-day mortality and an OR of 3.02 (95% CI, 1.97–4.56) for presence of a major complication (Table 5).
4. ROC Curve Analysis of Age and mFI-5 for Mortality
The ROC curve analysis showed superior discrimination of frailty (mFI-5) for mortality (AUC=0.743; 95% CI 0.661–0.825; p<0.001) in comparison with age (AUC=0.594; 95% CI, 0.512–0.667; p=0.043) (Fig. 2).
DISCUSSION
Resection of spinal tumors is performed to improve functional (ambulatory) status, reduce pain, and in certain cases, to improve survival chances. However, the associated mortality with the surgical intervention is well documented in both single-center and large national database studies [3,5,6,8,11,32-34]. Thus, it is critical to identify and validate prognostic predictors for preoperative risk stratification of spinal tumor patients. The current study reports that baseline frailty status (as measured by mFI-5) is a more robust predictor than chronological age of postoperative complications, i.e., 30-day mortality, major complication, unplanned readmission, unplanned reoperation, hospital LOS, and discharge to nonhome destination in spinal tumor patients. Our findings demonstrate the importance of frailty status assessment rather than just chronological age in preoperative risk stratification of spinal tumor cases.
Previously, the 30-day mortality rate after surgical intervention for spinal tumor was reported as 4.5% in a 2008–2014 NSQIP study [8], 3.3% in 2011–2014 NSQIP data, 0.55% in 1993–2002 Nationwide Inpatient Sample (NIS) data [11], and 0.46% in NIS data from 2003–2010 [3]. In the present NSQIP study utilizing data from 2015–2019, we found the 30-day mortality rate after spinal tumors surgery to be 1.6%. Among all the postoperative outcome variables studied, baseline frailty status (mFI-5 score) had the strongest association, as evidenced by the larger effect sizes with postoperative mortality. Previously, mFI-5 was reported to be an effective predictor of postoperative mortality in brain tumor surgery patients [35]. Another study found mFI-5 to be the best predictor of mortality after spine surgery for degenerative cervical myelopathy as compared with age, mFI-11, modified Charleston comorbidity index, and American Society of Anesthesiologists physical status classification [20]. Our study data show that mFI-5 is an effective predictor of mortality after surgery for spinal tumors.
In our study, the major and minor postoperative complication rates were 10.6% and 15.6%, respectively. This is comparable to previously reported major and minor postoperative complication rates of 11.5% and 19.8% in 2008–2014 NSQIP spinal tumor data [8], major complication rate of 14.4% in 2011–2014 NSQIP spinal tumor cases [6], and total complication rate of 17.5% from NIS data from 1993–2002 [11]. The presence of high postoperative morbidity in our large national data set and other previously published reports emphasizes the importance of identifying risk factors for postoperative complications. While age was a predictor of postoperative complications, we found that it was frailty status (mFI-5 score) which highly effective in predicting the presence of major postoperative complications, suggesting that mFI-5 can be used for preoperative risk stratification in this patient population. Although mFI-5 was previously reported to be an effective predictor of postoperative outcomes in brain tumor patients [36], to the best of our knowledge, the present study is the first to document baseline mFI-5 score as a robust predictor of postoperative complications in spinal tumor patients in a large national database.
LOS was previously reported to be a driver of hospitalization cost in spine surgery patients, and increased LOS was associated with a higher risk of postoperative complications such as infectious and adverse thromboembolic events [37-39]. Previously, mFI-11 and CCI were reported to be associated with extended LOS in spinal surgery patients based on 2008–2014 NSQIP data. Based on both univariate and multivariate analyses, we also report an association of higher mFI-5 score with increased LOS, and thus an indirect measure of increased cost of hospitalization due to increasing frailty status of the patients. Moreover, we found higher effects sizes of increasing frailty tiers for unplanned readmission and reoperation within 30 days of spinal tumor surgery. Our belief is that this can be explained by the fact that in spine surgery patients, the most common indications for early reoperation and readmission are compressing hematoma, SSI, and hardware failure, and patients with higher mFI-5 scores are more likely to develop postoperative SSIs and carry a higher risk of osteopenia/osteoporosis [20]. Previously, a systematic review of frailty and spine surgery outcomes reported that higher frailty scores were associated with nonhome discharge [40]. The present study data employing mFI-5 frailty score further corroborate these findings in spinal tumors patients.
There are a few limitations of the present study, mainly ones inherent to any analysis based on a large national database, and therefore the results need to be interpreted in a prudent manner. Firstly, the NSQIP data only records postoperative outcomes within the initial 30 days of the surgery. As a result, it is impossible to gauge long-term outcomes and survival in spinal tumor patients. Secondly, NSQIP data does not include tumor size, intraoperative complications, and postoperative neurological outcomes, variables relevant to spine tumors patients. Thirdly, the present study is a retrospective analysis of a prospectively collected national dataset and therefore may be subject to inherent selection bias. Despite these limitations, the current study represents the largest series of spinal tumors patients analyzing the effect of baseline frailty status in comparison with age on surgical outcomes. The large sample size provides the necessary statistical power to recognize mFI-5 frailty score as a robust predictor of postoperative outcomes in spinal tumors patients. Furthermore, given that our study is based on a large national data set, it therefore carries significant generalizability beyond single-center data.
CONCLUSION
In conclusion, our study represents one of the most detailed analyses with a large sample size of postoperative outcomes in spinal tumor patients and is the first to report a direct comparison of age and frailty status employing mFI-5 score. Baseline frailty status is increasingly being used in preoperative risk stratification of neurosurgical patient populations and is emerging as a better predictor of outcomes than chronological age across multiple neurosurgical procedures [19,20,40]. Based on multivariate analysis, we determined that “severely frail” status was associated with the highest effect sizes for 30-day mortality, presence of major complication, unplanned readmission, reoperation, and higher LOS. The results of this work show that frailty status (i.e., a measure of physiological reserve) influences postoperative outcomes more significantly than increased chronological age. Future large-scale multicenter prospective studies are warranted to validate these findings. The greatest clinical significance of present study is the improvement in preoperative risk stratification and subsequent preoperative patient counseling regarding the risks and benefits of proposed surgical intervention. Surgeons can use the knowledge that frailty is more significant than age in predicting outcomes after spinal tumors surgery to more accurately judge whether a patient would do well with spinal tumor surgery. Furthermore, this provides support to the conclusion that there are some severely frail spinal tumor patients that should not be offered a surgical resection. It is important to assess potential surgical patients with significant frailty to help determine whether their frailty phenotype is amenable to better outcome with individualized pre- and perioperative specialized care. In other fields, the use of prehabililation prior to surgery has been gaining traction. We believe this is worthy of investigation, but we believe that studies like the present one are important as critical first steps to identify whether worse outcomes are expected for specific neurosurgical diseases based on frailty.
Notes
Conflict of Interest
The authors have nothing to disclose.
Funding/Support
This study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author Contribution
Conceptualization: SFK, AD, CB, WC, RT, DAC, OT, KC, RM, CC, MS; Data curation: SFK, CB, RM, CC, MS; Formal analysis: SFK, AD, CB, SS, RT, DAC, MC, OT, JV, KC, CR, PS, MS; Funding acquisition: C Bowers, WC, RM, MS; Methodology: SFK, AD, CB, SS, WC, RT, DAC, MC, OT, JV, KC, JD, RM, CR, PS, CC, MS; Project administration: CB, WC, MS; Visualization: SFK, AD, CB, SS, WC, RT, DAC, JV, JD, CR, PS, MS; Writing - original draft: SFK, AD, CB, SS, WC, RT, DAC, MC, OT, JV, KC, JD, CR, PS, MS; Writing - review & editing: SFK, AD, CB, WC, RT, DAC, OT, KC, JD, RM, CR, PS, MS.