Anterior Endoscopic Cervical Discectomy: Surgical Technique and Literature Review
Article information
Abstract
The conventional surgical technique for radiculopathy with cervical disc herniation (CDH) is anterior cervical discectomy and fusion, with a good clinical outcome and fusion rate. However, significant perioperative morbidity related to extensive surgical exposure has been reported. Therefore, anterior endoscopic cervical discectomy (AECD) using a working channel endoscope has been developed to reduce surgical complications and tissue damage. The objective of this study was to describe a cutting-edge technique for AECD of soft CDH. The primary indication is cervical radiculopathy with or without axial neck pain due to soft CDH. The surgical procedure consists of 2 parts: (1) a safe anterior percutaneous approach under fluoroscopic control and (2) selective endoscopic discectomy and foraminal decompression using specialized mechanical tools under endoscopic visualization. The clinical outcomes are comparable to those of conventional surgery and show the benefits of minimally invasive spine procedure. Perioperative data revealed typical minimalism, including reduced muscle damage, blood loss, operative time, and recovery time. With technical advancements in surgical instruments and optics, AECD will become more practical and safer. AECD is effective in selected CDH cases with cervical radiculopathy. However, high-quality clinical studies are needed to verify the effectiveness of this endoscopic cervical spinal procedure.
INTRODUCTION
The standard surgical technique for cervical radiculopathy with cervical disc herniation (CDH) is anterior cervical discectomy and fusion (ACDF), which is a reliable surgical option with an acceptable fusion rate [1-6]. However, there are considerable surgical morbidities that may interfere with patients’ recovery to their normal lives: (1) approach-related problems such as difficulty in swallowing, hematoma, hoarseness, and esophageal injury [7,8]; (2) fusion-related events such as motion limitation, non-union, and hardware failure [9-11]; and (3) adjacent segment disorders [12,13].
Since Hijikata [14] and Kambin and Sampson [15] first introduced percutaneous lumbar discectomy, various endoscopic spine surgery techniques have been developed. With regards to the cervical spine, some pioneers have evolved minimally invasive surgical techniques via a percutaneous anterior cervical approach. A percutaneous approach using a small working channel endoscope instead of an open anterior approach may reduce approach-related adverse events and extensive tissue trauma. Currently, anterior endoscopic cervical discectomy (AECD) and modified procedures have been reported to be effective in appropriately selected CDH cases [16-25]. However, technical modifications of this procedure are variable and need to be integrated and standardized.
This article describes the basic technique of AECD, and discusses its clinical outcomes and technical modifications, including the scientific evidence.
INDICATIONS
The clinical indications for AECD are: (1) severe cervical radiculopathy with or without neck pain, (2) discogenic cervical headache, (3) cervical myelopathy with a high risk of requiring extensive open surgery under general anesthesia, and (4) persistent or progressive symptoms despite at least 3 months of nonsurgical therapy. The radiological indications are: (1) soft CDH compressing the spinal cord or exiting nerve root demonstrated on computed tomography (CT) and magnetic resonance imaging (MRI) and (2) preserved intervertebral disc space. The procedure can be performed when the situation meets the radiological and clinical criteria simultaneously [18,22].
The contraindications are: (1) calcified or hard CDH; (2) advanced spondylosis with collapsed disc space; (3) cervical myelopathy with cervical stenosis, ossification of the posterior longitudinal ligament, or ossification of the ligamentum flavum; (4) segmental spinal instability or kyphotic deformity; (5) other neurological or vascular diseases mimicking degenerative disc pathologies; and (6) a history of anterior neck surgery at the index level.
SURGICAL PROCEDURES
The step-by-step surgical procedure of AECD for cervical radiculopathy with soft CDH consists of (1) an anterior percutaneous approach under fluoroscopic control and (2) selective discectomy and foraminal decompression through endoscopic visualization [22,26].
1. Patient Position, Anesthesia, and Skin Marking
The procedure can be performed under either general or local anesthesia according to the patient’s condition and the surgeon’s preference. In the case of local anesthesia, the basic premedication comprises midazolam (0.05 mg/kg, intramuscularly) and fentanyl (0.8 μg/kg, intravenously) administration on call. The local anesthetics can be added as required during the procedure. The patient is placed in a supine position with their neck extended on a radiolucent spine table. After adequate anesthesia and positioning, essential anatomical structures are marked under fluoroscopic guidance, including the index level, carotid pulse, and medial margin of the sternocleidomastoid muscles.
2. Fluoroscopic-Guided Percutaneous Anterior Cervical Approach
The basic concept of the surgical approach is a percutaneous anterior approach through the safe working zone between the carotid artery and the trachea into the cervical disc. The deep fascia separates the vascular compartment (including the carotid artery and vein) and visceral compartment (including the trachea and esophagus). Therefore, a simple finger pressure between the vascular and visceral parts can easily create a safe working zone.
Contralateral access is recommended because it provides a better visual field for the lateral and foraminal zones of the disc. After confirming the operative disc level using anteroposterior (AP) and lateral fluoroscopic views, the surgeon palpates the carotid pulse and keeps it lateral from the surgical field. The surgeon then presses the space between the carotid vessels and trachea with their fingers, pushing the trachea to the contralateral side. When the surgeon feels the anterior surface of the disc with their fingertips, an 18-gauged approach needle is inserted into the disc space directly from the midzone of the anterior disc surface to the lateral target disc point (Fig. 1A). After confirming adequate needle trajectory and landing point on the AP and lateral fluoroscopic views, intraoperative discography is performed to stain the herniated disc fragment and identify the leakage status with a mixture of indigo carmine and contrast media. Next, a guidewire is introduced through the needle sheath into the disc, and a stab skin incision, less than 5 mm, is made horizontally at the index cervical disc level. After sequential dilation using dilators of different sizes, a round or rectangular working sheath is placed to ensure the surgical field of the herniated disc (Fig. 1B). A trephine can be inserted through the working sheath and cut into the annulus to reduce the intradiscal pressure and resistance.
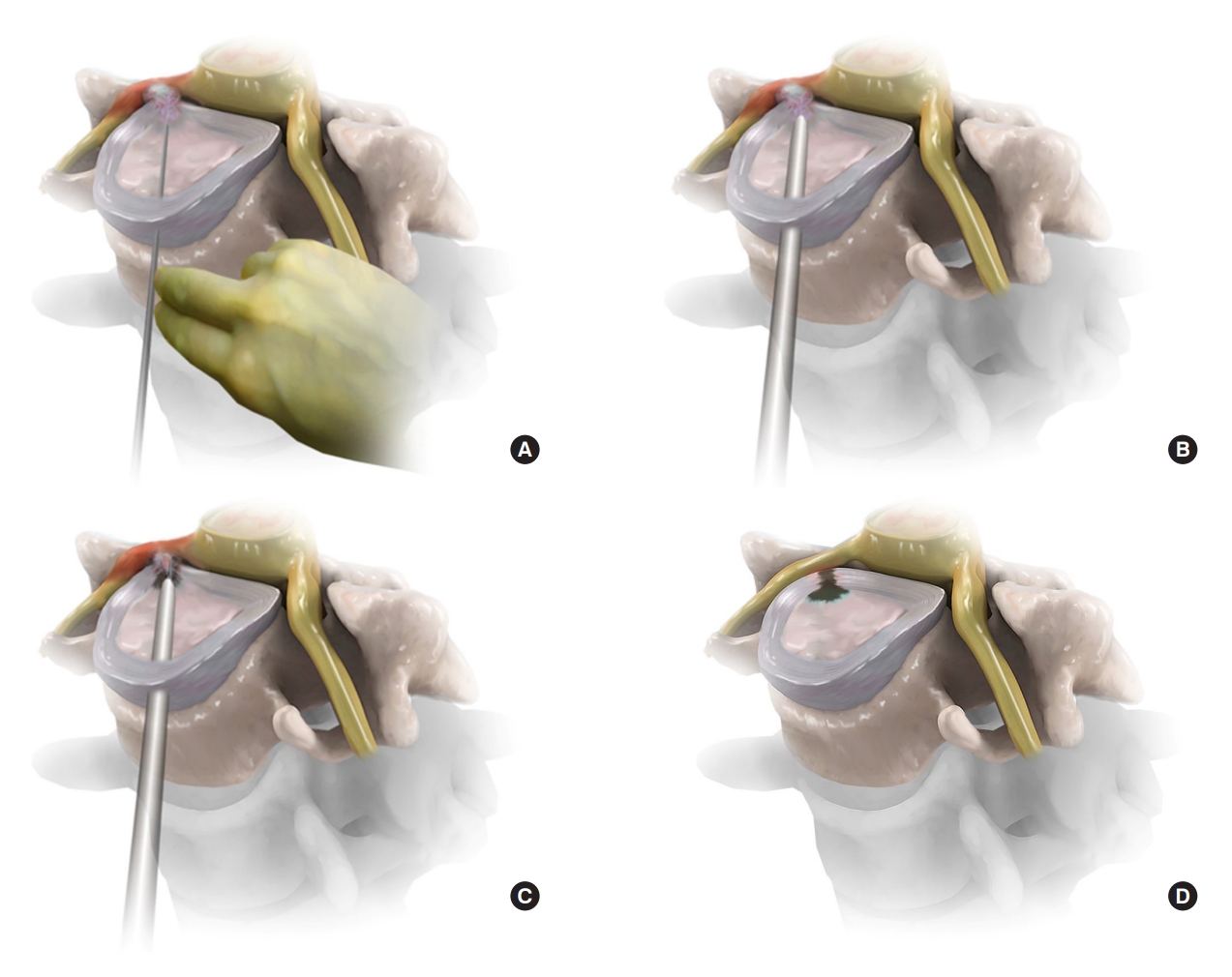
Schematic illustrations of anterior endoscopic cervical discectomy. (A) A contralateral needle insertion is made through the safety working space between the carotid artery and tracheoesophagus to the herniated disc under fluoroscopic control. (B) After intraoperative discography and serial dilation, the oval-shaped working sheath is placed at the posterior portion of the target disc point. (C) After the sufficient release of annular anchorage, the released herniated fragment is removed using endoscopic mechanical tools and supplementary devices. (D) The solid pulsation and free mobilization of the neural tissue under an endoscopic visualization can determine the final neural decompression.
3. Endoscopic-Guided Selective Discectomy and Foraminotomy
The main procedure in this step is the selective removal of herniated fragments under endoscopic visualization while preserving the central nucleus of the maternal disc. An ellipsoid working channel endoscope is introduced through the working sheath, and the posterior disc space is identified through endoscopic visualization. The surgical disc space is continuously irrigated with antibiotic-containing saline at a rate of 30–40 mL/min. Disc decompression is first performed in the posterior subannular area to reduce intradiscal pressure and create adequate working space. This initial decompression and release process should be performed using endoscopic forceps, a semiflexible radiofrequency tip, or a side-firing laser, until the annular fissure and herniated disc fragment are identified. Then, the annular anchorage is released using an endoscopic cutter or other devices until fibrotic adhesion and the herniated disc fragment are separated. The herniated element can then be selectively removed piece by piece using fine endoscopic forceps. During selective decompression, thecal sac pulsation can be gradually detected through the torn annular fissure. Decompression can proceed to the foraminal zone. After selective discectomy and foraminotomy, the epidural space and decompressed exiting nerve root can be seen through the opened annular fissure (Fig. 1C).
4. Final Checking Point
The final point of the procedure can be determined by the solid pulsation of the thecal sac and free mobilization of the exiting nerve root (Fig. 1D). Surgeons should examine whether there is any dural membrane breach in the surgical field. They should also check for any epidural or bone bleeding to prevent postoperative hematoma. After surgery, the endoscope is withdrawn, and the wound is closed with a one-point subcutaneous suture and skin tape. Postoperative MRI or CT scans may be checked for precise decompression of the primary pathology as required (Fig. 2). If there are no significant adverse events, the patient can be discharged within 24 hours postoperatively (Fig. 3).

A 48-year-old female patient underwent anterior endoscopic cervical discectomy at the C4–5 level. (A, B) An approach needle is introduced between the carotid artery and the tracheoesophagus. Note the endotracheal tube or tracheal air shadow is pushed contralaterally using the fingertips, ensuring the safety working space in the fluoroscopic view. (C, D) After placing the working sheath intradiscally, a selective discectomy is conducted under endoscopic visualization.
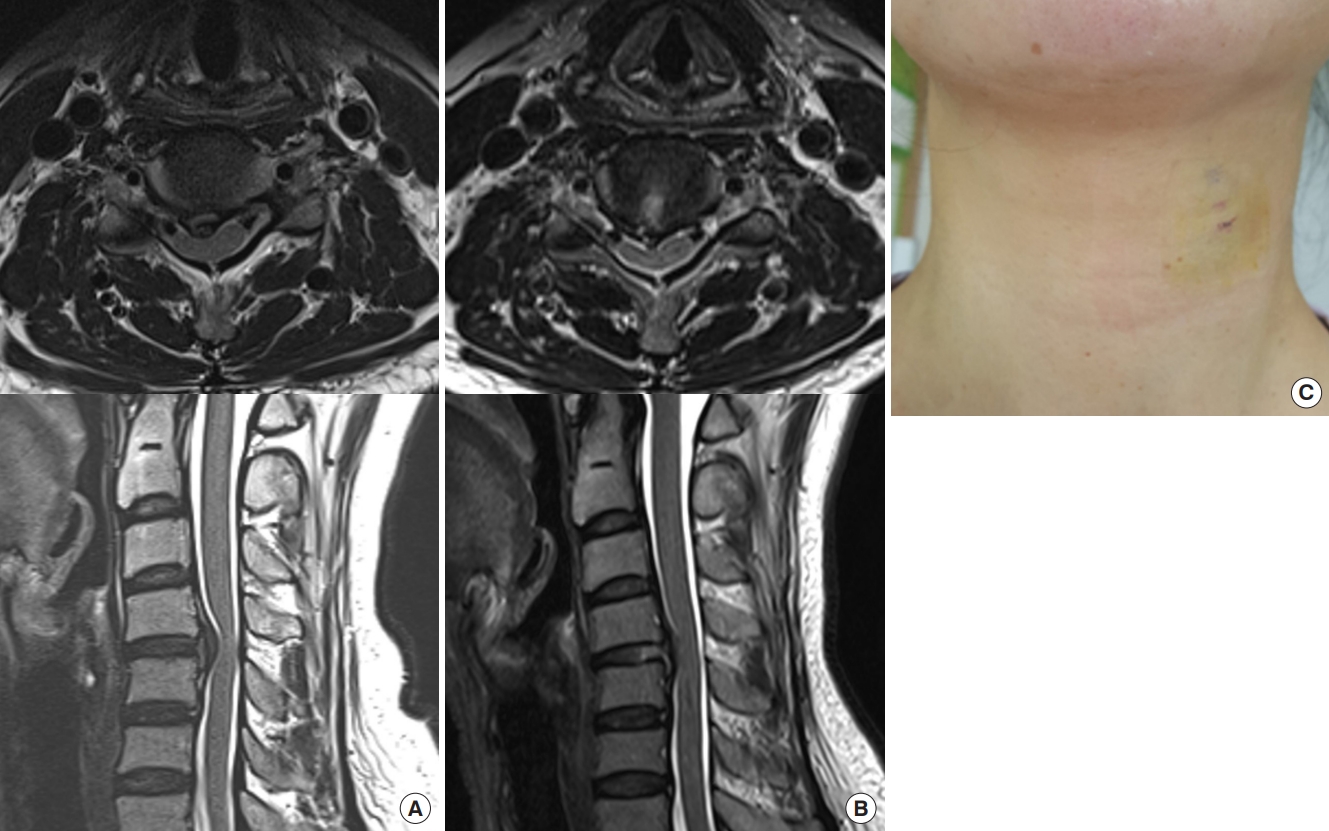
Postoperative status of a 48-year-old female patient with soft cervical disc herniation at the C4–5 level. (A) Preoperative axial and sagittal magnetic resonance (MR) images demonstrate an extruded disc compressing the spinal cord and exiting nerve root. (B) Postoperative axial and sagittal MR images show a well-decompressed status immediately after the procedure. Note the selective removal of the herniated disc while preserving the central nucleus. (C) The patient’s symptoms improved immediately without any adverse events. She could return to ordinary work within 1 month postoperatively with minimal surgical scar.
RESULTS
According to our comparative cohort study between the AECD group (51 patients) and ACDF group (64 patients), the 5-year clinical outcomes were identical between the groups [22]. The mean visual analogue scale scores for radicular pain improved from 4.58 ± 1.95 to 1.35 ± 1.34 in the AECD group and from 3.91 ± 1.78 to 1.14 ± 0.85 in the ACDF group. The neck disability index improved from 51.87 ± 21.47 to 7.82 ± 13.41 in the AECD group and from 58.27 ± 17.73 to 6.59 ± 10.14 in the ACDF group. According to the global results based on the modified MacNab criteria, the success rates were 88.24% and 90.63% in the AECD and ACDF groups, respectively.
We found that the endoscopic procedure showed the typical characteristics of minimally invasive surgery: shorter operative time, shorter hospital stay, and earlier return to work. The mean operative time was 55.20 ± 18.03 minutes in the AECD group versus 124.53 ± 35.68 minutes in the ACDF group (p < 0.001). The mean postoperative hospital stay was 2.18 ± 1.16 days versus 5.23 ± 2.39 days, respectively (p < 0.001). The time to return to work was 3.14 ± 1.08 weeks vs. 10.84 ± 3.12 weeks, respectively (p < 0.001).
Complications were similar between the groups. The most common complication was transient swallowing difficulty (1.96% vs. 4.69%, respectively), which improved within 4 weeks postoperatively. Two patients in the AECD group (3.92%) underwent subsequent ACDF because of recurrent disc herniation in 2 months. One patient in the ACDF group (1.56%) underwent subsequent posterior cervical foraminotomy and fusion because of postoperative foraminal stenosis and instability 12 months later. There was no statistical difference in the revision rate (p = 0.58).
DISCUSSION
1. Journal Review
The AECD technique has several benefits in minimally invasive surgery [26]. First, the anterior percutaneous approach with a thin working channel endoscope (3–4 mm in diameter) can preserve anterior cervical musculoskeletal structures. Therefore, postoperative scarring or surgical complications can be minimized. Overall, the recovery and rehabilitation times can be shortened compared with open surgery. Second, selective and delicate removal of herniated disc fragments can preserve the maternal nucleus. Moreover, unnecessary fusion or instrumentation can be avoided while maintaining segmental stability. Third, considering the surgical approach, herniated discs at any zone, from the central to the foraminal, and any degree of herniation, from an annular tear to extrusion, can be effectively removed under sophisticated endoscopic control. Finally, AECD may be performed under either general or local anesthesia according to the patient’s condition and request. Therefore, this technique can be applied to patients at risk for general anesthesia. Biomechanical or finite element studies revealed that anterior endoscopic cervical procedures showed a shorter surgical path, smaller surgical diameter, and less biomechanical influence on the cervical spine [27].
By contrast, AECD has inevitable shortcomings related to technical limitations. Spine surgeons are unfamiliar with cervical endoscopic procedures, which require a steep and long learning curve. Training courses and extensive clinical experience are mandatory for aspiring endoscopic surgeons to obtain good outcomes. In addition, surgical indications and clinical applications are limited to soft disc herniation. A collapsed disc, posterior stenosis, calcified pathologies, and severe myelopathy may be contraindicated in actual cases.
Various cases of CDH have been reported in different anatomical situations. Many technical reports and case series of AECD have been published, and the reported success rates have varied from 51% to 95% [20-25,28]. Most authors have concluded that endoscopic anterior cervical procedures are efficient for selected CHD, with the benefits of minimal invasiveness. However, scientific evidence of its clinical effectiveness is yet to be sufficiently elucidated. Some authors have published comparative cohort studies demonstrating the clinical efficiency of AECD with minimal invasiveness [22,29]. However, few randomized controlled trials (RCTs) have been conducted to verify the effectiveness of this procedure. Ruetten et al. [30] compared the clinical and radiological outcomes between full-endoscopic anterior cervical discectomy (54 cases) and ACDF (49 cases) over a 2-year follow-up period. They concluded that the endoscopic cervical procedure was a sufficient and safe alternative to ACDF in selected cases. There is also a lack of reliable meta-analyses or systematic reviews on AECD, but only technical or narrative reviews so far [26,28,31]. High-quality RCTs and systematic reviews are needed to verify the relevance and effectiveness of AECD compared with standard ACDF.
2. Technical Point
Critical technical points should be emphasized to achieve relevant and reliable results using this minimally invasive technique. The point-oriented percutaneous anterior approach is key to clinical success. The approach needle should be inserted from the contralateral side through the safe working zone, which is the interfascial space between the carotid sheath (vascular axis) and tracheoesophageal compartment (visceral axis). The surgeon’s fingertips can create a safe working zone under fluoroscopic guidance. Endotracheal tube shape or tracheal air shadow may be helpful indicators of the visceral axis. The access trajectory should be directed toward the annular fissure and herniated disc fragments. The second key to success is appropriate release of the annular anchorage before removal of the herniated disc fragments. Generally, there is a solid and tenacious adhesion between the annular fissure and offending pieces. A premature attempt to remove the elements without an adequate release process cannot result in sufficient decompression because the endoscopic devices are relatively delicate and small. The exact closing point of the AECD technique may be the last but most crucial key to success. To declare the endpoint of the surgery, the surgeon should confirm that the affected neural tissues “breathe” without any remaining tethering. The minimal condition for the definitive endpoint is solid pulsation and free mobilization of the thecal sac and nerve root, regardless of surgical exposure of the neural tissues in the endoscopic surgical field.
3. Technical Modification
With the development of endoscopic technologies, technical modifications have emerged with unique advantages. First, AECD via a transcorporeal approach was designed with the benefit of selective discectomy while preserving the maternal disc space [32-37]. Second, AECD with the interbody fusion technique combines endoscopic discectomy and immediate instrumented fusion [38-40].
4. Anterior Versus Posterior
The percutaneous endoscopic cervical procedure may be performed through the anterior or posterior approach. Both approaches might be practical if the surgeon applied the process for adequate indications [26,28]. In general, the direction of the approach may be determined according to the zone of neural compression. If the primary pathology is located at the lateral border of the myelon, the posterior approach may be useful because the posterior endoscopic approach is simple and more accessible than the anterior approach. In contrast, the anterior approach may be more effective for the case of central or paracentral pathologies since the cervical spinal cord must not be retracted into the medial direction during the procedure. Therefore, the surgeon should choose the path according to the patient’s pathology.
CONCLUSION
AECD and other modified techniques may be effective in treating cervical radiculopathy with CDH. Precise percutaneous anterior cervical localization and selective decompression of the critical point are key to success. It can result in relevant clinical outcomes with minimal invasiveness in terms of skin incision, operative time, blood loss, hospital stay, anesthesia, and recovery time. However, more high-quality clinical studies are needed to verify the effectiveness of this endoscopic cervical spinal procedure.
Notes
Conflict of Interest
The authors have nothing to disclose.
Funding/Support
This study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
