Three-Dimensional-Printed Titanium Versus Polyetheretherketone Cages for Lumbar Interbody Fusion: A Systematic Review of Comparative In Vitro, Animal, and Human Studies
Article information
Abstract
Interbody fusion is a workhorse technique in lumbar spine surgery that facilities indirect decompression, sagittal plane realignment, and successful bony fusion. The 2 most commonly employed cage materials are titanium (Ti) alloy and polyetheretherketone (PEEK). While Ti alloy implants have superior osteoinductive properties they more poorly match the biomechanical properties of cancellous bones. Newly developed 3-dimensional (3D)-printed porous titanium (3D-pTi) address this disadvantage and are proposed as a new standard for lumbar interbody fusion (LIF) devices. In the present study, the literature directly comparing 3D-pTi and PEEK interbody devices is systematically reviewed with a focus on fusion outcomes and subsidence rates reported in the in vitro, animal, and human literature. A systematic review directly comparing outcomes of PEEK and 3D-pTi interbody spinal cages was performed. PubMed, Embase, and Cochrane Library databases were searched according to PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analysis) guidelines. Mean Newcastle-Ottawa Scale score for cohort studies was 6.4. A total of 7 eligible studies were included, comprising a combination of clinical series, ovine animal data, and in vitro biomechanical studies. There was a total population of 299 human and 59 ovine subjects, with 134 human (44.8%) and 38 (64.4%) ovine models implanted with 3D-pTi cages. Of the 7 studies, 6 reported overall outcomes in favor of 3D-pTi compared to PEEK, including subsidence and osseointegration, while 1 study reported neutral outcomes for device related revision and reoperation rate. Though limited data are available, the current literature supports 3D-pTi interbodies as offering superior fusion outcomes relative to PEEK interbodies for LIF without increasing subsidence or reoperation risk. Histologic evidence suggests 3D-Ti to have superior osteoinductive properties that may underlie these superior outcomes, but additional clinical investigation is merited.
INTRODUCTION
Lumbar interbody fusion (LIF) is a staple of lumbar surgery and offers the ability to improve sagittal plane deformity, provide an additional fusion surface to decrease pseudarthrosis rates, and facilitate foraminal expansion and indirect decompression of the lumbosacral nerve roots. At present, the market is dominated by devices comprised of either titanium (Ti) alloy or polyetheretherketone (PEEK).
The ideal spinal interbody device should have 2 properties: (1) the ability to produce a rigid construct that facilitates alignment correction and indirect decompression through disc-space distraction, and (2) the ability to facilitate the bony fusion that is critical for long-term fusion success. Ti and Ti alloys (e.g., Ti6Al4V) serve as the materials for a bulk of commercially available devices as Ti and alloys are known to facilitate osseointegration at the bone-implant interface [1]. Alloys additionally have the advantage of increased corrosion resistance [2]. However, the Young’s modulus of bulk Ti and Ti alloys is 50–110 gigapascals [3,4] (GPa), which is far in excess of that of both cancellous bone (3–4 GPa) and cortical bone (14.6 GPa) [3]. This creates a mismatch between the interbody and the adjacent bone that can result in cage subsidence [5-8]. To address this, PEEK implants were developed in the late 1990s, which possess a Young’s modulus that far more closely approximates that of native bone, decreasing the risk of implant subsidence [9,10]. PEEK implants are additionally radiolucent and so can better facilitate radiographic monitoring of bony fusion during follow-up [9,11,12]. However, PEEK exhibits poor osseointegration due to its ability to form biofilms, increasing rates of bony nonunion (pseudarthrosis) [13]. To address the weaknesses of both materials, Ti-coated PEEK interbodies were produced, but they did not appear to significantly reduce subsidence rates relative to Ti implants [13] and were associated with the risk of surface coating delamination during implantation.
More recently, advances in 3-dimensional (3D)-printing and biomaterial surface treatment technology have facilitated the production of 3D-printed Ti (3D-pTi) interbody devices [14]. These devices have highly porous surfaces that facilitate osteoinduction [15], while at the same time have a low enough elastic modulus to match that of native bone [4,16,17]. However, given the relatively recent development of these devices, currently available evidence is limited. The objective of the present systematic review was to summarize the available biomechanical, animal, and human data directly comparing outcomes between 3D-pTi and PEEK lumbar interbody devices with respect to bony fusion, implant subsidence, reoperation/revision (in human studies), and construct stability/stiffness (in biomechanical studies).
METHODS
1. Search Strategy
Using the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analysis) guidelines the PubMed, Embase, and Cochrane Library databases were queried to identify all studies directly comparing outcomes between PEEK and 3D-pTi vertebral interbody implants from database inception to February 2022. Articles were identified using the following Boolean search string: “print*” AND (“peek” OR “polyetheretherketone”) AND “fusion”. Studies were included if: (1) they were available in English full texts or translations, and (2) they directly compared PEEK and 3D-pTi lumbar interbody implants with regard to biomechanical outcomes, material properties, fusion rate, subsidence rate, reoperation rate, and/or construct stability. Articles were excluded if: (1) they focused on an intervention other than lumbar fusion, or (2) were noncomparative studies, technical notes, abstracts, commentaries, clinical trials without published data, reviews or meta-analyses of previously published data. The present review was not registered and no protocol is available.
2. Screening
Two authors (NAP and JLG) independently screened titles and abstracts for relevance according to the inclusion and exclusion criteria. Any discrepancies during the screening process were resolved by consensus among all authors. Full text articles were then reviewed to determine final inclusion, and reference lists of all included studies were also queried to identify additional relevant studies.
3. Study Quality
The quality of evidence for the included cohort studies was assessed Cochrane risk of bias tool 2 for randomized trials and nonrandomized studies.
4. Data Extraction
Independent reviewers (NAP and JLG) extracted data from all eligible studies. Data extracted from human study variables included the occurrence of implant subsidence and device-related reoperation. Animal study variables included radiographic evidence of osseointegration on micro-computed tomography (μCT), histological analyses of osseointegration, including quantitative histomorphometry, and biomechanical testing results (flexion-extension, right/left lateral bending, right/left axial rotation and stiffness). Nonclinical study variables included energy effective strain, or a quantification of the energy stored in the composite based on an assessment of the effective strain, as well as human mesenchymal stem cell (hMSC) morphology, proliferation, and differentiation, DNA fluorescence assay, alkaline phosphatase (ALP), and calcium content.
RESULTS
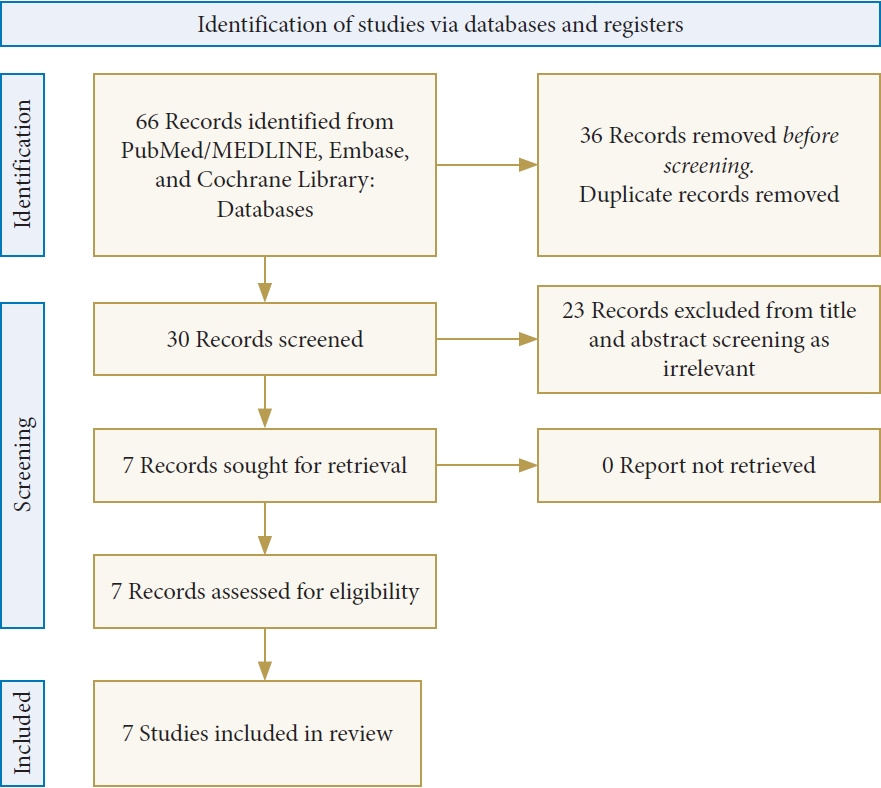
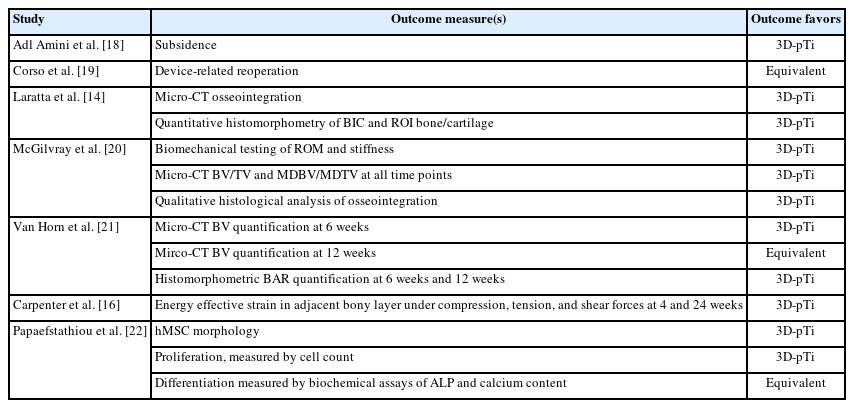
Overall, 30 unique studies were identified through the literature search, of which, 7 progressed to final study inclusion (Fig. 1). Of the 7 eligible studies (Table 1) included in this review [14,16,18-22], 2 studies were retrospective clinical cohorts [18,19], 3 studies were prospective animal studies using ovine models [14,20,21], and 2 studies were nonclinical laboratory examinations (1 finite element analysis [FEA]16 and 1 in vitro study) [22]. The mean Newcastle-Ottawa Scale score for the 5 studies was 6.4, with a range of 6 to 7. Points were lost in the "comparability” and “selection” assessments. Each study utilized a different manufacturer of their 3D-pTi. All studies used traditional PEEK for comparison except for one study which used porous PEEK. Excluding the nonclinical studies, there was a total population of 299 human and 59 ovine subjects, with 134 human (44.8%) and 38 ovine models (64.4%) implanted with 3D-pTi cages. Six studies reported results favoring 3D-pTi, while 1 study [19] reported equivocal outcomes for 3D-pTi and PEEK devices (Table 2).

Characteristics of direct comparative studies assessing 3D-pTi vs. PEEK lumber interbody implantation
1. Human studies
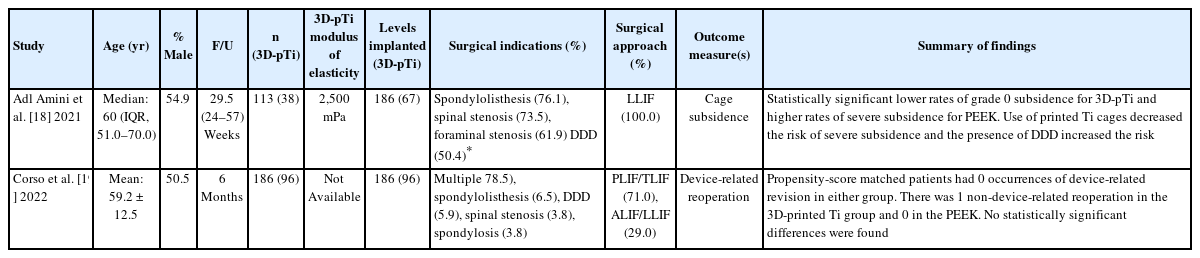
There were 2 retrospective, human cohort studies included (Table 3). Adl Amini et al. [18] included 113 patients (54.9% male) with a median age of 60 years and a mean follow-up of 29.5 weeks. Of the 113 patients, 38 received 3D-pTi implants and 75 received PEEK; 186 total levels were implanted with interbodies with 67 (36.0%) of those being 3D-pTi. Lateral LIF (LLIF) was used for all implantations with the primary structure of the 3D-pTi devices being variations of a grid-like lattice (Table 3). Of note, 3D-pTi cages were more likely to be used during surgeries involving the L1/2 and L2/3 disc spaces (p< 0.001) and for single-level procedures (55.3% vs. 48.0%, p = 0.007). The major outcome measure was cage subsidence with overall subsidence rate for grades I–III under the Marchi classification (0% to 24% collapse of the level) [23] being significantly less for the patient cohort treated with a 3D-pTi device compared to patients treated with a PEEK device (p = 0.003). When stratifying the patient cohort by cases of high-grade subsidence, defined as grade II or III, the 3D-pTi cages still demonstrated a significantly lower subsidence rate when compared to the PEEK cages (3.0% vs. 18.5%, p = 0.002). On multivariate analysis, patients treated with the 3D-pTi were less likely to develop severe subsidence when compared to those treated with PEEK cages (odds ratio, 0.05; 95% confidence interval, 0.01–0.30) (p= 0.001). Furthermore, a diagnosis of degenerative disk disease was also found to independently and significantly predict the development of postoperative severe subsidence (p= 0.034).
Corso et al. [19] included 186 patients (50.5% male) with a mean age of 59.2± 12.5 years and a minimum follow-up of 6 months. Of the 186 patients, 96 received 3D-pTi implants and 90 received PEEK; 186 total levels were implanted with 96 (51.6%) of those consisting of 3D-pTi implants. From a demographic standpoint, patients self-identifying as black and/or African American were more likely to be treated with a 3D-pTi (versus PEEK) device. Additionally, patients who underwent surgery involving implantation of the 3D-pTi device were more likely to have undergone surgery in 2020 as well as to have received treatment at an academic center.
In the present study, the major outcome measure was devicerelated reoperation. One instance of nondevice-related operation was observed in the 3D-pTi cohort, while there were no occurrences of device-related operation in either cohort. The most common surgical indications for 3D-pTi cage implantation included spondylolistheses, spinal stenosis, foraminal stenosis, and degenerative disc disease (Table 3). To control for the confounding effects of demographic variables, Corso et al. [19] also propensity-matched patients, finding that, among matched patients, nonerequired device-related revisions. From these results, Corso et al. [19] conclude that for nondevice-related reoperation events, 3D-pTi cages are associated with minimal risk when compared to non-3D printed cages.
2. Animal Studies
All 3 animal studies were prospective randomized trials employing ovine models (Table 4) [14,20,21]. All 3D-pTi devices used for animal studies used variations of a grid-like lattice structure. Table 4 provides scanning electron microscope illustrations of each device included in the studies analyzed. All known ovine subjects were female. Laratta et al. [14] included 14 ovine models with each receiving 2 levels of implantation, 1 3D-pTi and 1 PEEK. Follow-up was performed at 4 and 8 weeks. From a biomechanical perspective, the PEEK cage had lower static axial compression yield load, expulsion yield load, subsidence yield load and stiffness than the 3D-pTi cage (14,533 vs. 44,002; 517 vs. 620.8; 1,100 vs. 1,120; 13,807 vs. 42,252). The PEEK cage also had a higher Young’s modulus (GPa) than the 3D-pTi cage (3 vs. 2). Outcome measures included micro-CT osseointegration, quantitative histomorphometry of bone-implant contact (BIC) and region of interest (ROI) bone/cartilage. At each follow-up, they found that while 3D-pTi interbody devices showed successful osseointegration into the adjacent vertebrae, PEEK implants demonstrated no such evidence, instead showing only localized fibrotic tissue. Additionally, there was higher mean proportion of bone and cartilage content in the ROI as measured by histomorphometric analyses of the implant region for the 3D-pTi versus the PEEK implants (8,201,364±5,480,486 vs. 1,418,262±2,358,418; p=0.008 and 1,338,943±827,115 vs. 49,496±578,039; p=0.015 for bone and cartilage, respectively). Furthermore, quantitative histomorphometry of implant contact with bone and ROI of bone/cartilage showed significantly higher BIC in the 3D-pTi implants with a value of 39,295.3017±4,414.26266 compared to a BIC of 0 for the sheep treated with PEEK cages (p<0.001).
McGilvray et al. [20] included 27 ovine models with each receiving 2 levels of implantation, with combinations of 3D-pTi, PEEK, or Ti-coated PEEK. Overall, 18 3D-pTi levels were implanted. Follow-up was performed at 8 and 16 weeks. Outcome measures included range of motion (ROM) and stiffness testing, bone volume to total volume ratio (BV/TV) and mean density of bone volume to mean density of total volume ratio (MDBV/MDTV). First, micro-CT scans of multiple planes were taken, specifically of the coronal and midsagittal axes of the 3D-pTi and PEEK cages at 8 and 16 weeks. 3D renderings of these scans displayed significantly higher BV/TV and MDBV/MDTV for the 3D-pTi group at all time points (p < 0.01). Furthermore, there was a significantly increased percent bone for 3D-pTi compared to PEEK at 16 weeks (p= 0.04). Qualitative histological analysis of osseointegration showed higher osteoblast activity, fibrous neovascularization, and bony filling of implant pores in the 3D-pTi group.
Van Horn et al. [21] included 18 ovine models with each receiving 2 levels of implantation, with combinations of 3D-pTi, PEEK, or Ti alloy. Overall, 6 3D-pTi levels were implanted. Follow-up was performed at 6 and 12 weeks. Outcome measures included micro-CT BV quantification and histomorphometric bone apposition ratio (BAR) quantification. At 6 weeks, Van Horn et al. found that the BV was significantly higher in the group treated with the 3D-pTi cage when compared to the PEEK cohort (177.3± 44.1 mm3 vs. 116.9± 43.0 mm3, p= 0.05). However, at 12 weeks, the difference in BV between the 3D-pTi cohort and the PEEK cohort became insignificant (234.7± 35.9 mm3 vs. 218.8±21.8 mm3, p>0.05). Despite comparable BV among PEEK and 3D-pTi at 12 weeks, the BAR for the 3D-pTi cages were 2.7 times higher than the PEEK cages, reaching statistical significance (23.6%± 10.9% vs. 8.6%± 2.1%, p< 0.05). A similar result was found at 12 weeks with the BAR for the 3D-pTi cages being 2.6 times higher than the PEEK cages, also reaching statistical significance (36.5%± 10.9% vs. 14.0%± 5.0%, p< 0.05). Importantly, these results indicate greater integration of the 3D-pTI device into bony structure through measurement in the amount of bone inside the cage’s pores.
3. Nonclinical Studies
Among the nonclinical studies identified during our search, 2 examined bone porosity and 1 utilized FEA to measure compression, tension, and shear forces between implants [16], with the other utilizing an in vitro hMSC and extracellular matrix (ECM) model to measure cell proliferation and as well as differentiation amid the implant environment [22]. As was consistent with the studies on animal models, all 3D-pTi devices used in nonclinical used variations of a porous structure. Table 5 provides an illustration of the chosen lattice structures for of the included studies.
Carpenter et al. [16] aimed to analyze the load-bearing potential of the 3D-pTi and PEEK cages in order to determine differences in biomechanical functionality between the 2 devices. To accomplish this task, finite element (FE) models of the interaction between the pore structure and bone were used with observations collected at 4 and 28 weeks. Notably, the PEEK cages had increased load share transferred to the bone grown within the device (66% vs. 13%), tension (71% vs. 12%), and shear (68% vs. 9%) when compared to the 3D-pTi device (p< 0.05). Further, the 3D-pTi device had reduced spinal load magnitudes both at 4 and 12 weeks when compared with the PEEK device (4 weeks: 180 ± 300 microstrain vs. 784 ± 351 microstrain; 12 weeks: 121± 49 microstrain vs. 298± 88 microstrain). Overall, this resulted in 29.5% increased load distribution in 3D-pTi compared to 15.8% of porous PEEK.
Papaefstathiou et al. [22] performed an in vitro study comparing 3D-pTi to PEEK with observations collected at 7, 14, 21, 28, and 42 days. The porosity of the 3D-pTi implants used ranged from 70%–75%. Results showed 3D-pTi forming a dense layer of intermixed hMSC and ECM by day 28. For PEEK implants, cells were sparse on day 7 but multilayers were formed at day 42. Analysis of proliferation demonstrated 3D-pTi surfaces to have significantly higher cell number at all time points. Differentiation analysis showed increased ALP activity and calcium content per cell, although no significant differences were observed.
DISCUSSION
The 3 advantages of LIF relative to conventional posterolateral fusion are its ability to improve sagittal balance, facilitate indirect decompression of the lumbosacral neural elements through disc-space distraction, and provide an additional surface for fusion. Conventionally 2 materials have dominated the market: Ti alloy and PEEK. Both Ti alloy and PEEK devices first became widely available on the market in the late 1990s. Ti interbodies possess the advantage of being osteoinductive and are known to demonstrate high levels of osseointegration with native bone. However, these devices are radiopaque, which can limit radiographic assessment of fusion during follow-up [11]. Additionally, Ti alloy has an elastic modulus that far exceeds that of cancellous bone. While this offers potentially greater mechanical stiffness at the time of implantation, it also increases the risk of implant subsidence [8]. Though subsidence does not always necessitate operative revision, it is associated with loss of sagittal plane correction and neuroforaminal height loss, potentially counteracting 2 of the desired outcomes of interbody fusion. By comparison, PEEK has a much lower elastic modulus and an accordingly lower risk of implant subsidence. It is also radiolucent and can thereby better facilitate radiographic follow-up [9,12]. However, PEEK is largely chemically inert and demonstrates poor osseointegration secondary to its proclivity to form biofilms [24]. Ti-coated PEEK implants have been developed in an attempt to combine the advantages of both materials, but these hybrid devices do not appear to reduce subsidence risk relative to Ti implants [13] and are associated with the risk of surface coating delamination during implantation [25].
In this review, data from animal studies, human clinical series, and ex vivo experimental studies comparing PEEK and 3D-printed Ti interbody devices is presented. The data, while limited and primarily derived from animal and nonclinical studies, suggest that 3D-pTi devices are associated with decreased subsidence, increased osseointegration on micro-CT as well as qualitative histological analysis, increased BIC and ROI bone-to-cartilage ratio, increased ROM in all directions, increased stiffness, increased BV/TV, increased BAR, increased energy effective strain in the adjacent bony layer, increased inimplant cell proliferation, and favorable hMSC morphology as it relates to bony fusion. Taken together, 3D-pTi outperforms PEEK in 11 of the 14 outcomes. It is equivalent to PEEK in device-related reoperation, BV quantification at 12 weeks and cellular differentiation around the implant as measured by ALP and calcium content.
Advances in 3D printing technologies and materials science in the past 5–10 years have led to the availability of new 3Dprinted Ti interbodies [26-29]. 3D-pTi devices are designed to imitate trabecular bone with highly porous surfaces, which both facilitates bony ingrowth [30] and lowers the elastic modulus to the point that it more closely emulates that of cancellous bone [4,20,31-33]. 3D-pTi implants also have greater radiolucency relative to conventional Ti implants and so may allow for more accurate assessment of bony union [27]. However, it must be noted that not all 3D-printed cages are the same, as cage properties vary significantly with the porosity of the printed Ti. Notably, while highly porous devices can allow for greater load sharing [30], porosities greater than 70% may become detrimental to the point that they can compromise structural integrity [16]. Although high porosity may increase the load sharing capacity of 3D-pTi implants until it nearly approximates the load sharing properties exhibited by PEEK, below this critical threshold in-implant mass-to-volume ratio, intra-pore struts become thinner and thus, more vulnerable to degradation and buckling [16,34].
As suggested above, the elastic modulus of 3D-pTi cages directly correlates with the degree of porosity [16]. In one study, reducing the E (elastic modulus) of Ti from 100,000 MPa to 2,500 MPa through porous 3D modeling, demonstrated a similar elastic modulus to human bone (15,000–20,000 MPa) [4,18]. Prior FEA of PEEK implants, which have an elastic modulus comparable to cancellous bone, have shown lower von Mises stresses at the endplates of implanted interbodies in FEA relative to conventional Ti implants [9]. This same advantage should therefore be possessed by modern 3D-pTi interbody devices. Indeed, this was shown clinically by Adl Amini et al. [35] in their clinical series of 113 patients who underwent LLIF with 3D-pTi cages. Theoretically with modern 3D-printing technologies, the elastic modulus of the implant could be customized to the patient’s underlying bone density to allow for optimal implant-patient matching. However, the cost-effectiveness of such a solution may be infeasible at present.
Additionally, with respect to clinical outcomes, Corso et al. [19] reported no occurrence of 3D-pTi device-related revision surgeries within 6 months of the initial fusion procedure. For comparison, the randomized controlled trial conducted by Kersten et al. [36] showed an additional procedure rate of 2.1% within 6 months of PEEK interbody device implantation in transforaminal LIF (TLIF) procedures. Retreatment indications for corrective decompression of implanted PEEK cages included an adjacent level complication or pseudarthrosis. Revision surgery is also recommend at higher rates when PEEK cages are used (3 out of 24 patients, 12.5%) as compared to when 3D-pTi cages are used (revision was not recommended to any patient) [18].
Three-dimensional-printed Ti implants can also undergo surface treatment (e.g., adsorbed hydroxyapatite coating) to better facilitate bony ingrowth [37,38]. Plasma spraying can be used to adjoin hydroxyapatite and Ti at the molecular level [39]. This amalgam maintains the stiffness of the Ti while exploiting the osseointegrative properties of hydroxyapatite [40] to create a balanced implant not unlike 3D-pTi. While this is attractive in theory, studies have only demonstrated this coating’s effectiveness in Ti pedicle screw stability [41,42] and not yet in interbody spacer outcomes [39]. Previous literature shows that reductions in stress due to the creation of a highly porous environment increases the compressive shear strength [37,43,44]. Krafft et al. [38] created a novel 3D-pTi device, implanted in minimally invasive LLIF, with cage subsidence rates reported at 3.4% per implant, a rate lower than traditionally-used static PEEK cages (ranking from 10%–16.1%). There were specifically 2 cases of subsidence out of 59 interbodies implanted, both occurring within 4-level constructs and both resulting in asymptomatic outcomes not necessitating revision.
Histomorphological studies support the use of 3D-pTi implants over PEEK implants. 3D-pTi implants demonstrate robust osseointegration ingrowth as compared to PEEK implants, which show no demonstrable osseointegration in animal models [14]. Noiset et al. [12] demonstrated that PEEK lacks the ability to osseointegrate circumferentially around the cage and instead forms fibrous bony ingrowth. By contrast, 3D-pTi exhibits growth in a uniform manner due to the octahedral lattice configuration of the implant [45]. The porous texture of the 3D-pTi cage appears integral to its ability to promote osteogenesis through. The porous structure promotes de novo bone growth, remodeling, and capillary formation [46]. In vitro work by Olivares et al. [47] suggests that this may result from increased bone morphogenetic protein 2 expression on microtextured Ti alloy. Stimulation and promotion of bone growth and remodeling most likely enhances the 3D-pTi implant stability and fusion. The cellular properties found at the micro scale in 3D-pTi cages also contribute to the overall improved osseointegration. Increased friction related from the porous surface of 3D-pTi cages has been reported to increase cell adhesion for bony ingrowth [20]. Formation of F-actin filament highways in hMSCs caused improvements to cellular adhesion compared to PEEK and 2D-Ti scaffolds [22]. In addition, this mechanotransduction has been reported at the macroscopic level in ex vivo loading 3D spinal implants as well [48].
Increased bone-to-cage contact may also promote formation of an osteoblastic environment that promotes 3D-pTi cage-mediated fusion. Van Horn et al. [21] recorded an increased fusion-related bone volume within the 3D-pTi device compared to PEEK. This increased bony surface apposition, understood as a surface area to volume ratio, was noted to increase steadily during weeks 6 to 12 in subjects with 3D-pTi devices [21]. Increased bony contact with the porous surface of the 3D printed device has been shown to generate reductions in ROM, furthering stabilizing of the 3D-pTi device [20].
Notably, several other demographic and surgery-related factors have been associated with the functional and clinical outcomes described in the studies included in this review. A study from Zavras et al. [49] reviewing 144 patients who underwent anterior LIF (ALIF) found that older age, higher body mass index, patient frailty as quantified by the American Society of Anesthesiologists physical status classification grade, and a prior diagnosis of osteoporosis were each factors associated with increased subsidence. Similar results were described by Phan et al. [50] who found a positive association between obesity and increased rates of subsidence amongst patients who underwent ALIF. Regarding patient surgical characteristics, Peng et al. [51] in a review of 32 patients undergoing ALIF, found that lower intraoperative pressure and complete removal of the intraoperative plate was associated with significantly reduced cage retropulsion. Building on these results, Yao et al. [52], in a retrospective review of 93 TLIF patients, found that anterior cage positioning reduced subsidence. Several other studies have described similar outcomes in spinal fusion patient cohorts in regards to the geometry of device integration. Regarding the present review, only 2 of the included studies analyzed human cohorts. On multivariate analysis, Adl Amini et al. [18] found only 2 other demographic or surgical factors that were a significant predictor of subsidence: diagnosis of degenerative disk disease and volumetric bone density. Corso et al. [19] performed a propensity-matched study, eliminating the potential for confounding demographic variables to influence the final outcomes. However, as a result of cohort matching, they were unable to elucidate relationships between patient demographics, surgery-related factors and functional outcomes.
There are several limitations to the present study. Foremost is the relatively limited number of studies available providing direct comparison of 3D-pTi and PEEK interbody for LIF. Moreover, a majority of the studies included in the present review were animal or nonclinical studies, limiting the ability to reach broad conclusions about the advantaged of 3D-pTi, though the preliminary evidence here appears to support its use over PEEK implants. Notably, none of the included studies included comparison between variations on the LLIF implants, namely ALIF devices, TLIF devices and posterior LIF devices, which may also have applications in clinical workflows. Deeper investigation into the generalizability of the highlighted results for these variations is warranted though previous reviews of literature have found that indications for the interbody fusion device used are contingent on anatomy rather than material used. As a result, we would not expect significantly different comparative outcomes for the device variations. Further, we were unable to compare demographic and surgery-related factors across studies although multivariate and cohort matching within studies identified independent relationships between device materials and functional outcomes. Additionally, each of the studies analyzed used different metrics for device characterization, such as young’s modulus, and efficacy, such as subsidence rates and patient-driven outcomes, rendering comparison across studies, even in similar populations, difficult. Future studies may look to develop a standardized set of comparison tools for interbody devices.
Furthermore, additional analysis on the cost-effectiveness of these implants is merited. Crucial to development of a potential cost-effectiveness argument in favor of 3D-pTi, is the elimination of osteobiologics and their cost burden to a successful fusion surgery, owing to the superior osteointegrative character of 3D-pTi relative to standard interbodies. A most cost-effective solution may lie somewhere in between; Malone and colleagues [53] utilized a 3D-pTi coated with a low cost β-tricalcium phosphate-hydroxyapatite ceramic graft in a retrospective series of 90 patients undergoing lateral lumbar fusion surgery and demonstrated successful fusion criteria in 99.3% of patients at 1-year follow-up without any instance of revision surgery or high-grade subsidence. Future studies investigating the cost-effectiveness of this implant must consider how it may obviate standard osteobiologics for achieving fusion. An additional limitation is the utilization of different 3D-pTi implants in each study. As demonstrated by prior biomechanical data, implant porosity significantly affects the elastic modulus and other biomechanical properties, along with the ability to facilitate osseointegration. The exact impact on the clinical effectiveness of these devices is unclear. Further, none of the included study investigated the influence of unique lattice structures on-device efficacy. Future studies may look to analyze whether implant porosity and structure significantly impact device outcomes. moreover, none of the included clinical studies reported greater than 1-year follow-up. As bony union can take more than a year, it is possible that fusion rates at later follow-up time points do not show a difference between 3D-pTi and PEEK implants, though the histopathological data support 3D-pTi as superior. Finally, the studies included in this review used devices made of 3D-pTi devices and commonly available PEEK materials. Since these studies have concluded, advancements in PEEK technology, such as addition of inorganic and organic phases, different minerals and modifications to structure and roughness, have occurred and future efforts should therefore compare 3D-pTi technology to these more modern, modified PEEK materials.
CONCLUSION
The present systematic review highlights the paucity of clinical data directly comparing outcomes between 3D-printed Ti implants and PEEK implants for LIF The clinical and preclinical data that is available supports the superiority of 3D-pTi in terms of fusion outcomes and osseointegration. The available clinical series additionally showed decreased rates of cage subsidence and no reports of reoperations or revision surgeries. Despite the promising preliminary data, additional human investigation, including cost analyses are merited.
Notes
Conflict of Interest
Dr. Pham reports consultant fees with Medtronic and Thompson Surgical. The other authors have nothing to disclose.
Funding/Support
This study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author Contribution
Conceptualization: NAP, NJB, ZAP, NBH, CK; Data curation: NAP, SO’B, CDR; Formal analysis: NAP, SO’B, CDR, JG; Methodology: NAP, SO’B, CDR, JG, NBH; Project administration: NAP, ZAP, LDDA, MHP; Visualization: NAP, JG, NJB, ZAP, NBH; Writing - original draft: NAP, SO’B, CDR, NBH, CKB, JG; Writing - review & editing: NAP, SO’B, CDR, CKB, MHP, SC.