Proximal Junctional Kyphosis or Failure After Adult Spinal Deformity Surgery - Review of Risk Factors and Its Prevention
Article information
Abstract
Proximal junction kyphosis (PJK) is a common imaging finding after long-level fusion, and proximal junctional failure (PJF) is an aggravated form of the progressive disease spectrum of PJK. This includes vertebral fracture of upper instrumented vertebra (UIV) or UIV+1, instability between UIV and UIV+1, neurological deterioration requiring surgery. Many studies have reported on PJK and PJF after long segment instrumentation for adult spinal deformity (ASD). In particular, for spine deformity surgeons, risk factors and prevention strategies of PJK and PJF are very important to minimize reoperation. Therefore, this review aims to help reduce the occurrence of PJK and PJF by updating the latest contents of PJK and PJF by 2023, focusing on the risk factors and prevention strategies of PJK and PJF. We conducted a search on multiple database for articles published until February 2023 using the search keywords “proximal junctional kyphosis,” “proximal junctional failure,” “proximal junctional disease,” and “adult spinal deformity.” Finally, 103 papers were included in this study. Numerous factors have been suggested as potential risks for the development of PJK and PJF, including a high body mass index, inadequate postoperative sagittal balance and overcorrection, advanced age, pelvic instrumentation, and osteoporosis. Recently, with the increasing elderly population, sarcopenia has been emphasized. The quality and quantity of muscle in the surgical site have been suggested as new risk factor. Therefore, spine surgeon should understand the pathophysiology of PJK and PJF, as well as individual risk factors, in order to develop appropriate prevention strategies for each patient.
INTRODUCTION
Adult spinal deformity (ASD) is a broad spectrum of alignment abnormalities of the lumbar spine or the thoracolumbar spine, including primary degenerative sagittal imbalance, iatrogenic spinal deformity, and spinal scoliosis [1,2]. As the elderly population increases due to the recent improvement in medical quality and an increase in life expectancy, the prevalence of ASD is increasing to such an extent that some paper reports that the frequency of ASD in adults aged 60 years or older is up to 68% [3]. Patients with ASD suffer from severe back pain, radiculopathy, gait disturbance, and disability due to compression of the nerve root and spinal cord caused by deformation of the coronal and sagittal alignment, and the quality of life is decreases. These symptoms are an important factor in determining correction surgery [4,5]. Correction surgery using long segment instrumentation for ASD requires high skill among spine surgeries. Therefore, it is mainly performed by experienced spine surgeons. Nevertheless, the overall complication rate of correction surgery for ASD is 13%, and the revision rate in long term follow-up is about 9% [6,7]. Proximal junctional kyphosis (PJK) and proximal junction failure (PJF) are common complications that occur in about 46% of cases after correction surgery of ASD [8-10], but revision surgery is required in severe cases of PJK and PJF. Additional revision surgery in patients who have undergone a long segmentation instrument for ASD can be another disaster. Therefore, in order to reduce the incidence of PJK and PJF, this paper focuses on the risk factors and prevention of PJK and PJF after correction surgery of ASD, and aims to provide overall information about PJK and PJF through current literature review. Based on this, it is hoped that it will be helpful to establish prevention strategies for PJK and PJF.
MATERIALS AND METHODS
We conducted a search across multiple database, including PubMed, Embase, CENTRAL, and Web of Science for papers published by February 2023. The search keywords were “proximal junctional kyphosis,” “proximal junctional failure,” “proximal junctional disease,” and “adult spinal deformity.” In this study, a search strategy was employed using keywords, medical subject headings (MeSH) terms alone, or a combination of both. The MeSH terms included “kyphosis” and “spinal diseases” used. 438 papers in PebMed were identified, 425 articles in Web of Science, 292 in Embase, and 18 in CENTRAL. A total of 1,173 articles were identified, of which 412 were duplicate articles. Papers written in languages other than English, and case reports & articles were excluded (n=356), and studies not focusing on PJK and PJF were also excluded from this study (n=302). Finally, 103 articles were included in this review. The details of this process are highlighted in Fig. 1. Each article title and/or abstract was screened for relevance by 2 reviewers. Subsequently, the same reviewers conducted a full-text review of the relevant articles and determined whether each article satisfied the criteria for inclusion.
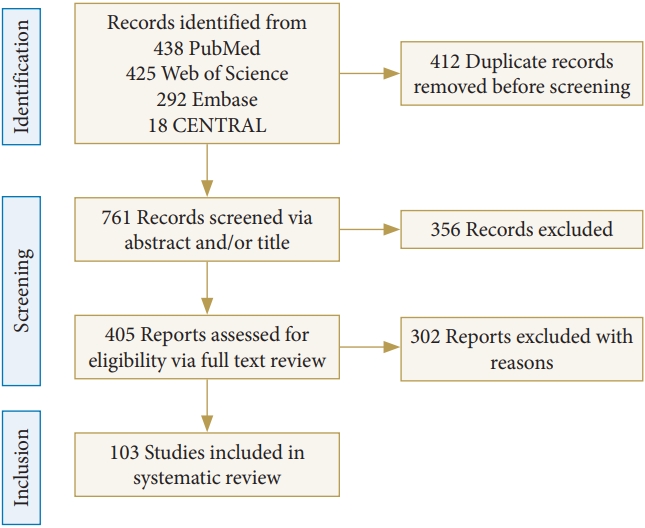
Flow diagram (PRISMA format) of the screening and selection process of proximal junctional kyphosis or failure after adult spinal deformity surgery. PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
1. Definition, Pathophysiology, Incidence, Classification
PJK is a complication that commonly occurs in the uppermost instrumented vertebra of long-level instruments and indicates the presence of abnormal kyphosis at that level. There are various methods of measuring PJK. The most common way to measure PJK is to measure the sagittal Cobbs angle between the inferior endplate of the upper instrumented vertebra (UIV) and the superior endplate of the vertebra 2 levels above the UIV [2,11,12]. This method is mainly used because of its high reproducibility with intraclass and interclass correlations ranging from 0.78 to 0.92 and 0.55 to 0.80 [13]. In 1989, Bernhardt and Bridwell [14] reported on the normal sagittal alignment of thoracic kyphosis, thoracolumbar junction, and lumbar lordosis (LL) in normal individuals. Then, in 1999, Lee et al. [15] reported abnormal kyphotic angles of the proximal level of instrumented fusion of at least 5 degrees during a follow-up period of at least 2 years after posterior spinal fusion surgery. Subsequently, based on the study findings that there should be a difference of at least 11 degrees in radiologic measurements after long-level fusion to have 95% confidence in kyphosis, Glattes et al. [16] redefined the angle of PJK to be greater than 10°. To date, it is most commonly used to define PJK as an increase in the angulation of the PJK by at least 10° greater than the preoperative measurement in literature [2,11,17-19]. Despite numerous studies, no consensus has been reached on the optimal PJK angle after adult spinal deformity surgery, with many studies proposing a critical cutoff value of either 15° or 20° [20-22]. PJK is commonly seen in fusion surgery, and there is much controversy about its clinical significance, as it rarely results in significant neurological outcomes or requires revision surgery. Some studies consider PJK to be a simple radiological finding, as there is no difference in its association with clinical symptoms regardless of its presence or absence [2,16,22]. PJK is a progressive condition that has been observed to continue even after 2 years following surgery [23,24]. PJF refers to the worsened form of the PJK disease spectrum and is characterized by mechanical failure and/or spinal instability at the UIV or UIV+1, including vertebral fracture, subluxation, and fixation failure. This can cause pain, neurological deficits, and may require revision surgery in up to 47% of affected patients [11,18,21,23,25]. PJK and PJF are not separate diseases but rather included in one disease spectrum. In analogy to tumor, depending on the severity of the disease, they can be classified into benign PJK without significant clinical relevance and malignant PJF with important clinical significance (Fig. 2).
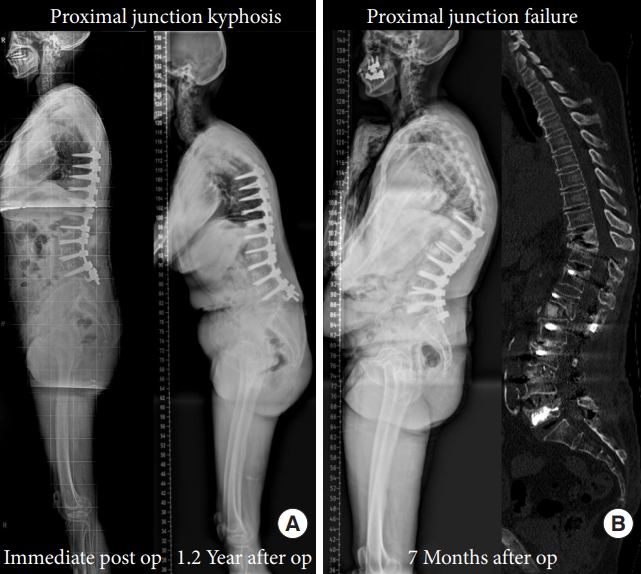
Whole spine lateral x-ray and computed tomography image of proximal junctional kyphosis (A) and failure (B).
The pathophysiology of PJK and PJF may differ slightly. PJK is believed to occur due to partial damage to the posterior tension band near the UIV during long-level instrumentation surgery, as well as the action of deformity correction force during surgery and compensation for reduced kyphosis in the thoracic region [18,26]. This kyphosis, which is thus induced or compensated, increases loading near the UIV. In addition, biomechanically, long-level instrumentation induces stress increase near the UIV due to the long lever arm and nonphysiological center of motion. This causes force to occur near the UIV, and motion near the UIV increases to distribute this force. As a result, degenerative changes progress more rapidly near the UIV, and vertebral bodies that cannot withstand the force may also experience fractures and instrumentation failure, leading to PJF [27-29].
The incidence of PJK varies widely due to various factors such as its definition, follow-up period, and characteristics of the patient population. Many studies report the incidence of PJK to be within 20%–40% [2,12,23,30,31]. According to a recent paper published in Korea, among 78 ASD surgical patients who were followed up for more than 2 years from January 2012 to December 2017, 25 patients (32.1%) were reported to have developed PJK [32]. In contrast, the incidence of PJF is reported to be lower than that of PJK, ranging from 1.4% to 35% [21,33,34]. It is important to know not only the incidence of PJK and PJF, but also the timing of their occurrence. PJK is usually diagnosed within 1 year after surgery, and when PJK occurs, approximately 53% of PJK progression angle occurs within 3 months of occurrence [23]. In other study, it has been reported that approximately 59% of PJK progression angle occurs within 8 weeks of occurrence [24]. Although PJF is an aggravated finding of PJK, PJF progresses rapidly. The average time for revision surgery with PJF was 10 months, and 87% of these cases underwent revision within 2 years [34]. Similarly, in another study, PJF was diagnosed within an average of 3 months after surgery and revision surgery was performed within 7 months [21].
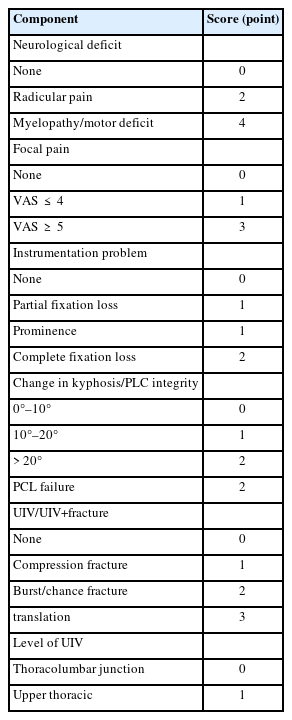
There have been few studies on the classification of PJK. Yagi et al. [23] classified PJK based on type and grade, and spondylolisthesis. This method provided a simple and easy means of communicating PJK type among clinicians, but had a major drawback in that it did not provide decision criteria for management, including revision surgery [11]. To address this issue, Hart et al. [25] and the International Spine Study Group (ISSG) classified PJK and provided decision criteria for management. They used a PJK severity scale consisting of 6 components, including neurological deficit, focal pain, instrumentation problem, change in kyphosis/posterior ligament complex integrity, fracture location, and level of UIV. This scale had good reliability and repeatability, and its score showed a strong correlation with clinical outcomes and indication for revision surgery. A score of 7 or higher was recommended for revision surgery (Table 1).
2. Risk Factors and Prevention
PJK and PJF can be caused by various factors, but can be broadly classified into 3 categories: patient-related, radiographic, and surgical risk factors.
1) Patient-related risk factors
Patient-related risk factors include old age, osteoporosis, high body mass index (BMI) and sarcopenia. Many studies have reported old age as a risk factor for PJK after long-level instrumentation in adult spinal deformity. While age of 50 or older is often mentioned as a risk factor, other studies have also reported age of 60 or older as a risk factor [11,22,24,35]. Along with advanced age, important risk factors for PJK/PJF are osteoporosis and low bone mineral density (BMD) [11,23,34]. Patients with low BMD, including osteoporosis and osteopenia, have a 30.9% increased risk of developing PJK compared to those with normal BMD. Furthermore, osteoporosis and age over 60 are considered as risk factors for PJK [23,36]. Mikula et al. [37] measured the bone density of UIV and UIV+1 with Hounsfield units (HU) using computed tomography and reported that low HU was the risk factor for PJK and PJF with an optimal cutoff of 159 HU. Moreover, it has recently been reported that high vertebral body quality (VBQ) is related to the occurrence of PJK/PJF by measuring bone quality with MRI-based VBQ scoring [38]. Yagi et al. compared the changes in average BMD and the incidence of PJK between the postoperative teriparatide (TP) group and control group among osteopenia patients who underwent long segment fusion with ASD. Hip BMD increased by 6.7%+4.9% in the TP group, but decreased by 1.4%+4.2% in the control group at 2 years follow-up. The incidence of PJK type 2 was also significantly lower in the TP group (4.6%) than in the control group (15.2%, p=0.02) [39,40]. There are studies that have applied cement augmentation to prevent proximal junctional complications in patients with osteoporosis. In one study, prophylactic vertebroplasty was performed on UIV and UIV+1, resulting in a decrease of PJK and PJF by 8% and 5%, respectively, at 6 months follow-up, but at 5-year follow-up, the incidence of PJK was not significantly different (28.2%) [41]. Another study reported no junctional complications during the follow-up period after cement augmentation at all instrumented levels and UIV+1 [42]. There is currently no guideline that determines up to which level prophylactic cement augmentation should be performed for PJK and PJF. However, considering the changes in biomechanical loading, shearing, and vertebral body/disc nutrition due to cement augmentation, many authors recommend performing cement augmentation at the UIV and/or UIV+1 [43-46]. Bridwell et al. [22] reported that high BMI (median, 25.6 kg/m2) and the presence of comorbidities are related to the development of PJK of 20° or more. However, other studies have explained that BMI is not related to PJK/PJF [47]. Recently, the importance of muscles has been emphasized, and the concept of sarcopenia has been introduced. Furthermore, many studies have reported the association between paraspinal muscles and the risk of PJK. In 2016, Hyun et al. [48] proposed that lower thoracolumbar muscularity and higher fatty degeneration could induce PJK. Subsequently, Pennington et al.49 measured the cross-sectional area of paraspinal muscles at the UIV and reported that smaller paraspinal musculature at the UIV strongly predicted PJK. Recently, the emphasis has shifted to the quality of muscles, and a total muscle-fat index of paraspinal extensor muscles > 4.08 has been reported to be independently related to PJK [50]. Moreover, the importance of multifidus among paraspinal muscles has been emphasized, and the importance of bone quality, along with muscle quality, has also been integrated. Lower HUs and severe multifidus sarcopenia at the UIV are proposed as independent predictors of PJK and PJF [10]. Therefore, the state of muscle as well as bone should be evaluated before long-level instrumentation in ASD patients. In addition, the concept of frailty was also presented. Frailty is a new medical concept that represents an individual’s vulnerability to external injury due to physiological age increase and physiological function decrease [51]. Health deteriorates at varying rates among individuals, and from a medical perspective, there is a discrepancy between physiological age and chronological age. Assessment of frailty has been developed as a method to quantify an individual’s physiological age [51,52]. Recently, these frailty indices have shown good results in predicting complications after surgery [53,54]. In a study, the researchers utilized the ISSG ASD prospective patient database to develop the adult spinal deformity frailty index (ASD-FI). They reported that patients with greater frailty measured by ASD-FI had a higher incidence of major complications such as PJK and were associated with worse outcomes [51,55]. Recently, the Frailty-Adjusted Realignment Score was proposed by combining the ASD-FI and GAP scores, and it was reported to be useful in minimizing the risk of mechanical complications [56].
2) Radiographic risk factors
After undergoing deformity surgery, if the load distribution is not stabilized in the implants and spine alignment, a compensation mechanism occurs. Therefore, spine surgeons should perform instrumented fusion to minimize the occurrence of compensation mechanisms following surgery [57]. Malalignment of the thoracolumbar spine can cause compensatory changes, including cervical hyperlordosis, pelvic shift, knee flexion, hip extension, and pelvic retroversion. Cervical alignment can be influenced by thoracic alignment, and there is a correlation between global sagittal alignment and reciprocal changes with the magnitude of sagittal deformities [58,59]. T1 slope is associated with cervical lordosis and C7 sagittal vertical axis (SVA), and changes in each parameter can influence the others. Cervical lordosis increases after greater iatrogenic reduction in thoracolumbar deformity surgery [60]. There is a reciprocal relationship between LL and thoracic kyphosis. Thoracic kyphosis and pelvic retroversion can be corrected after restoration of LL [61]. The magnitude of the T1 pelvis angle is strongly associated with reciprocal changes in knee angle, hip extension, and posterior pelvic shift. If sagittal malalignment is corrected, posterior pelvic shift, knee flexion, and hip extension are also corrected [62]. Therefore, an important factor in the occurrence of PJK and PJF is the presence of radiographic abnormalities of global spinal alignment. As performance of spine surgery involves artificially fixing a dynamic spine structure, it is crucial to balance the loading distribution of the inherent gravitational force of the spine both before and after the surgery. Many radiological risk factors have been reported for PJK and PJF, which can be classified into preoperative and postoperative factors [63]. Maruo et al. [64] reported that preoperative thoracic kyphosis (TK) of 30° or more is an important predictive factor for PJK, and a meta-analysis study identified TK > 40° as a risk factor for PJK [23,65]. Additionally, other studies have reported that preoperative proximal junctional angle of 10° or more and pelvic incidence > 55° are associated with a higher risk of PJK [64]. Regarding the occurrence of PJK and PJF, several studies have emphasized the changes in spinal alignment after surgery. Yagi et al. [23] reported that postoperative changes in SVA of 50 mm or more are related to increased incidence of PJK. Another study showed that postoperative SVA of less than 50 mm is significantly associated with a risk of PJF [33]. Furthermore, a change in LL of 30° or more before and after surgery has also been reported as a risk factor for the occurrence of PJK [64].
The Scoliosis Research Society (SRS)-Schwab classification suggested that the targets for obtaining satisfactory alignment and good clinical outcome were pelvic incidence-LL (PI-LL) <±10°, pelvic tilt < 20°, and SVA < 40 mm [66-68]. However, it was not clear how achieving Schwab goal affected the occurrence of mechanical complications, and mechanical complications were common despite achieving the ideal correction of the Schwab criteria. One study reported that Implant-related complications occurred in 31.7%, even though Schwab’s target value was achieved, and 52.6% of them required reoperation [69]. In addition, the Schwab criteria were simply measured with radiologic curves, angles, and distant parameters, and the definitions of “normal” and “pathologic” alignment were unclear. These radiologic parameters are purely descriptive rather than analytic. So, correlations with these parameters do not necessarily imply causation. When using SVA, PI-LL, and pelvic tilt, using only the linear numerical value without considering the correlation of each parameter can lead to misleading, especially in patients whose PI value is close to the upper-normal or lower normal limit [57]. Examples of causing misleading in each parameter are as follows. Patients with higher PI may have higher PT and SS, and higher PT do not necessarily imply increased retroversion and disability. A pelvic tilt of > 20° can be interpreted as a pathologic condition under the Schwab criteria, but may be an anatomical characteristic or reflect a large PI. So, the ideal target of pelvic tilt < 20° cannot be applied to all types of PI. A lower PT may be required in patients with low PI, but pelvic tilt < 20° in patients with high PI may be disabling. Ultimately, in the Schwab criteria, PT did not consider anteversion, which could be the cause of mechanical failure. PI-LL <±10 has limitations when applied to patients with extreme PI values. Additionally, incorrect load distribution during surgical correction of LL can lead to mechanical failure, so the lordosis distribution in the lower (L4–S1) and upper (L1–3) lumbar spine which is important for changing the load distribution should be considered [70]. However, the Schwab criteria did not take this into account. SVA < 40 mm can be masked by compensatory pelvic retroversion without considering negative malalignment, which is a potential cause of mechanical failure [57,70,71]. In order to overcome these limitations of the Schwab criteria, a new method has been proposed for predicting mechanical complications after surgery in ASD patients called the global alignment and proportion (GAP) score. Since PI is a relatively constant parameter, other parameters can be evaluated in relation to PI. Therefore, GAP score uses PI-based proportional parameter rather than absolute numeric. Furthermore, the GAP score indicates the standing sagittal alignment and shape as either “normal” or “pathological” for every magnitude of PI [57]. This involves considering each individual’s ideal global spinopelvic alignment, considering appropriate alignment goals based on the complexity of their spinal structure. Measurements such as relative LL, relative pelvic version, relative spinopelvic alignment, lordosis distribution index, and age factor are used to score postoperative alignment characteristics and classify them into proportionate, moderately disproportionate, and severely disproportionate categories [57,72]. This classification demonstrated that patients with proportionate alignment had mechanical complication rates of 6%, moderate disproportion in 47%, and severe disproportionate alignment in 95% [57]. Therefore, understanding the GAP score within the current concept of spine balance is important for preventing PJK. However, external validation of the GAP score for the association between proportional alignment categories and mechanical failure has not been consistent across studies [73]. Furthermore, since this method was developed and analyzed postoperatively, it is difficult to apply it reliably before surgery. Therefore, there are still differing opinions on whether the GAP score reflects mechanical failure or not [74]. Recently, in order to improve the predictive ability of mechanical complication, Modified GAP scoring with BMI and BMD (GAPB), which combines BMI and BMD with the GAP score, has been proposed [75]. Furthermore, Noh et al. [76] reported that they further improved the predictive ability of mechanical complications using machine learning based on GAPB.
With the increase in the elderly population, there has been a growing discussion regarding the definition of ideal sagittal balance in elderly patients. As age increases, LL tends to decrease with an increase in pelvic tilt to maintain whole sagittal balance following lumbar spine degeneration [77]. Roussouly et al. [78] classified the spine shape of normal adults into 5 types and presented an algorithm for restoring sagittal alignment, considering the association between pelvic incidence and lumbar degenerative changes. Degenerative loss in LL shifts the gravity line forward, and consequently compensatory mechanisms such as pelvic retroversion, knee flexion, and ankle extension occur to shift the gravity line posteriorly, resulting in posterior pelvic shift [79]. Due to these reasons, sagittal parameters in the normal population can vary with aging. The ideal target of LL relative to PI may be lower in elderly patients. Therefore, age-adjusted targets for sagittal alignment have been proposed [80]. In a study, it was reported that despite undergoing deformity surgery based on the previous target in elderly patients, there was a high incidence of PJK. This was attributed to overcorrection according to the age-adjusted target. Additionally, it was found that overcorrection was more commonly observed in the smaller PI group, which increased the occurrence of PJK. Therefore, while achieving sufficient correction is crucial for patients with high PI. Spine surgeons should be cautious about overcorrection in elderly patients with lower PI due to the high correlation with PJK [32]. Lafage et al. [81] reported effective prediction of PJK and surgical outcome through sagittal age-adjusted score considering inspired by SRS classification, the concept of the GAP score, and age-adjusted alignment targets. However, there is no universal target for spinopelvic measurement that considers all of these yet.
3) Surgical risk factors
It is also very important for spine surgeons to consider the surgical aspect in predicting and preventing PJK and PJF. The surgical risk factors of PJK and PJF include the following; Selection of UIV/lowest instrumented vertebra (LIV), surgical approach, proximal implant construction, rod characteristics, and posterior soft tissue (muscle, posterior ligamentous complex [PLC], facet capsules) injury. Choosing an appropriate UIV can be a challenging issue. In general, neutral and stable vertebrae are chosen as UIV [82]. Some studies have reported a lower incidence of PJF when the UIV is located in the upper thoracic (UT) region compared to the lower thoracic (LT), thoracolumbar, or lumbar region [21,36]. However, the UIV in the UT region can lead to PJK caused by subluxation and soft tissue injury, while the UIV in the LT region can lead to PJK and secondary vertebral body fractures [21,64]. Since thoracic hyperkyphosis is a risk factor for proximal junctional problems, fusion extended to the UT level in patients with thoracic hyperkyphosis can reduce the occurrence of PJK and PJF [43,83]. Additionally, several studies have reported that long-level instrumentation extending to S1 can increase the incidence of PJK [24,33,84]. However, if the LIV is L5, revision surgery can be increased by accelerating the degeneration of the L5–S1 disc [85]. Therefore, the decision to choose the LIV at L5 or S1 remains controversial. The incidence of PJK is more than 3 times higher in patients who have undergone a combined anterior-posterior approach than in those who have undergone a posterior-only fusion. It is speculated that this is because the combined approach involves an anterior release, allowing for greater correction and resulting in a larger difference in sacral sagittal vertical line. However, the exact reason for this is unclear [8,11,86]. One important aspect in the occurrence of PJK and PJF is also what to use for proximal implant construction. Pedicle screw is commonly used, but it is rigid and can cause PJK by increasing facet violations at the proximal level [16,20,87]. Therefore, various other methods for semirigid fixation have been devised. It has the concept of topping off, which reduces peak stress by creating a gradual transition of motion at the proximal end of the rigid spinal construct [12,88]. One of the representative methods is the spinal hook, which theoretically reduces facet violation and provides dynamic motion, thus decreasing the risk of PJK. In a study involving 47 patients who underwent transverse hook or pedicle screw fixation at the UIV and were followed up for 2 years, the spinal hook group showed a PJK incidence of 0%, while the pedicle screw group showed an incidence of 29.6%, demonstrating that spinal hooks significantly reduce the occurrence of PJK [20]. In a biomechanical study using cadavers, the loading stress at the proximal level after long-level instrumentation was compared among the groups using supralaminar hooks (17%±11.5%), hybrid constructs (19%±8.2%), and bilateral screws (23%±8.3%). The loading stress in supralaminar hook group was the lowest, indicating that it is effective in reducing loading stress at the proximal level [89]. There is a method to reconstruct the PLC by providing Mersilene tape (polyester fiber) to the lamina or spinous process of UIV-1, UIV, and UIT+1 to reduce the loading stress at this level; tether, sublaminar tape [12,88,90,91]. Doodkorte et al. [12] conducted a systematic review of biomechanical studies and found that tether showed a gradual increase in the transition zone of range of motion (ROM) compared to pedicle screws, and also demonstrated a decrease in intradiscal pressure. In a systematic review of clinical studies, the incidence of PJK was significantly lower in the tether group compared to the pedicle screw group (18% vs. 45%, p=0.045) [88]. Sublaminar tape, like tether, also reduced the ROM of flexion-extension and lateral bending at the index level and decreased the intradiscal pressure [12]. In a prospective study that included 40 consecutive patients who underwent sublaminar tape application at UIV+1, 7.5% of PJK and no cases of radiological PJF occurred during a median follow-up period of one year [92]. The role of the rod is also important in the development of PJK and PJF due to the influence of the long-level arm of instrumentation. Using a stiff rod such as cobalt chromium (CoCr) can increase stability, but due to higher rigidity, it can cause PJK by increasing loading and increasing ROM at both ends of the rod, especially proximal [46,90]. In actual clinical practice, PJK occurred more frequently in the CoCr rod group than in the titanium alloy (Ti) rod group (CoCr 60% vs. Ti 26.5%) [93]. Not only the stiffness of the rod, but also its shape is important in construct design. Contouring of the rod at the proximal level can reduce additional loading forces on the construct and decrease screw pullout strength and junctional stress [2,94]. Kim et al. [90] recommend careful kyphotic bending of the proximal portion of the rod to ensure complete seating of the rod within the screw head at the proximal 2 levels and to prevent additional loading at this level. In addition, the use of a transition rod within a single rod that varies in diameter and strength to modify the rigidity of the construct has been reported to reduce the incidence of PJK [95]. However, due to the difficulty in fitting a rod to individual patients and the lack of clinical data, transition rods are not widely used in actual clinical field. Multiple rod constructs have been used to improve stiffness and stability at pedicle subtraction osteotomy sites in adult deformity correction [96], but a recent study reported that multiple rod constructs did not reduce pseudoarthrosis and rod fracture after PSO for ASD correction [97]. Finally, the easiest but arguably most difficult aspect that a deformity surgeon must be cautious about is posterior soft tissue injury, which is an iatrogenic injury caused by the surgeon. Damage to the paraspinal muscles and posterior ligament complex at the proximal level during surgery has been identified as a risk factor for PJK and PJF in many studies [11,18,23,90,98,99]. Therefore, when exposing the proximal level, it is essential to preserve the interspinous ligaments, supraspinous ligaments, proximal facet, and capsule. This has led to the proposal of minimally invasive surgery as an approach to reduce the risk of injury [100,101]. The risk factors mentioned above are summarized in Table 2.
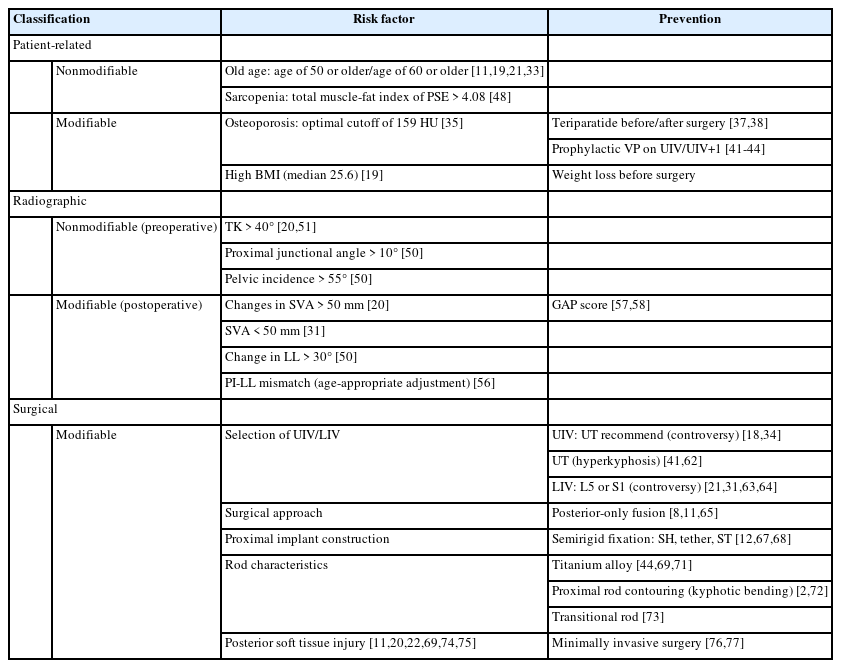
Risk factors and potential prevention strategies for proximal junctional kyphosis and proximal junctional failure
Recently, the application of minimal invasive surgery (MIS) in ASD has been reported because of the relatively short surgical incision, short hospital stays, reduced bleeding loss. Although open surgery demonstrates powerful correction results, it is associated with a higher incidence of PJK due to factors such as more damage to the normal spinal structures and larger implants compared to MIS [102,103]. MIS offers advantages in reducing iatrogenic injuries and minimizing the occurrence of mechanical complications, as it does not require large incisions and extensive muscle dissection. It preserves the muscles, ligaments, and nerves associated with mechanical complications, which can help reduce the incidence of PJK [100]. While MIS cannot currently replace open surgery in ASD, further attempts are needed, and selection of MIS in appropriate patients can minimize PJK.
CONCLUSION
Advances in implant technology and surgical skills have enabled long-level fusion and instrumentation for treating adult spine deformity, but these advancements have also led to new complications including PJK and PJF. PJK and PJF occur due to a combination of various factors, including surgical, radiological, and patient-related risk factors. Therefore, spine surgeon should understand the pathophysiology of PJK and PJF, as well as individual risk factors, in order to develop appropriate prevention strategies for each patient.
Notes
Conflict of Interest
The authors have nothing to disclose.
Funding/Support
This study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author Contribution
Conceptualization: BJL, DJJ, JHP, Korean Spinal Deformity Society (KSDS); Formal Analysis: BJL, DJJ, JHP; Investigation: BJL, DJJ, JHP, KSDS; Methodology: BJL, DJJ, JHP, KSDS; Project Administration: BJL, DJJ, JHP, KSDS; Writing – Original Draft: BJL, DJJ; Writing – Review & Editing: BJL, DJJ, SSB, HYC, SJH, JHP, YC, KSDS.