Robotic-Assisted Spine Surgery: Role in Training the Next Generation of Spine Surgeons
Article information
Abstract
Objective
This study aimed to assess the degree of interest in robot-assisted spine surgery (RASS) among residents and to investigate the learning curve for beginners performing robotic surgery.
Methods
We conducted a survey to assess awareness and interest in RASS among young neurosurgery residents. Subsequently, we offered a hands-on training program using a dummy to educate one resident. After completing the program, the trained resident performed spinal fusion surgery with robotic assistance under the supervision of a mentor. The clinical outcomes and learning curve associated with robotic surgery were then analyzed.
Results
Neurosurgical residents had limited opportunities to participate in spinal surgery during their training. Despite this, there was a significant interest in the emerging field of robotic surgery. A trained resident performed RASS under the supervision of a senior surgeon. A total of 166 screw insertions were attempted in 28 patients, with 2 screws failing due to skiving. According to the Gertzbein-Robbins classification, 85.54% of the screws were rated as grade A, 11.58% as grade B, 0.6% as grade C, and 1.2% as grade D. The clinical acceptance rate was approximately 96.99%, which is comparable to the results reported by senior experts and time per screw statistically significantly decreased as experience was gained.
Conclusion
RASS can be performed with high accuracy within a relatively short timeframe, if residents receive adequate training.
INTRODUCTION
With the rapid advancement of technology over the past 2 decades, numerous surgical robots have been invented and are currently being used in various fields. The field of spinal surgery, in particular, has seen significant trends, with the introduction of the first robotic-assisted pedicle screw fixation system (Mazor Robotics Ltd., Caesarea, Israel) in 2004. Since then, various types of robotic surgical instruments have been developed, each with many advantages, leading to their widespread use [1,2].
Based on the outcomes of robotic-assisted spine surgery (RASS) to date, the integration of robotics in spine surgery offers numerous benefits. Several studies have documented improved accuracy in pedicle screw placement [3-5], freedom from radiation exposure during surgery [6], reduced blood loss, and shortened hospital length of stay [7]. These positive results have increased interest in robotic spine surgery and led to explosive growth in the robot-related medical device market. In response to these concerns, various efforts have been made to strengthen training programs aimed at bridging the gap between technology and core surgical competencies [2,8,9].
Some researchers have argued that adopting spinal robotic systems would not be a hurdle in the training process of young spine surgeons [10], based on the idea that they are familiar with handling new technologies and can quickly adapt to robot systems with an advanced user interface and artificial intelligence [11]. Therefore, from this point of view, young surgeons would be able to use robotic systems to their full potential in operations with complex spinal deformities [12]. In fact, several articles have reported that the use of spinal robotics among trainees revealed no significant difference in the time required for screw placement between junior and senior residents [13].
However, there are concerns about exposing young spine surgeons to robotic surgery at an early stage [10]. Excessive reliance on technology may lead to a lack of proficiency in essential skills that must be mastered during the training process, such as understanding the anatomical landmarks of bony structures and the tactile sensation involved in manually cannulating a pedicle. These concerns are valid and should be taken into consideration when developing training programs for young spine surgeons.
Compared to medically advanced countries where spinal robots devices have been rapidly introduced, our country has shown a very slow progress in spinal robots surgeries. The infrastructure for spinal robotic surgery in our country is not well distributed, and a reimbursement system for medical insurance has not yet been established. As a result, there was less interest in robotic surgery, and relatively insufficient consideration was given to the training process for robotic surgery. Fortunately, the domestically developed CUVIS-spine robot system (CUREXO Inc., Seoul, Korea) has been recently introduced and rapid spread of spinal robotic surgery infrastructure is expected in the future. Therefore, significant efforts are needed for the design of an effective robotic surgery education system.
In considering how to develop an effective robotic surgery training program, we designed this study. Our research is structured around 2 main components. First, we carried out a survey to evaluate the level of awareness and interest in RASS among young neurosurgery residents. Subsequently, we selected a resident from our institution to participate in a hands-on training program, which utilized a dummy to educate young neurosurgery residents. After completing the training, the resident performed RASS while being monitored by an experienced supervisor.
MATERIALS AND METHODS
1. Targets of the Questionnaire Survey
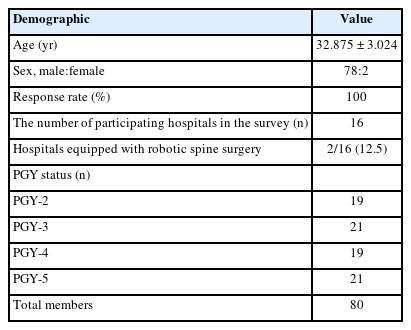
We carefully selected a total of 16 hospitals from among the neurosurgery training hospitals in the Republic of Korea, ensuring that the selection was not regionally concentrated. The questionnaire was distributed via email from June 1 to July 31, 2023. To encourage responses, we personally contacted the survey participants by cell phone. All questionnaires were fully completed and returned, resulting in a 100% response rate. The survey respondents included 80 trainees from the 16 hospitals. The average age of the respondents was 32.875 years, with a gender distribution of 78 men and 2 women. Among the 80 trainees, there were 19 residents in their first year (postgraduate year [PGY]-2), 21 in their second year (PGY-3), 19 in their third year (PGY-4), and 21 in their fourth year (PGY-5). The survey-related information is summarized in Table 1.
2. Content of the Questionnaire Survey
We developed a questionnaire consisting of 10 questions to evaluate neurosurgical trainees’ level of involvement in spinal fusion surgery, as well as their awareness of and interest in RASS. The first and second questions were designed to determine the most commonly used methods for pedicle screw placement at the trainees’ affiliated hospitals and their extent of participation in spinal fusion surgery at their current institutions. The third and fourth questions sought to gauge the trainees’ preferences for screw fixation methods and to gather their opinions (advantages and disadvantages) on each method. The fifth, sixth, and seventh questions aimed to assess the trainees’ awareness of robot-assisted surgery. Questions 8 to 10 explored the trainees’ willingness to actively engage in future RASS training programs. The original survey can be found in Supplementary Material 1.
In the second stage, we assessed a trainee’s proficiency and the speed at which he adapted to the robotic equipment. We selected a resident currently in training at our hospital, a 29-year-old in his third year of the neurosurgery residency program (PGY-4). He was chosen because he has the most experience in percutaneous screw fixation surgeries among the residents currently undergoing training. Under the guidance of an expert, he has already gained substantial surgical experience, having performed approximately 40 cases of percutaneous pedicle screw fusion (PPSF) and 10 cases of open spinal surgery. Although he did not participate in the cadaver training program, he is considered to have a sufficient understanding of the anatomical structures necessary for the successful execution of PPSF. He participated in all surgeries related to this study, while the remaining trainees did not participate in the study.
The robot training program consisted of 2 steps. The first step involved an educational session on understanding the configuration and use of the robot system, which lasted approximately 1 hour. The second step took place in the operating room, where the trainee used a dummy to perform intraoperative scanning with the O-arm. The dummy was specifically designed for training of RASS. The inside of the dummy composed of the radiopaque materials with imitation of real spinal bony structure. So, practical training using radiographic imaging equipment is possible. The trainee planned the screw placement based on imaging data and executed the insertion of pedicle screws (Fig. 1). To ensure familiarity with the equipment, these processes were repeated 3 times. This repetition allowed the trainee to become proficient in both the procedure and the operation of the robotic surgery equipment. The training process using the dummy and the actual surgical procedure are presented in the Supplementary Video Clip.

Robot training process using the dummy: O-arm scanning (A), planning of screw insertion pathway (B), drilling of entry point (C), verifying the entry point (D), tapping (E), screw insertion (F), confirming the position of screw (G).
The application of the procedure to real patients began in June 2023 and continued for 3 months, with a total of 28 patients undergoing RASS. The mean age was 66.35 years (range, 53– 84 years). The inclusion criteria for the study were degenerative lumbar diseases such as degenerative lumbar spinal stenosis and spondylolisthesis. Eligible patients had persistent lower back pain and radicular pain lasting for at least 3 months, which correlated with preoperative magnetic resonance imaging findings and did not improve with medical treatment. Patients with infectious spondylitis, traumatic vertebral fractures, and neoplasms were excluded from the study. All patients underwent interbody fusion using the oblique lateral interbody fusion (OLIF) technique and RASS with the CUVIS-spine robot system. The CUVIS-spine robot system is a domestic robotic system that employs image-guided navigation to provide surgeons with precise spatial positioning and orientation of anatomical structures during pedicle screw insertion surgery. It consists of a main console with an optical tracking camera, a workstation with a planning program, a floor-mounted robotic arm, and a staff console with a touchscreen monitor. This robotic system utilized the scan-and-plan method, and intraoperative computed tomography (CT) images were used for registration. Intraoperative imaging was performed using the O-arm system (Medtronic, Dublin, Ireland). The study protocol was approved by the Institutional Review Board (IRB) of Pusan National University Yangsan Hospital, which waived the requirement for informed consent due to the retrospective nature of this study (IRB No. 55-2023-055).
3. Surgical Procedures
All patients underwent the conventional OLIF technique in the right-lateral decubitus position. After cage placement, the patients were repositioned prone on a radiolucent table for pedicle screw insertion. A 3-cm skin incision was made at the level of the 2 upper spinous processes of the upper instrumented vertebra to mount the optical tracker. Subsequently, we positioned the robotic arm adjacent to the surgical bed and installed the registration tool on the arm. Intraoperative CT scanning was then performed, and the scanned images were directly transferred to the main console. The surgeon used the planning program to plan and preview the screw positions in the target vertebra. During the preview, we could confirm the positions of all screws and adjust the screw location considering the alignment of the rods. Next, the robotic arm was guided to the planned target position, where we made a skin incision and inserted an expander to dilate the working corridor. We identified the insertion point and verified the drilling point with a ball-tip probe. The process of drilling, tapping, and screw insertion was then conducted with the assistance of the robotic arm. The system displayed the lateral repulsive force generated when surgical instruments contacted the bone surface, aiding in the correct insertion of screws along the planned pathway. After completing all procedures, intraoperative O-arm imaging was performed again to confirm the placement of the screws. Finally, we used a conventional fluoroscopic C-arm to align the rods and complete the surgery.
4. Assessment of the Learning Curve
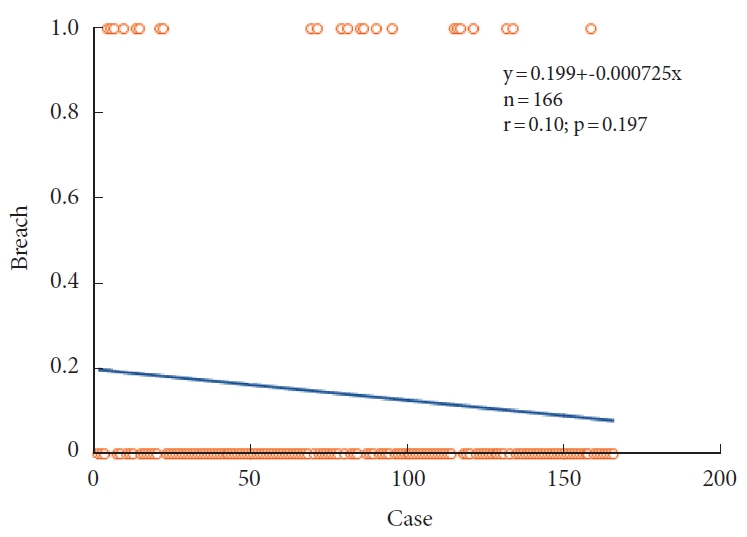
To assess the trainee’s adaptation to an unfamiliar robotic-assisted navigation system, we first evaluated their performance based on 3 categories: (1) number of supervisor interventions, (2) time per screw, and (3) accuracy analysis of screw position. We closely monitored the trainee throughout the entire surgical procedure and actively intervened when necessary. Surgical intervention was defined as any action in which the supervisor actively participated in the procedure, including verbal guidance and direct assistance. This encompassed determining the location and length of the skin incision, selecting screw length, and guiding the trajectory of screws and entry points for drilling. These actions were quantitatively measured and recorded. Second, we analyzed the surgical operation time for screw insertion to gauge the trainee’s proficiency. We measured the time interval between the intraoperative O-arm scan used for planning and the postoperative O-arm scan used to confirm screw placement. The time per screw was calculated by dividing the total operation time using the robot by the number of screws inserted during the patient’s procedure. Third, the accuracy of screw placement was classified according to the Gertzbein and Robbins scale classification [14]. Grade A was assigned when a screw was placed entirely within the pedicle; grade B for a cortical breach of the pedicle within 2 mm; grade C for a breach from 2 mm to 4 mm; grade D for a breach from 4 mm to 6 mm; and grade E for a breach of more than 6 mm.
5. Statistical Analysis
Statistical analysis was conducted using IBM SPSS Statistics ver. 28.0 (IBM Co., Armonk, NY, USA) and MedCalc ver. 19.6 (MedCalc Software Ltd., Ostend, Belgium). Continuous variables that followed a normal distribution were presented as means with standard deviations. Learning curves were analyzed using binomial logistic regression, and the threshold for statistical significance was set at a p-value < 0.05.
RESULTS
1. First Stage: Results of the Survey
1) Main methods of pedicle screw placement and degree of participation in spinal fusion surgery
The first question was about the primary methods for pedicle screw placement employed in contemporary hospitals. The method most frequently utilized in hospitals that train residents is fluoroscopic-guided open pedicle screw fixation, accounting for 41%, followed by the PPSF technique at 34%. Navigation and robotic surgery were limited to surgeries performed using the percutaneous pedicle screw technique. Navigation-guided techniques and robotic systems, being highly expensive, are available in only a limited number of hospitals, which explains their lower usage rates.
In the second question, we categorized surgical experience based on the number of operations an individual had participated in. Those with no surgical experience were classified as “none,” those with fewer than 10 operations as “low,” those with 10 to 20 operations as “middle,” and those with 30 or more operations as “high.” Surgical experience was differentiated between cases in which individuals acted as operators and those in which they served as assistants. The survey results are summarized in Table 2. Operations involving navigation guidance and robotic assistance were expected to have lower levels of experience due to the limited availability of equipment, and this was indeed confirmed by the survey results. However, it was also noted that many trainees lacked experience in the spinal surgery process. While spinal surgery experience is not a prerequisite for becoming a neurosurgeon, we believe that spine training education should be further emphasized.
2) Preference of screw fixation methods and the trainee’s thoughts
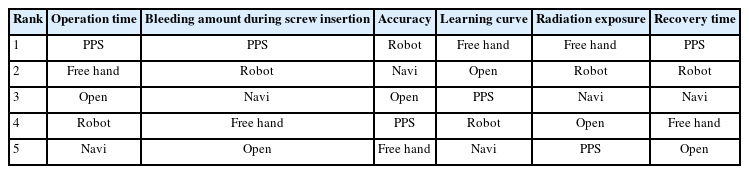
The third and fourth questions focused on the preferences for screw fixation methods and the trainees’ perspectives on the advantages and disadvantages of each method. We used a method of ranking by summing the assigned rankings for each surgical method and then assigning rankings based on the order of the lowest total score received. Consistent with the previous responses, the preference for the open method and PPSF was the highest across the board. Trainees reported difficulty in confirming anatomical landmarks, which led to a decreased preference for the freehand technique due to the risk of cortical breach. Preferences for the navigation-guided screw method and robot-assisted screw method were lower, attributed to the limited availability of navigation equipment. The results of survey question 3 are presented in Table 3. In response to the fourth question, trainees indicated that the percutaneous screw fixation technique offers the most advantages in terms of operation time, estimated blood loss, and recovery time. Navigation-guided surgery and robotic-guided surgery were recognized for their accuracy as a primary advantage, and it was noted that the learning curve for these methods was not steep. The results of survey question 4 are presented in Table 4.
3) Level of awareness of robotic-guided surgery
The fifth and seventh questions were designed to assess the level of awareness regarding robotic-guided surgery. For the fifth question, only 2 participants answered that they are familiar, 46 have heard about it, and 32 answered that they do not know what it is. Most trainees demonstrated a low awareness of robotic-guided surgery; however, their level of interest was relatively high. A binary question to examine the detailed content of robotic-guided surgery was posed in the seventh question. Trainees frequently selected the correct answer we anticipated, confirming their awareness of the relatively accurate details of the process and outcomes of robotic surgery. The questions and results of the seventh question are summarized in Supplementary Material 2.
4) Interest in robotic-guided surgery and willingness to participate in future training programs
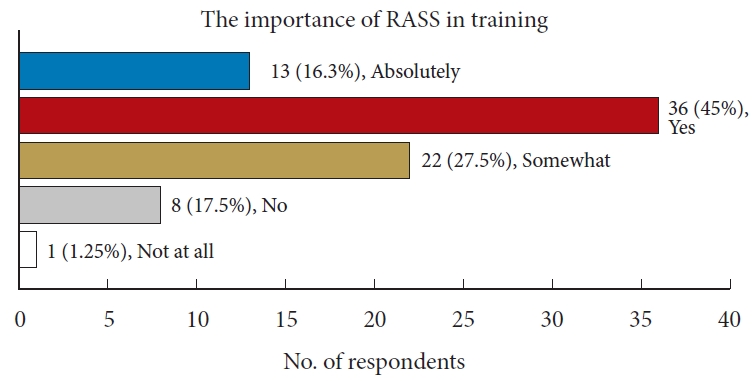
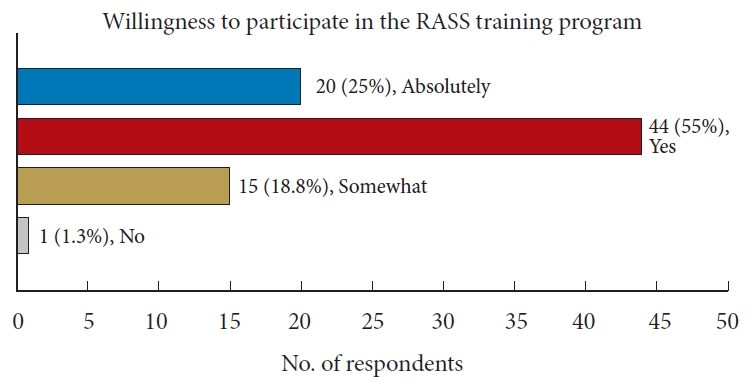
Questions 6, 8, 9, 10 inquired about the trainees’ interest and willingness to engage in future robot-assisted surgery training programs. Question 6 revealed that two-thirds of the respondents expressed interest in RASS (Fig. 2). A significant majority, approximately 88%, acknowledged the necessity of training in robotic surgery (Fig. 3). Furthermore, 59% of respondents were optimistic about the potential for robot-assisted surgery to become a mainstream surgical approach in the future in question 9. When asked about their willingness to participate in a training program, 79% responded affirmatively (Fig. 4). These findings indicate a strong interest among residents in robotic surgery training.
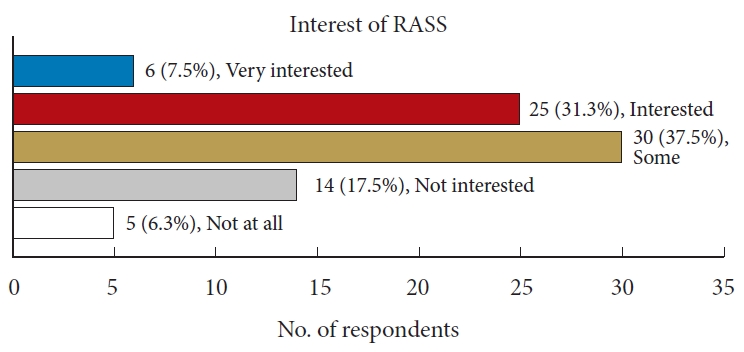
Result of question 6, interest of RASS. Two-thirds of the respondents expressed interest in RASS. RASS, roboticassisted spine surgery.
2. Second Stage: Clinical Results
The medical records and radiological examinations of 28 patients were retrospectively analyzed. Our study comprised 13 male and 15 female patients, ranging in age from 55 to 84 years. The average age of the patients was 69.36 years.
Among the 28 patients, 2 had previously undergone spinal fusion surgery, and the surgical procedure was performed to extend the range of the earlier fusion. In 6 patients, posterior laminectomy surgery was conducted to remove elements causing neural compression after the insertion of an OLIF cage. A total of 166 robot-assisted screw insertions were attempted, with 164 screws successfully placed. Robot-assisted screw insertion failed in 1 patient due to severe skiving, necessitating a switch to fluoroscopic-guided screw insertion. Patient demographics are summarized in Table 5.
3. Results of the Screw Position
We analyzed the placement of 166 screws: 4 screws were placed in the L1 vertebra, 16 in the L2 vertebra, 34 in the L3 vertebra, 54 in the L4 vertebra, 54 in the L5 vertebra, and 4 in the S1 vertebra. Out of the 166 screws, 142 were classified as grade A (85.5%), 19 as grade B (11.6%), 1 as grade C (0.6%), 2 as grade D (1.2%), and the remaining 2 screws were considered failures due to skiving. We considered grades A and B to be clinically acceptable results, and the screw placement accuracy rate of the robotic RASS was 96.99% (Table 6).
4. Analysis of the Learning Curve
We measured the learning curve in terms of 3 metrics. The first metric was the degree of supervisor intervention. On average, the supervisor intervened approximately 5.1 times per surgical case. While the level of intervention was notably high during the first and second cases, it showed a gradual decline. By the sixth case, the average level of intervention had stabilized. The trend in supervisor intervention relative to the surgeon’s growing experience is depicted in Fig. 5. The second metric was the change in time per screw, which was determined by dividing the total time spent using the robot by the number of screws placed. The average time per screw was 9.85 minutes. As with supervisor intervention, the initial 2 cases required a significantly longer time per screw, but this time quickly decreased and the graph’s slope flattened, indicating stabilization. The binary logistic regression analysis revealed that the time per screw statistically significantly decreased as experience was gained (Fig. 6). The final metric was the accuracy analysis of screw placement. Out of 166 screws, those classified as grade A were considered nonbreached, while all others were categorized as breached. Although there was no statistical significance, there was a trend indicating a decrease in the occurrence rate of breached screws over time (Fig. 7).
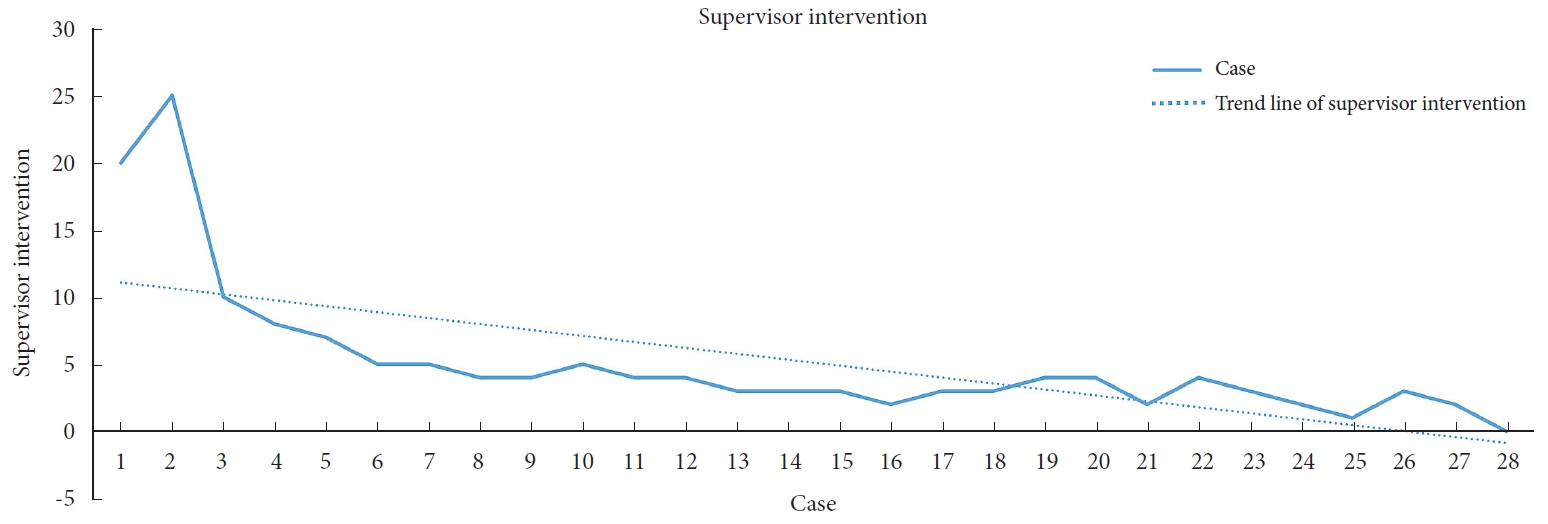
Change of supervisor intervention according to the accumulation of experience. The trend line of the supervisor intervention approaches the average value around 6th case and gradually decreases.

Change in time per screw according to the accumulation of experience. The binary logistic regression analysis revealed that the time per screw statistically significantly decreased as experience was gained (p=0.05).
DISCUSSION
Since the development and U.S. Food and Drug Administration approval of a robotic spine surgery system overseas, numerous studies have reported on its accuracy, risk factors, and learning curve [2,9,14,15]. Although Korea entered the field later, the CUVIS-spine robot system was developed using domestic technology and currently being used and clinical results are reported [5,16]. Our experiences with spine surgery have been shared at various domestic conferences to benefit other spinal surgeons. Our findings have garnered considerable support and interest. However, some experienced spine surgeons have voiced concerns. Traditional neurosurgical training emphasizes responsibility and autonomy, with patient safety as the foremost priority, while cautiously integrating new technologies. The field of spine surgery has seen the introduction of many new surgical instruments and techniques. Yet, the acquisition of these new skills is increasingly difficult for trainees due to restricted working hours and other limitations. It is a daunting task to master conventional surgical techniques in spine surgery, as well as additional robotic skills, within the 4-year neurosurgical training period. Furthermore, in a state of technical immaturity, an excessive dependence on technology can lead to concerns about the potential neglect of essential skills that should be mastered during training.
In light of these concerns, we reflected further on the direction we are currently pursuing. This prompted us to conduct a survey on the training situation of residents in the field of spinal surgery and their perception of robotic surgery, and developed and implemented our own curriculum for robotic spine surgery.
The survey results aligned with some of our expectations but also brought to light some surprising findings. As anticipated, fluoroscopic-guided open pedicle screw fixation and PPSF were the most common methods for screw insertion used in other hospitals. However, the adoption of robots or navigation systems for screw insertion was significantly lower, likely due to the prohibitive costs and limited availability of these advanced technologies in hospital settings. An unexpected discovery was the extremely low level of trainee involvement in spinal surgery (survey question 2, Table 2). It was revealed that over half of the trainees had limited exposure to spinal fusion surgery until they obtained their specialist licenses. This lack of practical experience can be attributed to the current laws regulating trainee working hours in Korea, as well as a trend towards a training system that is predominantly centered around fellows. This has highlighted a substantial gap in resident training, which is disappointing from an educational perspective. This situation underscores the need to reevaluate and potentially restructure surgical training programs to ensure that residents receive comprehensive and hands-on learning experiences in the field.
The survey on trainees’ preferred methods of screw insertion (survey question 3) indicated a strong preference for open procedures and PPSF, with a significantly lower preference for the freehand technique. This tendency appears to stem from their current stage of training and possibly a less thorough grasp of surgical anatomy. Trainees are inclined to favor techniques that provide a clearer and more accessible identification of the entry point, which explains their preference for open procedures and PPSF over the freehand technique. The value placed on visualization could render robotic surgery particularly appealing.
Despite the challenges in the educational environment, trainees’ eagerness to participate in robotic training programs (survey question 10) and interest about the RASS (survey question 6) suggest a positive outlook. Their expectation that RASS will become a standard practice in the future underscores the need to integrate these technologies into training curricula. Developing such curricula is crucial to prepare future surgeons with the skills and knowledge required to proficiently perform robotic spine surgery, which is gaining significance in the rapidly advancing field of surgical technology.
Before establishing a robotic spine surgery curriculum at our hospital, a review of the existing literature underscored the necessity for a systematic course that transitions from simulation training to hands-on surgical procedures. Pennington et al. [2] recommended structuring robotic surgery education into 3 phases: didactic education, preclinical training, and live surgery. They provided clear objectives for each phase and detailed the process for evaluating and enhancing problem-solving skills in anticipation of surgical complications. We utilized their framework as a model to develop a comparable training curriculum and to educate our trainees. Patient safety is another critical consideration, underscoring the importance of conducting training during actual surgery under the supervision of seasoned professionals. This method ensures that residents not only master the technical aspects of robotic surgery but also develop vital competencies in a controlled and secure setting. The strategy is designed to narrow the divide between theoretical knowledge and practical experience, while placing the highest priority on patient safety.
Setting the criteria for selecting participants for the practical training has some difficulty. We decided to conduct the study with participants who had sufficient experience in PPSF, considering that RASS is similar to PPSF. We set a minimum criterion of surgical experience at least 40 cases of PPSF for participant selection. To achieve this, a minimum of 6 months of clinical experience is required. Subjectively, with this level of experience accumulated, it can acquire essential skills that must be needed for RASS, such as understanding the anatomical landmarks of bony structures and the tactile sensation involved in manually cannulating a pedicle.
Our hospital introduced the CUVIS-spine robot system around October 2021, and we have recently reported the surgical outcomes of the first 100 cases of robot-assisted spine surgery conducted by senior surgeons [16]. When comparing these outcomes with those of previous procedures, the overall results were similar when performed by experienced surgeons. In our study, the accuracy of screw placement was 96.99%, which is slightly lower than the previously reported accuracy of 98.12%. However, our results are nearly comparable to those reported in other literature on robotic spine surgery [17,18]. Notably, the time per screw, which represents the duration of robot-assisted screw fixation, showed a significant improvement in our study, with an average of 9.84 minutes compared to the previous average of 21.04 minutes. These results are likely due to the accumulated experience in operating the robotic system.
We demonstrated a very short learning curve. Initially, trainees faced challenges, displaying unfamiliarity and discomfort during the early stages of their training. However, they adapted to the robotic apparatus more quickly than anticipated. Supervisor intervention reached the average around the 6th case and the time required to place each screw reached the average by the third case. Over time, the accuracy of screw placement exhibited a consistent improvement. In a meta-analysis examining the learning curve associated with RASS, the number of cases needed to overcome the learning curve for spine robots ranged from 3 to 30 cases, or 15 to 60 screws [2]. A comparative study on the accuracy of screw insertion between residents and attending spine surgeons found no significant difference between the 2 groups [13]. Taking into account these varied findings, we cautiously conclude that the barriers to mastering robotic surgery are not insurmountable.
In view of increasing demand for spinal fusion surgery due to aging population and the rapid advancement of robotic surgery, robotic surgery is a highly promising field in the future. Integrating robotic surgery in regular training programs would be a natural choice in line with technological advancements. According to our study, young trainees can be easily adapted to robotic surgery and demonstrated similar result comparable to those of attending surgeons. With the reduction in the burden of spinal fusion surgery due to RASS, trainees can have more opportunities to focus on the fundamental aspects of spinal surgery, such as decompressing of neural elements, correcting sagittal imbalance and inserting interbody structures for anterior support. However, excessive reliance on technology can diminish the ability to adapt to abnormal situations and hinder the advancement of surgical skills in spinal surgery. So, trainees without experience in spine surgery should avoid attempting RASS without understanding traditional surgical procedures.
The limitations of our study include its inability to survey residents from other hospitals across the country, which restricts the representativeness of our sample. Another constraint is the limited state of spinal robotic surgery infrastructure, as only 2 hospitals in the nation are equipped with spinal robots.
Most respondents lack actual experience with robotic surgery. So, there may be bias due to respondents’ lack of experience and this should be taken into account when interpreting the survey results. Furthermore, the study focused on a single trainee with considerable experience in percutaneous spine surgery, and the overall sample size was relatively small. Nevertheless, given the increasing need for and interest in robotic spinal surgery, mastering this skill is undeniably crucial for the next generation of spine surgeons. We argue that it is imperative to develop a comprehensive training curriculum for robotic spinal surgery that augments traditional spinal surgical methods. Such an integrated educational approach would ensure that emerging surgeons are proficient in both conventional and cutting-edge surgical techniques.
CONCLUSION
The survey results indicated a deficiency in actual surgical participation during the training of residents. However, the interest in RASS among neurosurgery residents was high. RASS can be performed with high accuracy in a relatively short period of time if residents receive sufficient training.
Supplementary Materials
Supplementary Materials 1, 2 and video clip can be found via https://doi.org/10.14245/ns.2448006.003.
Notes
Conflict of Interest
The authors have nothing to disclose.
Funding/Support
This study was supported by a 2023 research grant from Pusan National University Yangsan Hospital.
Author Contribution
Conceptualization: DWS, GSS, SY; Data curation: DWS, SHL, SWL, GSS; Formal analysis: JSL, YHK, BKO; Funding acquisition: DWS, JSL; Methodology: SHL, SWL, SKS; Project administration: JSL, SHL, JHL, YHK; Visualization: JHL, YHK; Writing - original draft: JSL, BKO; Writing - review & editing: SKS.