Analysis and Temporal Evolution of Extubation Parameters for Patients Undergoing Single-Stage Circumferential Cervical Spine Surgery
Article information
Abstract
Objective
Airway obstruction after postoperative extubation is a dreaded but uncommon complication in patients undergoing circumferential cervical spine surgery (CCSS). The cuff leak test (CLT) has been utilized to assess air leak around the endotracheal tube which may reflect airway swelling. In this prospective observational study, we analyze the temporal evolution of CLT and perioperative factors that may influence it.
Methods
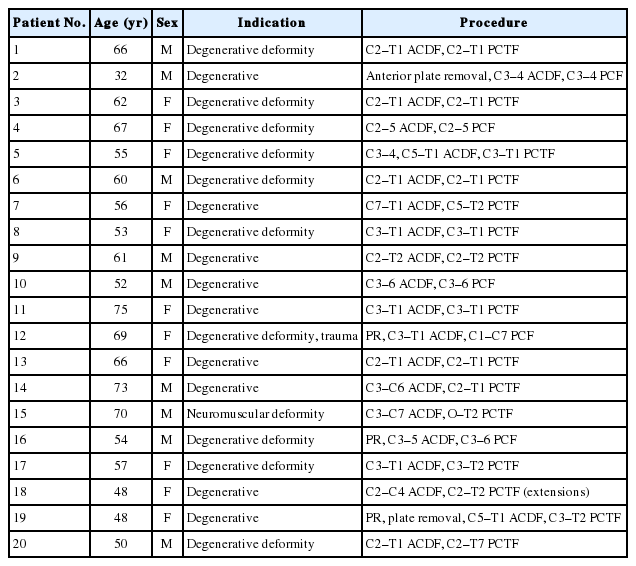
Twenty patients undergoing single-stage CCSS were managed according to our extubation protocol. Patients were maintained intubated overnight following surgery. They were extubated if a CLT > 200 mL and both intensive care unit (ICU) and Neurosurgery teams agreed that it was safe. Patients extubated in the first postoperative day (8 of 20) comprised the early group, and the remaining patients (12 of 20) the delayed group. Patient and operative data were analyzed as a single group and comparing both groups.
Results
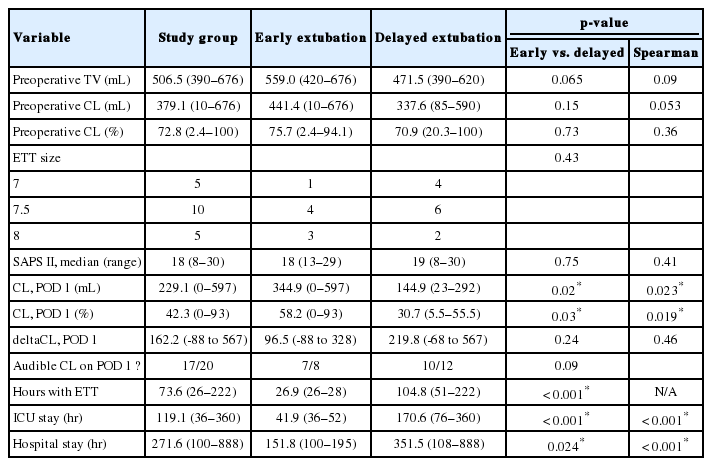
The main indication for surgery was cervical deformity. Median number of levels fused was 5 anteriorly (range, 1–6) and 6 (range, 1–13) posteriorly. Patients were kept intubated for an average of 73.6 hours (range, 26–222 hours) and stayed in the ICU for 119.1 hours (range, 36–360 hours). There were 4 failed extubations and 3 patients (15%) required a tracheostomy. Patient profiles between both groups were very similar across most patient variables but differed significantly regarding infraglottic luminal area (p < 0.05). Patients with larger preoperative cuff leak values tended to have a shorter intubation period (p=0.053).
Conclusion
This study objectively demonstrates the difficulties in airway management following CCSS and provides useful insight for preoperative planning and counseling. Local anatomic factors influence airway outcome more than operative factors. The study format does not allow for testing of interventions but we suggest that patients with favorable anatomy (larger infraglottic luminal area) may benefit from a less strict airway management protocol.
INTRODUCTION
Extubation of a patient following large reconstructive procedures of the cervical spine is a critical portion of postoperative care. Inability to secure an airway is a serious concern due to laryngeal swelling and limited cervical motion with potentially catastrophic consequences [1,2]. Airway compromise in the postoperative period can occur due to factors extrinsic or intrinsic to the larynx. Extrinsic compression following the anterior approach to the cervical spine may happen due to soft tissue swelling, hematoma or accumulation of cerebrospinal fluid (CSF) or hemostatic agents [3,4]. Intrinsic obstruction may happen due to laryngeal swelling, vocal cord paralysis, local trauma secondary to intubation (such as arytenoid cartilage dislocation), laryngospasm or increased secretions [3,5]. Some of these causes have also been linked to other surgical procedures performed in the neck, for example, hematoma after endarterectomy or vocal cord paralysis after thyroidectomies. However, anteriorposterior cervical reconstruction has particular characteristics that may render it as one of the most dangerous operations in terms of airway obstruction [6,7]. The tracheoesophageal bundle is directly retracted in the anterior approach as described by Smith and Robinson, occasionally for prolonged periods of time; the addition of a posterior component may worsen both extrinsic and intrinsic factors due to recumbent positioning and overall longer intubation time [8,9]. Finally, should reintubation become necessary, this situation is further complicated by decreased mobility of the cervical spine secondary to the performed instrumented fusion. Depending on available resources and the experience of the person performing the intubation, an infraglottic airway may be the only option [10].
Large anteriorposterior cervical reconstructions have become more common as a result of spine surgery becoming safer in the 1980s and 90s, increased longevity of patients and emergence of cervical deformity as a treatable pathology. Certain complications of these procedures also may become evident only after very prolonged follow-up, such as adjacent level degeneration and or post-laminectomy kyphosis [11,12]. Extubation of these patients is a very challenging task; surprisingly little information is found in the literature regarding this specific population. We present here our data compiled from a prospective, observational study of patients undergoing single-stage circumferential cervical spine surgery (CCSS) (posterior – anterior – posterior [540] or anterior – posterior [360] cervical fusion) and analyze its temporal evolution and patient- and operations-pecific factors. Our hypothesis is that a quantitative cuff leak test (CLT) > 200 mL is predictive of a successful extubation following CCSS.
MATERIALS AND METHODS
This study was approved by the Institutional Review Board (IRB) of Rush University Medical Center (IRB No. 13121101). This is a prospective and observational study. Twenty consecutive patients undergoing CCSS from 03/2014 to 09/2015 at our hospital were enrolled. Exclusion criteria were past or present tracheal or pulmonary pathology such as patients with diagnosis of mediastinal neoplasm or history of transplant, prior traumatic intubation, penetrating trauma to the neck and neoplastic or infectious indications for surgery. Surgery was performed by 2 of the authors (VCT and RBVF) and patients were treated according to the usual protocol introduced by the senior author (VCT) in 2009 based on personal experience and anecdotal data obtained from other leaders in the field. This protocol relies heavily on the quantitative aspect of the CLT for evaluation of airway patency.
The anterior cervical surgery is performed from a left-sided approach according to usual technique [9]. Most reconstructions were fairly large with cervical deformity as the most frequent indication (Table 1). Perioperative steroids were not used. A Thompson table-mounted retractor was used for all anterior cervical approaches (Thompson Surgical Instruments, Traverse City, MI, USA). The anterior cervical reconstruction is always performed first to obtain the necessary correction (Fig. 1) unless posterior release is necessary (Fig. 2). Negative-pressure drains were placed in both the anterior and posterior cervical wounds. Patients were sent intubated to the Neurosciences Intensive Care Unit and were jointly managed by the Neurosurgery and Intensive Care teams; adequate analgesia and anesthesia were provided as necessary. A team of respiratory technicians is involved in the care of all Neurosurgical Intensive Care Unit (NSICU) patients. Airway patency was assessed using a measured and qualitative (audible or nonaudible) CLT as originally described by Miller and Cole but modified for a more modern ventilation strategy, incorporating lower tidal volume ventilation (8-mL/kg ideal body weight), newer ventilation modes and physiologic PEEP, as these advances were introduced a posteriori [13].

Sample case, patient No. 3. Patient with a history of prior uninstrumented C4–5 anterior cervical decompression and fusion and then underwent C3–7 laminectomy for continued weakness. She presented with progressive myelopathy and symptomatic cervical kyphotic deformity. Underwent a C2–T1 anterior cervical discectomy and fusion and C2–T1 posterior instrumentation. (A) Preoperative lateral radiograph. (B) T2 sagittal magnetic resonance image. (C) Postoperative lateral radiograph.
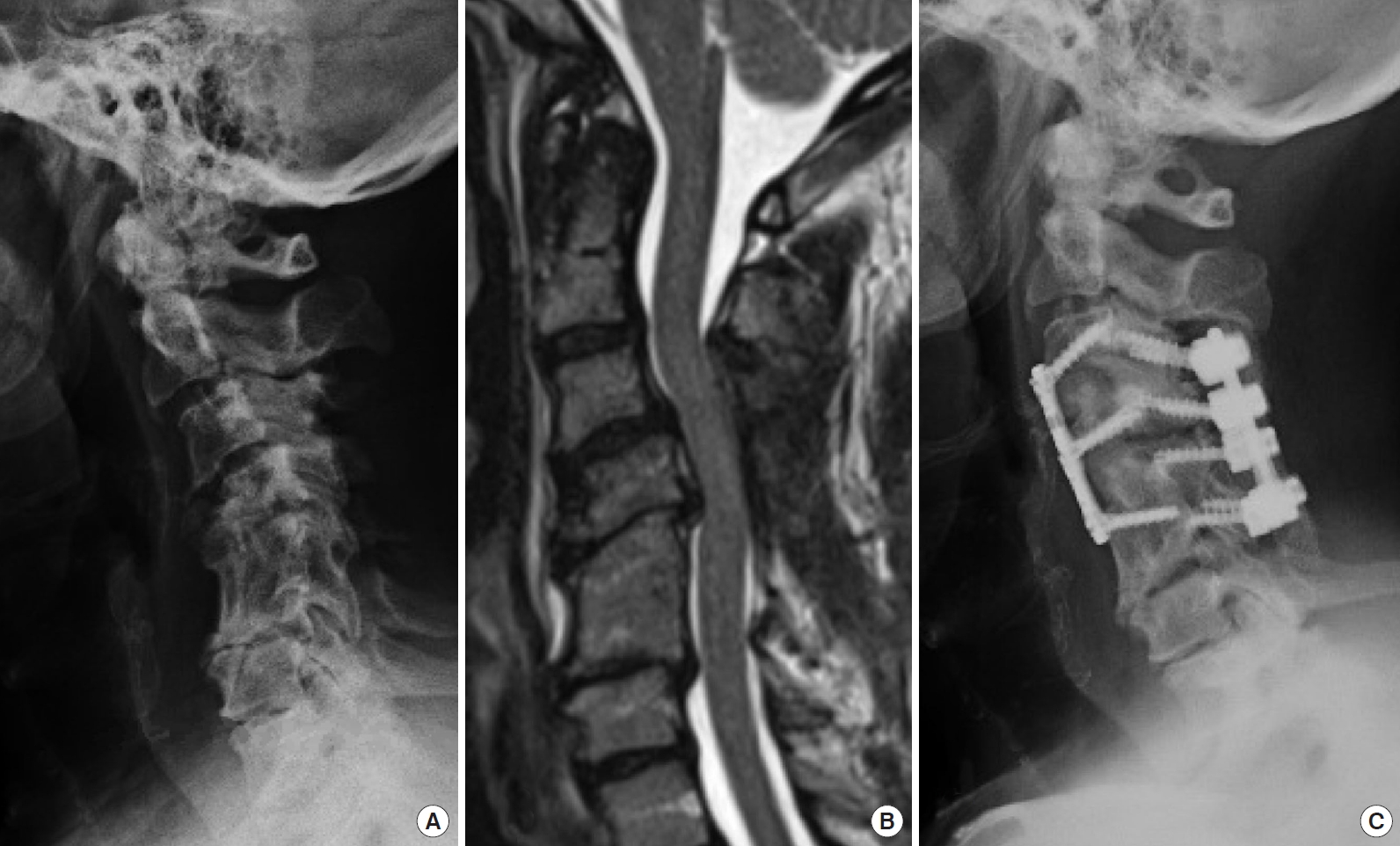
Sample case, patient No. 10. Patient with a history of a prior uninstrumented anterior C5–6 fusion and C4–6 posterior laminectomy with symptomatic cervical kyphotic deformity and myelopathy. Underwent a C3 laminectomy and posterior release, C3–5 anterior cervical discectomy and fusion and C3–6 posterior instrumentation. (A) Preoperative lateral radiograph. (B) T2 sagittal magnetic resonance image. (C) Postoperative lateral radiograph.
1. CLT Testing Protocol
Patients were tested in the operating room after general anesthesia and endotracheal intubation (preoperative CLT), and then daily in our ICU at 7 AM. The Evita ventilator (Dräger Medical AG & Co. KGaA, Lübeck, Germany) in our NSICU was reconfigured to turn off automatic leak compensation. The patient was placed in a semi-Fowler position, secretions suctioned from the endotracheal tube (ETT) and oropharynx and the ventilator set according to the following parameters: pressure-regulated, volume-controlled ventilation mode, respiratory rate 12 breaths/min, FiO2 0.4, PEEP 5 cmH2O and tidal volume of 8 mL/kg based on ideal body weight. Six respiratory cycles were observed until equalization between inspired and expired tidal volumes took place, with an acceptable error margin of 20 mL [5,14]. After adequate suctioning, the ETT cuff was then deflated and then 2 additional respiratory cycles were performed to allow the subject to cough. Expired tidal volume was then recorded over the following 6 respiratory cycles, the cuff reinflated and ventilatory parameters returned to baseline. A quantitative cuff leak (CL) was calculated as the difference between the mechanical inspired volume and the average of the 3 lowest tidal volumes with the cuff deflated [5,14]. The evaluator also assessed the CL qualitatively whether it was audible or not.
2. Extubation Protocol
Patients were eligible for extubation starting on the morning following surgery. Criteria for pursuing extubation included awake and cooperative patient or at baseline cognitive status, satisfactory ventilatory parameters (ability to maintain adequate oxygenation with an FiO2 of 0.4), hemodynamic stability, presence of an audible CL and CLT equal or greater than 200 mL, adequate cough, and minimal suctioning requirements. Extubation was always performed by the ICU team including a respiratory therapist, with Anesthesia (for fiberoptic airway access) and Neurosurgery (for emergent infraglottic airway access) backup. If both ICU and Neurosurgery teams did not agree on pursuing extubation before 12 PM, it would be delayed until the following morning. If a patient failed extubation or if extubation criteria were not met by postoperative day (POD) 10 and no there was no perspective of imminent extubation, tracheostomy was offered to the patient. We avoided the use of steroids in all patients due to evidence of decreased spinal fusion rates.
3. Data Collection and Analysis
Pre- and postoperative patient and surgery data were collected and are demonstrated in Table 2. Subjects were additionally screened preoperatively for sleep apnea with the STOP-Bang tool [15]. In most patients, laryngeal morphology could be evaluated through the preoperative cervical computed tomography (9 patients) or magnetic resonance imaging (7 patients) utilized for planning of the spinal reconstruction. Luminal area was measured at the level of the glottis and the infraglottic space and as well as a measured and a calculated AP laryngeal diameter according to the formula of Higgenbottam and Payne, based on the subject height (anteroposterior [AP] diameter= [33.9× height (m)]–33.7) [16]. Both the measured and calculated AP diameter are commonly described in the otolaryngology and pulmonary literature and thus included in our data. Laryngeal morphology could not be measured in 3 patients due to artifact from prior instrumentation and in 1 patient due to severe cervical kyphosis. Finally, length of time with an ETT was measured from its insertion before surgery till extubation.
Patients were analyzed initially as a single group and, a posteriori, divided into 2 groups based on whether they were promptly extubated at the first CLT (early group, POD 1, up to 36 hours after surgery) or required more time until they could be extubated (delayed group). Protocol violations were recorded and are discussed. Statistical analysis was performed with Prism 8 (Graphpad Software, San Diego, CA, USA) with a significance level set at 0.05 [17]. Nonparametric methods were utilized throughout the study due to the small number of subjects. These methods included Spearman correlation, Fisher exact test (contingency analysis) and Mann-Whitney (continuous variables) and are indicated where appropriate.
RESULTS
Patient baseline and anatomical characteristics are presented in Table 2. Patients in both groups were comparable except for infraglottic cross-sectional area (p=0.031), while height (and its derived parameter, calculated tracheal diameter) approached significance at 0.066. Operative data are demonstrated in Table 3 and are roughly similar between early and delayed groups. Perioperative critical care and hospital stay data, including CL data, are demonstrated in Table 4. Study groups differed in preoperative tidal volume, POD 1 CLT (absolute value and percentage), duration of endotracheal intubation, ICU, and hospital stay (p<0.05). In 16 out of 20 total patients, a preoperative CLT yielded a value > 200 mL. Analysis of preoperative and operative variables using Spearman rho failed to identify a correlation with duration of postoperative intubation, although duration of surgery (p=0.085), infraglottic area (p=0.083) and preoperative leak test volume (p=0.053) approached statistical significance. Analysis of postoperative variables demonstrated only expected and interrelated correlations – for example, larger CL value correlated with shorter intubation (CLT was the main defining factor of ETT permanence) and duration of intubation correlated with ICU and total hospital length of stay (Table 4).
Protocol violations during the extubation process were classified into either a negative violation (failure to extubate despite CL> 200 mL) or positive violation (extubation despite CL< 200 mL). There were 9 violations out of a total of 51 tests (Fig. 3). In the early group, there was only one positive violation which resulted in a successful extubation. In the delayed group, there were 3 positive violations in which all ultimately resulted in successful extubations. Positive violations all occurred in the setting of a CLT< 200 mL but with present audible leak; the attending surgeon also considered these cases to be smaller or shorter and CLT values were relatively close to 200 mL (186, 165, and 135 mL). There were 5 negative violations in the delayed group – these happened when despite CLT > 200 mL, patients were considered to be not ready for extubation due to neurological, pulmonary or a local (anterior cervical swelling) factors.
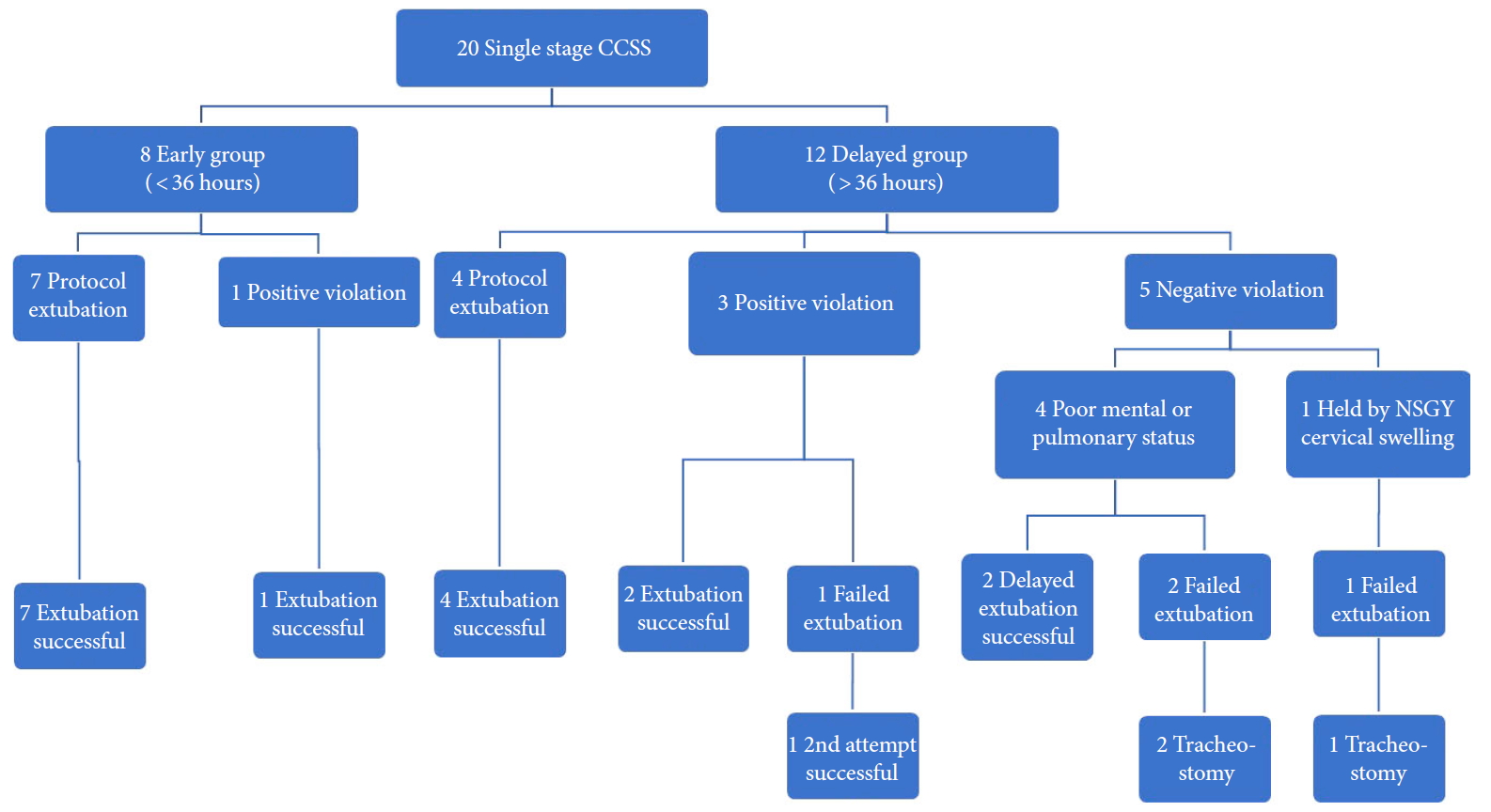
Flowchart demonstrating extubation protocol and patient outcome. “Positive violation” of protocol indicates extubation despite cuff leak < 200 mL. “Negative violation” indicates extubation held despite cuff leak > 200 mL. CCSS, circumferential cervical spine surgery; POD, postoperative day; NSGY, neurosurgery. Extubation protocol: Awake and cooperative patient or at baseline cognitive status, satisfactory ventilatory parameters (ability to maintain adequate oxygenation with an FiO2 of 0.4), hemodynamic stability, presence of an audible cuff leak and cuff leak test equal or greater than 200 mL, adequate cough, minimal suctioning requirements along with intensive care unit and Neurosurgery team agreement.
There were total of 4 failed extubations, of which 3 patients ultimately required a tracheostomy. One additional patient never reached a CLT> 200 mL or even an audible leak and underwent tracheostomy on POD 9 without attempted extubation (Fig. 3). All patients who failed extubation were reintubated within 4 hours of extubation – an effort was made to proactively reintubate patients at the end of the day if their respiratory status was tenuous. All patients undergoing a tracheostomy were decannulated by POD 50 and resumed a fully oral diet.
DISCUSSION
Airway obstruction following postoperative extubation following anterior cervical spine surgery represents a much-dreaded complication. It has many potential causes and is particularly concerning to surgeons because of its potential to quickly progress to respiratory arrest and death complicating a procedure that most of the time is considered “straightforward.” Airway complication rates are quoted around 1% of all anterior cervical procedures, usually restricted to 1- or 2-level pathology and are frequently discussed in the literature in the context of outpatient anterior cervical surgery and its safety [18,19]. This has led authors to identify patients at-risk for unplanned reintubation. CCSS is invariably one of the worst risk factors not only because of the magnitude of the operation but also since it may serve as a marker of severe pathology and/or morbid patients [20,21]. Even beyond the operative time, it obvious that a C5–6 ACDF patient is very different from a posterior-anterior-posterior C2–T2 reconstruction patient.
Terao et al. [21] reported out of a small series of 10 patients undergoing combined, single-stage CCSS, 3 were extubated according to the usual protocol for general spine surgery: immediately at the end of surgery, in the operating room – all 3 of them developed respiratory distress and required reintubation, which was unsuccessful in 1 of them, leading to emergent crycothyroidotomy. Analyzing their complete series of cervical spine surgeries, they calculated the odds ratio of a postoperative airway emergency in CCCS at 178.5.
Emery et al. [1] were the first to report on mortality following multilevel anterior cervical corpectomies and suggested a specific protocol to protect these patients from a respiratory prospective. Based on their negative experiences, they modified their postoperative protocol to include maintenance of the ETT for 24 to 72 hours postoperatively, pre-extubation inspection of the glottis area with laryngoscopy and a qualitative CLT. Epstein [22] identified obesity (> 99.8 kg), operative time greater than 10 hours, revision cervical surgery, extension to C2, more than 4 units of transfused blood, advanced age, CSF fistula, length of fusion and severe myelopathy (Nurick 4 or 5) as risk factors for delayed extubation following multilevel cervical corpectomies. These authors also demonstrated most patients could be successfully extubated on the first POD; fiberoptic inspection of the glottic area to assess laryngeal swelling was performed before extubation in every patient. Three out of 58 patients (5.17%) remained intubated on POD 7 and underwent elective tracheostomies. The basis for such rationale including the pre-established duration for tracheostomy was not explained in detail. Kwon et al. [23] further expanded the experience of the group of Emery et al. [1], this time involving CCSS only. Similar risk factors were demonstrated for prolonged intubation past POD 1 (operating time > 10 hours, higher blood loss) with the noted addition of intravenous fluid administration – average rate was 291 mL/hr for patients extubated on day 1 and 1,056 mL/hr for patients with delayed extubation. Kwon et al. [23] ceased to perform fiberoptic inspection of the larynx as previously described but instead relied on the CLT, once again dichotomized to “present” or “absent.” In the only study of major complications in patients undergoing correction of cervical deformity, Smith et al. [24] reported 4 cases out of 78 of respiratory failure (5.1%) from the ISSG database. One patient from this cohort died. It is not mentioned whether these occurred following CCSS, which comprised 29 of 78 patients (27%).
As seen in the literature review, airway management in these patients can be a challenging task with potentially catastrophic consequences. The most cited methods to evaluate the potential for extubation of a CCSS patient include radiological evaluation (lateral radiographs and measurement of the prevertebral soft tissue), fiberoptic inspection of vocal cords, and the CLT. All of these methods are problematic. Prevertebral tissue is always swollen following extensive anterior cervical reconstruction and unexpected vocal cord swelling can quickly develop following extubation. The CLT has been incorporated by at least 2 groups performing CCSS though its evaluation was qualitatively and the test protocol is not detailed nor seems to have been standardized [23]. CLT has been studied extensively in the general medical population and, even though its utility has been contested, it may be an important tool in high-risk extubations [5,25,26]. The most important work on a general medical population for standardization of the CLT was performed by Miller and Cole in 1996 [5]. As in other recent studies, a “positive” CLT has been defined as a leak value below the predetermined threshold value, while a “negative” test is a leak greater than this value. The event predicted by the CLT was respiratory failure due to airway compromise and this was clinically defined as post extubation stridor (PES) in all studies, even if it did not lead to reintubation [5,14]. The first conclusion drawn from CLT studies in a general medical population is that it is difficult to determine a useful test based on the low likelihood of PES in an unselected population, found to be around 4% to 6% [5,14,26]. Secondly, several ventilator parameters affect the actual leak around the breathing tube – whether the cuff remains deflated during both inspiratory and expiratory phases, tidal volume, pulmonary compliance and ventilation mode all have been shown to exert a significant influence. It has been suggested that any leak threshold may be reached if the ventilator is adjusted accordingly [27]. Most studies in the literature have chosen to follow the recommendations of Miller and Cole laid out in 1996; these are noted by the elevated tidal volume according to ideal body weight, which was a common practice at the time. These studies have then measured and not included the CLT in the decision to proceed with extubation; results were then analyzed in a 2×2 contingency table and plotted on a receiver-operating characteristic curve in order to determine the ideal leak threshold. In this manner, values around 110 mL or 15% of the inspired tidal volume have been found to be the most accurate predictors of PES in a general medical population. In these studies, CLT consistently exhibited an elevated negative predictive value (0.97 to 0.99 to predict safe extubation) but highly variable positive predictive value (PPV). No other study could match the PPV of 0.80 reported by Miller and Cole [5] – a much larger series by Kriner et al. [26] reported a dismal value of 0.12, while Antonaglia et al. [28] found a PPV of 0.25 with a CLT of only 70 mL.
A few alternatives have been sought to improve the value of CLT. Simultaneous fiberoptic inspection has been done and, while CLT accurately correlated with the presence of laryngeal swelling, it could still not accurately predict PES in patients undergoing long-term (average, 29 days) intubation [29]. A delta CLT value has been studied, trying to rule out other factors such as ETT diameter or anatomical variations as confounding factors. The CLT was performed immediately after intubation and before tentative extubation: its sensitivity and specificity were worse than the simple pre-extubation CLT [14].
The low PPV raises the question of what action should be taken for patients with a positive CLT – approximately 4 out of every 5 patients in a general ICU still managed to be extubated successfully; should CCSS patients who fail the CLT be offered a tracheostomy without a trial extubation? The literature offers little insight other than raising awareness to the issue. While the observational design of our study does not permit determination of a CLT threshold, we are able to identify a few risk factors within this specific and homogenous group: patient height, calculated AP tracheal diameter, infraglottic luminal area, and tidal volume. Calculated AP tracheal diameter and tidal volume were both based off patient height and so considered dependent variables. Local anatomy (a narrower trachea) is thus shown to be the determinant factor for prolonged intubation, failure to extubate and increased ICU and hospital stay. We had imagined that CLT would plateau after a certain point and in this study, no patient in this group was successfully extubated past POD 7. The senior author has successfully extubated a number of patients between day 7 and day 14 which is considered the time to move to tracheostomy – there are some factors such as depressed mentation, inadequate pulmonary status, or airway secretions that can still improve after day 7. It should be recognized, however that patients not close to extubation on day 7 will probably need a tracheostomy and it is reasonable for the team to discuss with the patient and family. Following this study, the junior author has modified his protocol to allow for extubation of “low-risk” CCSS patients (taller, infraglottic luminal area greater than 170 mm2 – the lowest value in the early group in this study) with a CLT audible to members of all 3 teams involved in patient care (neurosurgery, critical care, and respiratory therapy) to be extubated irrespective of the CLT value. We have had no failures so far and it has also resulted in an average CLT at extubation of 100–130 mL; with lower values, the leak is usually not audible.
CONCLUSION
This study has been very important to our group as its results enabled us to more appropriately counsel patients on expectations during the first few PODs following a CCSS. The results of this study suggest that patients with favorable anatomy, those who are taller with a larger infraglottic luminal area and those undergoing a shorter procedure may benefit from a more flexible airway management protocol. Strictly speaking, our hypothesis was rejected as there were occasional failures to successfully extubate despite a CLT> 200 mL but for most patients, extubation was successful even when the threshold of 200 mL was not reached. Accordingly, we have lately shifted our practice to incorporate a qualitative threshold of “audible leak” over a fixed value, while incorporating close surveillance in the first few hours after extubation to obtain an infraglottic airway if necessary. We hope this study demonstrates recovery from CCSS is not easy and may assist our colleagues to safely navigate this critical period while avoiding the dreaded respiratory failure complication.
Notes
The authors have nothing to disclose.