Correlation of Early Outcomes and Intradiscal Interleukin-6 Expression in Lumbar Fusion Patients
Article information
Abstract
Objective
To determine if there is correlation between intradiscal levels of interleukin-6 (IL-6) and early outcome measures in patients undergoing lumbar fusion for painful disc degeneration.
Methods
Intervertebral disc tissue was separated into annulus fibrosus/nucleus pulposus and cultured separately in vitro in serum-free medium (Opti-MEM). Conditioned media was collected after 48 hours. The concentration of IL-6 was quantified using enzyme-linked immunosorbent assay. Pearson correlation coefficients quantified relationships between IL-6 levels and pre- and postoperative visual analogue scale (VAS) back pain and Oswestry Disability Index (ODI), as well as change in VAS/ODI.
Results
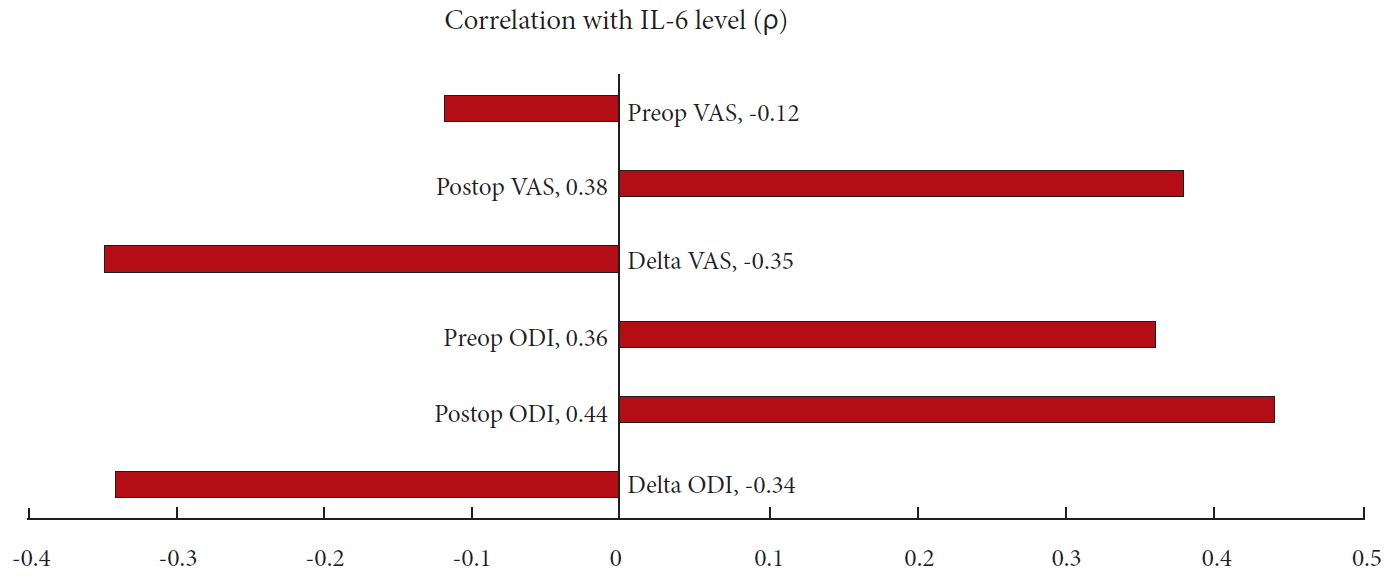
Sixteen discs were harvested from 9 patients undergoing anterior lumbar interbody fusion (mean age, 47.4 years; range, 21–70 years). Mean preoperative and 6-month postoperative VAS were 8.1 and 3.7, respectively. Mean preoperative and postoperative ODI were 56.2 and 25.6, respectively. There were significant positive correlations between IL-6 expression and postoperative VAS (ρ = 0.38, p = 0.048) and ODI (ρ = 0.44, p = 0.02). No significant correlations were found between intradiscal IL-6 expression and preoperative VAS (ρ = -0.12, p = 0.54). Trends were seen associating IL-6 expression and change in VAS/ODI (ρ = -0.35 p = 0.067; ρ = -0.34, p = 0.08, respectively). A trend associated IL-6 and preoperative ODI (ρ = 0.36, p = 0.063).
Conclusion
The direct association between IL-6 expression and VAS/ODI suggests patients with elevated intradiscal cytokine expression may have worse early outcomes than those with lower expression of IL-6 after surgery for symptomatic disc degeneration.
INTRODUCTION
Lumbar disc degeneration is a significant contributor to low back pain [1,2], accounting for almost 10% of lumbar fusion surgery performed in the United States [3]. Despite extensive study of the degenerative process in the disc, the exact cause for discogenic back pain has not been elucidated. Compounding the difficulty in understanding this condition is the fact that only a minority of those with degenerative changes are affected by severe, chronic back pain [4].
Despite the high incidence of surgery for painful disc degeneration, there are no highly accurate testing strategies to confirm the presence of pain at a particular disc level and the results of this type of surgery remain suboptimal. Provocative discography, which has been widely used, has been shown to have the potential for false-positive results [5,6]. Magnetic resonance imaging (MRI) studies, which are quite sensitive to degenerative changes, correlate poorly with pain or disability [4]. Research on the molecular basis of degenerative disc disease has greatly increased our insight into the biology underlying this complex process. In recent years, the role of specific molecules including cytokines and enzymes which degrade the intervertebral disc (IVD) extracellular matrix has become more clear [7]. In addition, expression levels of systemic inflammatory mediators (tumor necrosis factor [TNF]-α, sTNF-R1, interleukin [IL]-1β, and IL-6) have been studied with respect to the incidence of acute low back pain [8]. Associations were found between IL-6 and qualities of pain when combined with depressive symptoms, as well as TNF-α with pain severity and disability when combined with several other factors [8]. In fact, intradiscal injections of antibodies to IL-6 have recently been studied for the treatment of low back pain with promising short-term results [9], with mixed results seen for TNF-α inhibitors [10,11]. In another earlier study comparing disc tissue from patients undergoing fusion or discectomy, there were significantly higher levels of IL-6 and IL-8 in the fusion group, suggesting that these play a role in painful disc degeneration [12].
It is possible that the intradiscal concentration of IL-6 in patients with chronic low back pain may correlate with functional outcomes before and after surgery. A clearer understanding of this potential relationship may help predict which patients will have better or worse outcomes in the postoperative period. Previous work has shown that certain cytokines are more highly expressed in painful disc tissue versus nonpainful disc tissue [13] but correlation with clinical outcome following surgery has never been studied. The goal of this study is to identify the association, if any exists, between the intradiscal IL-6 concentration at a particular disc level and clinical outcome before and after lumbar fusion surgery. We hypothesize that there will be a positive correlation between the levels of expressed IL-6 and the degree of improvement in pain and disability as measured by visual analogue scale (VAS) and Oswestry Disability Index (ODI).
MATERIALS AND METHODS
This study was approval by the Institutional Review Board (IRB) of Jefferson University (IRB No. 08D.525). Patients with severe discogenic low back pain recalcitrant to nonsurgical management were recruited for study participation according to specific inclusion and exclusion criteria. Adult patients (>18 years) scheduled to undergo anterior lumbar interbody fusion (ALIF) for painful lumbar disc degeneration were included. All patients had a positive preoperative discogram (≥ 7/10) at the operated level(s) with a control level (≤ 3/10). Patients had lumbar dominant pain symptoms that were worse with sitting than with standing and had failed at least 6 months of aggressive, nonoperative rehabilitation and nonsurgical care. They also had to be willing and able to participate in the study follow-up regimen and complete preoperative VAS and ODI questionnaires to establish baseline scores. MRI needed to demonstrate Pfirrmann grade III or IV degeneration on MRI of 1 or 2 levels between L3 and S1. Patients were excluded if they had evidence of radiculopathy, symptomatic spinal stenosis, grade II or greater spondylolisthesis, scoliosis greater than 15 degrees, presence of an auto-immune disease including but not limited to rheumatoid arthritis, sclerodema, lupus, ankylosing spondylitis, patients taking immunosuppressive drugs or cytokine inhibitors, patients with prior lumbar fusion, and patients with active litigation or evidence of secondary gain.
1. Tissue Collection and Preparation of the Conditioned Medium
At the time of surgery, disc tissue was collected and transported in saline to the laboratory within 30 minutes of harvest in sterile tubes containing 25-mL phosphate-buffered saline (PBS) and antibiotics (penicillin 100 U/mL, streptomycin 100 μg/mL). Annulus fibrosus and nucleus pulposis were grossly separated based on morphology and were washed 3 times with PBS and finely minced into approximately 3-mm cubes and were weighed in preweighed tubes with 20-mL PBS. The washed tissue pieces were centrifuged at 1,200 rpm for 5 minutes and PBS was completely removed. The washed tissue pieces were then cultured for 48 hours at 37°C (95% humidity) in serumfree Opti-MEM (Gibco, by Life Technologies, Carlsbad, CA, USA) in 25-cm2 culture flasks with a density of 1.0 g tissue/3-mL medium. Conditioned medium (CM) was collected after 48-hour incubation, centrifuged at 1,200 rpm for 10 minutes, filtered at 0.20 µm and the supernatant was stored at -80°C until further use, similar to a previously published protocol [14].
2. Cytokine Assay With Enzyme-Linked Immunosorbent Assay
The concentration of IL-6 in CM was quantified using enzyme-linked immunosorbent assays (Quantikine ELISA kits, R&D System, Minneapolis, MN, USA) according to the manufacturer’s instructions. All samples were assayed in duplicate on the enzyme-linked immunosorbent assay plate overnight at 4°C. Colorimetric absorbance was measured with a BioTek, Synergy HT (BioTek Instruments, Inc. Winooski, VT, USA) and analyzed with 4-parameter-logistic-curve data analysis software (http://www.myassays.com).
After surgery, patients completed VAS and ODI outcomes questionnaires at 6 months. Demographic information for each patient was recorded including age and number of levels fused. In patients with multilevel procedures, the disc with the highest expression of IL-6 was used for statistical analysis. We theorized that the disc with the highest level of IL-6 expression would be the most symptomatic for the patient, and therefore the most representative of the patient’s dysfunction. As some discs had low IL-6 expression, we felt that taking an average would not accurately represent the patient’s level of pain and dysfunction. Pearson correlation coefficients (rho) were used to quantify relationships between expression levels of IL-6 and preoperative and postoperative VAS back pain and ODI, as well as change in VAS back pain and ODI. Significance was assumed for p < 0.05.
RESULTS
Sixteen discs were harvested from 9 patients undergoing ALIF for discogenic pain (mean age, 47.4 years; range, 21–70 years) (Table 1). There were 3 revision cases in which the primary cases were laminectomies without discectomies. Therefore, the disc tissue should not have been significantly compromised by the original surgery. The mean preoperative and postoperative (6 months) VAS back pain scores were 8.1 and 3.7 respectively. The mean preoperative and postoperative ODI (6 months) were 56.2 and 25.6, respectively. There were significantly positive correlations between intradiscal IL-6 expression and postoperative VAS back pain (ρ = 0.38, p = 0.048) and ODI (ρ = 0.44, p = 0.02). These findings suggest that patients with higher levels of intradiscal IL-6 expression have more back pain and higher disability after surgery. No significant correlations were found between intradiscal IL-6 expression and preoperative VAS back pain (ρ = -0.12, p = 0.54). Trends were seen associating IL-6 expression and change in VAS back pain, and change in ODI (ρ = -0.35, p = 0.067; ρ = -0.34, p = 0.08; respectively). This suggests that patients with higher levels of intradiscal IL-6 expression trended towards less improvement in VAS and ODI after surgery compared to those with lower IL-6 expression. Similarly, a trend was seen associating IL-6 expression and preoperative ODI (ρ = 0.36, p = 0.063) (Tables 2, 3) (Fig. 1).
DISCUSSION
This study demonstrates that patients with higher expression of intradiscal IL-6 have more pain and disability in the early postoperative period after ALIF and less pain improvement than those with lower expression of intradiscal IL-6. No significant correlation could be found with intradiscal IL-6 levels and preoperative VAS scores. There are several possible explanations for this finding. First, VAS pain levels are measured at 1-time point, and may not accurately represent a patient’s typical pain over the course of time. A more accurate representation of overall dysfunction is seen with ODI, where trends were seen with IL-6 levels. While other studies have found associations with low back pain and cytokine levels, they compared to either nonpainful discs from surgical specimens or cadaver samples with no history of back pain [12,13]. Our study did not have a control group of nonpainful discs. The Back Complaints in the EldersBrazil study could not find a significant correlation of several serum cytokine levels with “present pain intensity” and “low back pain frequency,” [8] but found associations when models included several other variables. This emphasizes the multifactorial nature of low back pain. In addition, due to the nature of this study, it is not possible to have a control group of patients with no pain or dysfunction. If there was a control group to compare to, it is possible we would find differences in preoperative VAS pain.
Another possible explanation for our findings is that all patients included in our study had some degree of overexpression of IL-6 and disc degeneration. Then, the inflammatory response triggered by surgery increased the pain and disability more so in the patients with higher intradiscal IL-6 concentration, as explained by their worse outcomes within the first 6 months of surgery. Part of the degenerative process of the disc includes ingrowth of nerves [15-17]. This may be the source of exaggerated pain and disability in the postoperative period in those with elevated IL-6 levels. While the disc is removed in its entirety, the ingrowth of nerves from the surrounding tissues likely become irritated from the surgery. In addition, the chronic elevation of IL-6 levels can cause these nerves to be hypersensitive and elicit an exaggerated pain response [7,18-20].
Few studies have attempted to characterize the association between cytokine expression and clinical symptoms in a systematic manner and no studies have attempted to correlate expression of inflammatory mediators with outcome measures after surgical intervention. Burke et al. [12] evaluated cytokine expression differences between the 2 groups of patients: those with discography-positive discogenic pain and those undergoing discectomy for sciatica secondary to herniated nucleus pulposus (HNP). Disc samples from patients with low back pain were found to have elevated expression of IL-6 and IL-8 compared with samples from patients with HNP and sciatica. However, postoperative outcomes were not evaluated. Using a similar study design, Lee et al. [21] evaluated cytokine expression in patients with presumed IVD degeneration presenting with chronic back pain and patients with leg-dominant symptoms with evidence of HNP on MRI. Groups were defined entirely on the basis of MRI findings of HNP and not based on clinical criteria or discography. They found increased expression of IL-8 in patients with greater degrees of disc degeneration and at levels with presumed discogenic pain as opposed to levels with HNP. Similarly, Le Maitre et al. [22] investigated expression of IL-1β and associated regulatory proteins in disc tissue isolated either from postmortem examinations of patients with no history of back pain/sciatica or from patients undergoing anterior lumbar discectomy for lumbar fusion or disc replacement to treat discogenic back pain. This study found that IL-1β expression was significantly elevated in patients with discogenic back pain and that IL-1β was important in upregulation of other genes associated with the degenerative cascade, including degradative enzymes. Our study did not find a correlation of IL-6 and preoperative low back pain, but this may be due to not having a control group of patients with no low back pain.
Limitations to this study include a relatively small sample size and lack of follow-up greater than 6 months. However, there are only small percentage of patients that undergo surgery due to isolated discogenic pain, and our study had strict inclusion criteria. There are numerous other variables that could have contributed to the clinical outcomes, such as fusion rate, body mass index, smoking status, preoperative pain medications, medical comorbidities, etc. While there is variability among the patients, including the number of levels fused, all surgeries included an ALIF followed by posterior stabilization, and were performed by the same surgeon, using the same instrumentation and techniques. The indication, low back pain secondary to disc degeneration, was consistent among all patients. Also, all patients had a positive preoperative discogram. In addition, while there was heterogeneity in the posterior techniques used, the disc tissue was removed during the anterior portion for all patients, which limits the variability that could be seen with different posterior fusion techniques (open versus percutaneous, +/- decompression). While the study design attempted to exclude patients with nondiscogenic pain by performing a preoperative discogram, it is possible that another source of pathology contributed to pain in some patients. Based on our review of previous studies and those from our lab, we focused on evaluating IL-6. However as previously mentioned there are several other inflammatory cytokines that could more strongly correlate with outcomes.
Future studies will attempt to find correlations of preoperative serum cytokine levels and intradiscal cytokine expression. If a direct correlation exists, patients that may have worse outcomes in the earlier postoperative period could be identified earlier, and additional strategies for pain control or alternative treatments such as targeted intradiscal cytokine antibody therapy could be offered. However, without a preoperative test that correlates closely with intradiscal cytokine expression, the results are still theoretical in nature at this time.
CONCLUSION
The direct association between IL-6 expression and VAS/ODI suggests patients with elevated intradiscal cytokine expression may have worse early outcomes than those with lower expression of IL-6 after surgery for symptomatic disc degeneration.
Notes
The authors have nothing to disclose.
Acknowledgements
This work was supported by a research grant from North American Spine Society (NASS Translational Research Grant Awarded to DGA).