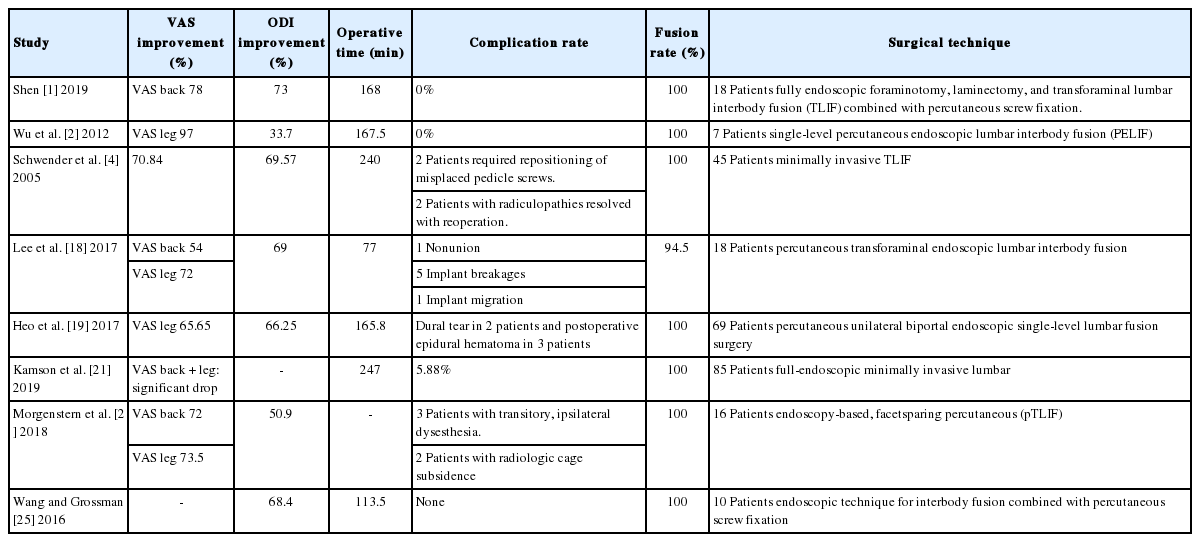
Uniportal Endoscopic Lumbar Interbody Fusion
Article information
Abstract
The cause of radiculopathy is the compression of the nerve root which can be secondary to sliding of the vertebra and reduced disc height. In some patients, decompression alone does not resolve this problem. We describe the uniportal endoscopic transforaminal lumbar interbody fusion technique. Full-endocopic foraminotomy and discectomy are followed by cage implementation and percutaneous instrumentation. The goal of this surgical method is decompression of nerve roots, segment stabilization, disc height, and sagittal alignment restoration. Uniportal endoscopic facet sparing transforaminal transkambin lumbar interbody fusion is a good surgical option to treat degenerative disc disease, mechanical instability, and spondylolisthesis. This method shows favourable clinical outcomes in selected patients.
INTRODUCTION
Transforaminal transkambin interbody fusion is a routinely used and well-accepted technique for lumbar fusion. Goals of the transforaminal lumbar interbody fusion (TLIF) are the decompression of central canal stenosis or exiting nerve roots, enlargement of the neuroforamina through disc height restoration, segment stabilization, and sagittal alignment restoration with an appropriate anterior load bearing.
Conventional TLIF has drawbacks due to long skin incision, muscle stripping, epidural fibrosis, blood loss, and possible nerve root injury. Minimally invasive tissue-sparing approaches became the standard in many areas and are preferred over traditional open surgery in both hospitals and ambulatory surgery centers. Reduced destruction of soft tissue results in reduced postoperative pain and length of hospital stay [1-3]. Despite many advantages, the learning curve especially in endoscopic procedures is a frequently discussed issue [4-7].
The use of full-endoscopic uniportal or biportal techniques with a rigid endoscope is becoming more popular for decompression of central and lateral recess stenosis [8-13].
The tranforaminal region (Kambin triangle) is the key for the transforaminal fusion techniques as it defines the point of entry. The endoscopic transforaminal approach can potentially reduce the risk of uncontrolled bleeding, excessive blood loss, and the need for blood transfusion. The disc height determines the cranial and caudal dimension of the neuroforamen. Length of the pedicle, facet joint arthritis, and hypertrophic yellow ligament influence the size of the Kambin triangle.
Spinal stenosis, disc protrusion, or herniations as well as osteophytes on the endplates of the motion segment can cause a change in position of the neural structures. The traversing nerve root might be shifted anteriorly in spinal stenosis. In case of herniated discs, the exiting nerve root can be pushed to posterior, medial or lateral, depending on the location of the extruded disc material.
Wang et al. [14] report on an effective and safe transforaminal approach utilizing the endoscope to implant an expandable cage. The disc space was filled with bone graft and percutaneous screw fixation was performed under local anesthesia. The use of conscious sedation allows for live neurological monitoring through patient’s feedback.
Critical steps of endoscopic fusion are the avoidance of exiting nerve root injury, preparation of the foramen, decompression of the spinal canal, removal of disc material, and preparation of the endplates [15]. The fusion rate with or without the use of additional bone substitute as well as the prompt postoperative stability appears to be essential and depend on implant design as well as on the technical steps of the surgery.
In order to restrict the rising costs in health care systems short hospital stays and less use of morphines are encouraged in the postoperative care of lumbar fusion patients [14].
MATERIALS AND METHODS
To accomplish a TLIF, first decompression and cage insertion are performed endoscopically. Thereafter screws and rods are implanted percutaneously.
Different types of endoscopic TLIFs are described in the literature. There are uni- and biportal techniques as well as microendoscopic methods [15-20]. The following surgical technique describes a uniportal endoscopic transforaminal TLIF procedure as it is used by the author.
Surgical technique TLIF procedure (steps 1–8)
1. Full-Endoscopic Foraminotomy and Discectomy (Steps 1–4)
Step 1. Preoperative preparation and set-up
Patient selection is an important factor in terms of surgical success, which is why all necessary diagnostic measures must be carried out first. In most cases, radiological imaging with x-ray and magnetic resonance imaging (MRI) is sufficient for diagnosis. Additional computed tomography scans as well as functional x-rays are sometimes necessary to assess disc calcification, bone formation, and stability.
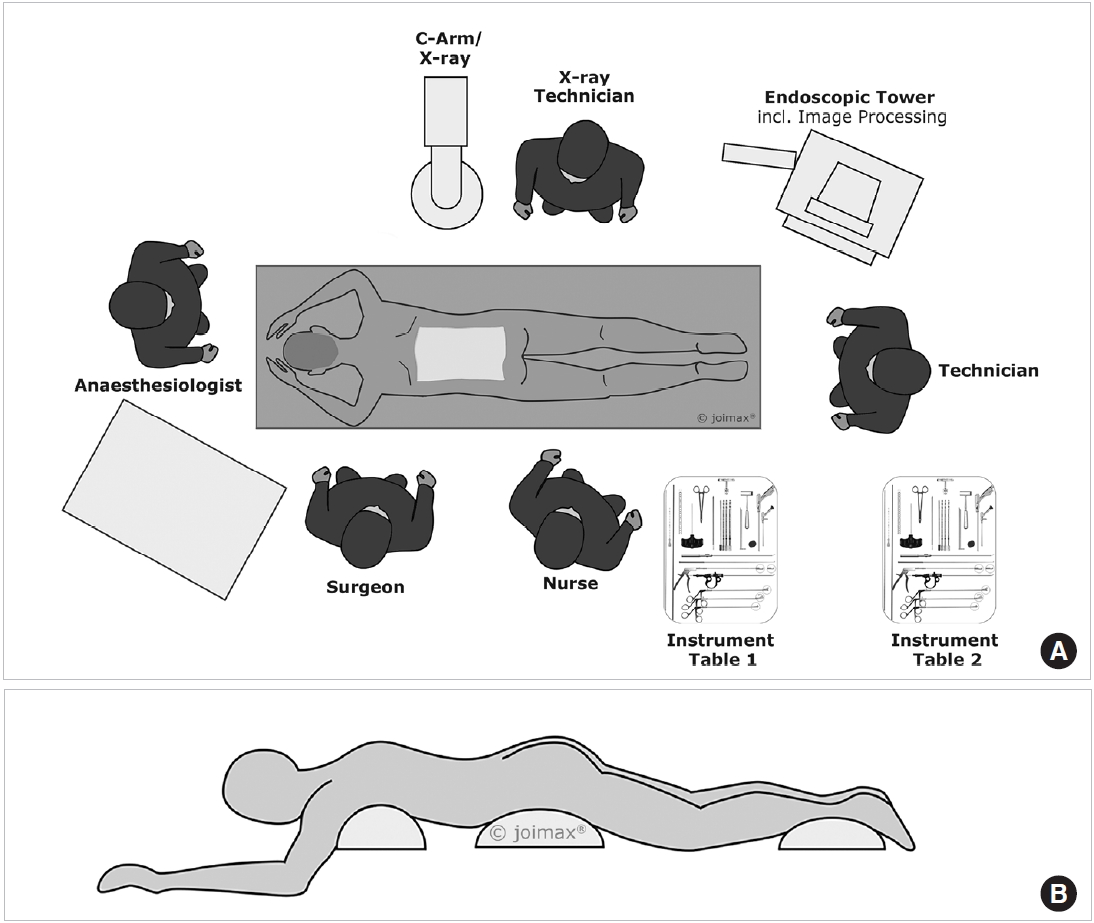
The operating room should provide the surgeon with sufficient space to perform the procedure and the possibility to limit the radiation exposure. The surgery room should be large enough to accommodate the endoscopic tower, C-arm, operation table, and instrument tables. The surgeon stands on the opposite site of the C-arm, the endoscopic tower is positioned at the foot of the patient (Fig. 1A).
Step 2. Anesthesia and patient positioning
The procedure can be performed under local anesthesia with the patient in prone position. A possible scheme for conscious sedation is a combination of Midazolam, Remifentanil plus oxygen via nasal cannula. The drugs are dosed individually by titration and also based on the different stages of intervention. The author prefers general anesthesia without (long lasting) muscle relaxants. Two cushions under chest and hip of the patient or a Wilson frame help to achieve less lordosis of the lumbar spine. At the same time, the pressure is taken away from the inner organs, reducing the risk of epidural vein bleeding (Fig. 1A, B).
Step 3. Approach planning
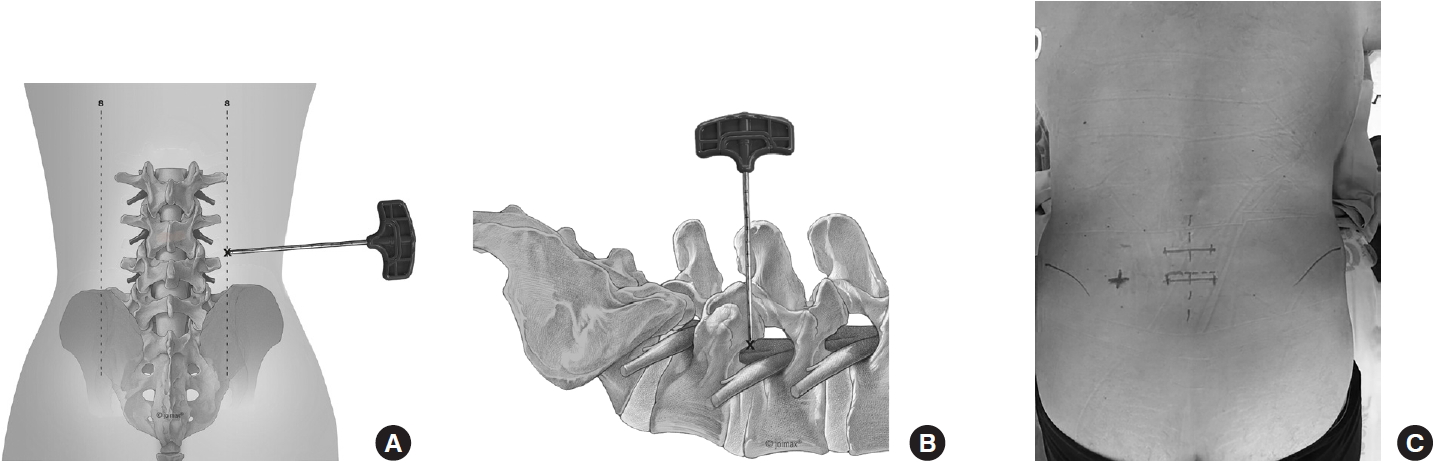
First, the spinous process line, the iliac crest line, pedicular line, and the correct intervertebral disc space are marked. Then the pedicles of the cranial and caudal vertebra are identified and marked to faciliate the later screw placement (Fig. 2A–C).
Depending on the pathology the first step of the surgery might be the screw placement and possible distraction or reduction of spondylolisthesis. Disadvantages of starting with screw placement are less haptic feedback when determining the final implant size due to ligamentotaxis and a lower quality of intraoperative fluoroscopy.
Step 4. Endosopic procedure (Supplementary video clip 1)
The incision should be performed on the clinically symptomatic side. In case of scoliotic deformity, an incision on the convex side of scoliosis could offer the advantages of a larger neuroforamen and more medial position of the traversing nerve root and dural sack.
The entry and incision point are about 8 cm (in most patients between 7 to 9 cm) off the spinous process line depending upon the level of fusion. The needle trajectory ranges from 40°–50°. The target point of the procedure is the center of the disc space into which the implant is inserted. The trajectory should be parallel to the disc space.
A clear anteroposterior (AP) and lateral x-ray without rotation is the key to an exact entry point determination. The author prefers to change the angulation of the table over changing the rotation or angulation of the fluoroscopy.
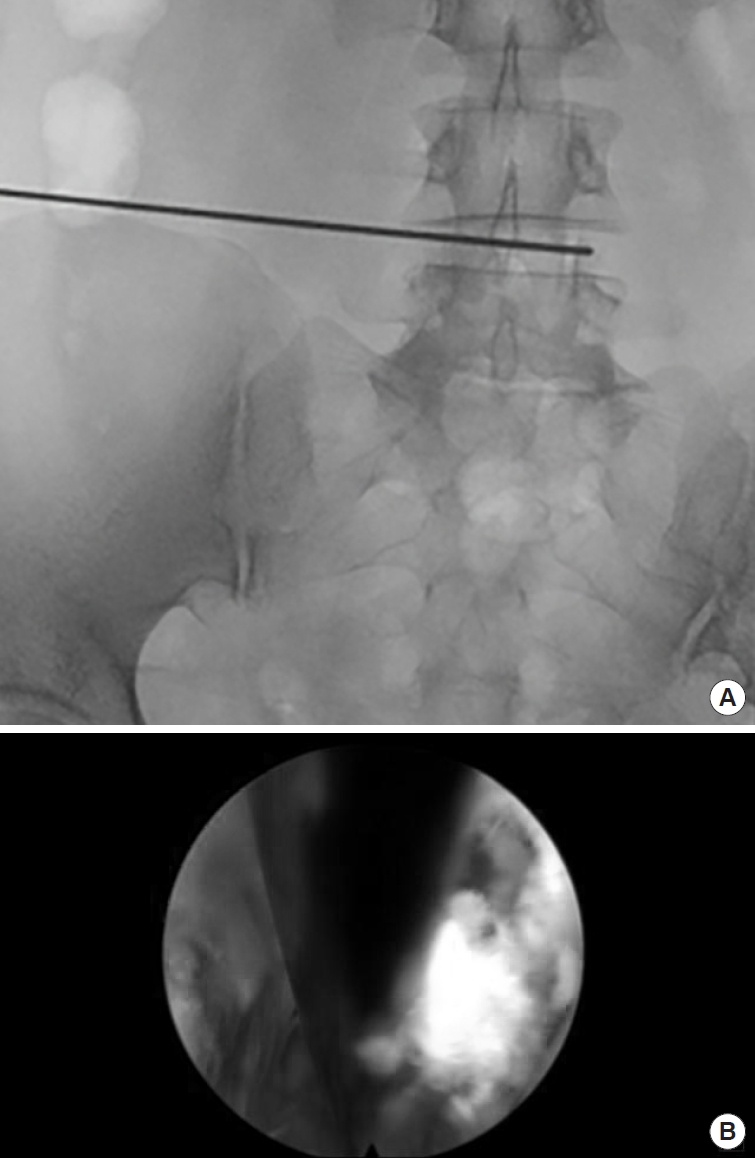
The needle is first advanced to the superior articular process (SAP) and then pushed forward to the posterior longitudal ligament. A haptic response is received when reaching the ligament. In the AP image, the needle tip should touch the posterior ligament and be located in the medial half of the interpedicular line. In the lateral x-ray, the needle tip should still be dorsal to the posterior margin of the vertebras. An oblique image can be helpful to target the center of the disc. The optimal angulation should be 45° to the patient’s back (Fig. 3). Then the needle is advanced to the center of the disc under radiological control in AP (Fig. 4A) and lateral views. The approach is comparable to a discogram - without compromising the parallel placement of the needle to the upper and lower endplate of the disc space. Additionally, methylen blue in combination with radio-opaque contrast fluid can be used to perform a discogram and color the disc.
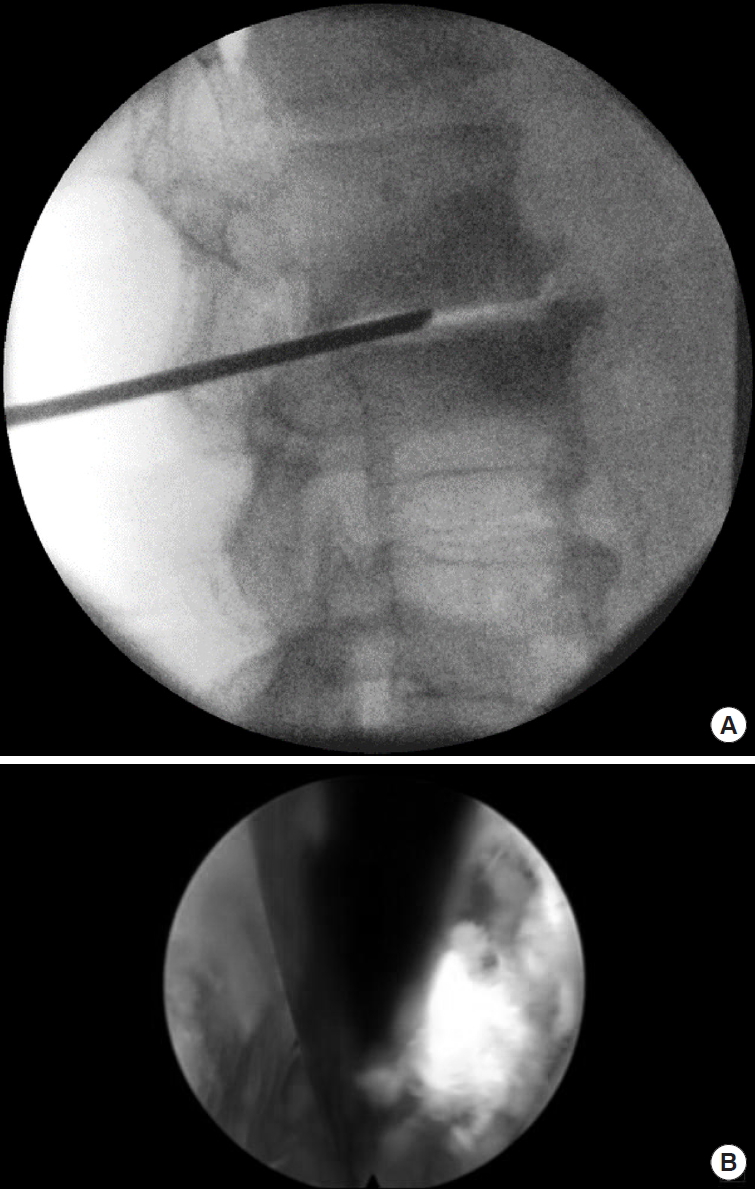
(A) Placement of the needle parallel to the upper and lower endplate at the center of the disc. (B) Intraoperative placement of Kirschner wire.
The guidewire is inserted. The skin and lumbar fascia are incised. In the case of limited space in the neuroforamen, a Jamshidi needle is helpful to perform a trans-SAP approach for a controlled foraminotomy without increased risk of exiting nerve root compromise.
The aim is the anatomical restoration of the neuroforamen by foraminoplasty to create space for the final implant.
The bone drills are used in a small to large diameter sequence to prepare the endplates (Fig. 5A, B). The reaming is done under fluoroscopic guidance. The fluoroscopic control also prevents end plate damage by confirming the correct drill bit diameter corresponding to the hight of the intervertebral disc space. Iatrogenic damage is also prevented by the design of the drill itself. A thorough preparation of the end plate up to the spongiosa is important. This step already allows to estimate the final dimensions of the interbody implant.
The guiding rod and dilators are then advanced over the guidewire into the foramen, followed by the working tube for the endoscope. After removal of the instruments, the endoscope is inserted into the working tube. Disc material is removed and the endplates are inspected. Alternatively to the drills, currettes and spoons can be used to prepare the endplates under fluoroscopic control.
Then the endoscope is retracted together with the working tube to inspect the foraminal area. The upper part of the pedicle as well as the SAP are identified and prepared for foraminoplasty. Next, the traversing nerve root is identified and decompressed. For decompression, the foramen is widened with an endoscopic diamond burr. Then the endoscope is turned towards cranial to identify the exiting nerve root, followed by turning it to inspect and decompress the extraforaminal region. Then the endoscope is rotated back to identify the area of the superior facet, pedicle, and traversing nerve root (so-called arch) and entry to the disc space.
Under visual control, the guidewire is reinserted through the endoscope (Fig. 6A, B) close to the exiting nerve root. Its final postion is controlled with AP and lateral x-ray. The final size of the implant is then determined by using the endplate raspatories and trial implants.
2. Cage Insertion, Endoscopic Control, and Percutaneous Instrumentation (Steps 5–7)
Steps 5. Cage insertion
Finally, the cage with or without bone subtitute is mounted on the cage inserter. It is advanced over the guidewire and can be hammered into the disc space under AP and lateral x-ray control. The final position should be in the center of the disc space in both projections. The cage can then be released and the instrument removed. During this step, a parallel position of the cage should be achieved and confirmed by the AP and lateral x-ray (Fig. 7A–E).

(A) Titanium cage insertion over-the-wire. (B) Cage insertion. (C) Cage insertion. (D) Final control x-ray (anteroposterior) cage placement. (E) Final control x-ray (lateral) cage placement.
The guiding rod, working tube, and endsocope are consecutively inserted and remaining tissue is removed from the foraminal area, allowing a postcage placement view on the implant, traversing nerve as well as exiting nerve root.
Steps 6. Instrumentation
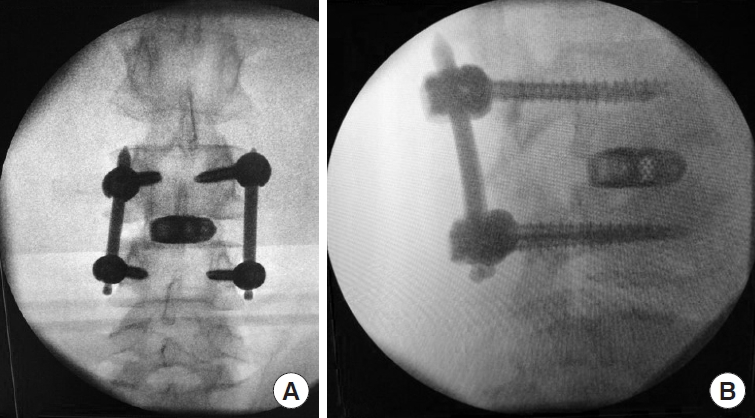
Under fluoroscopic guidance, the pedicles are identified. After inserting the pedicle probe to obtain a perfect intrapedicular placement, the pedicle holes are tapped. The appropiate sized pedicle screws are then placed under fluoroscopic guidance and their position controlled in AP and lateral x-ray images. The rods are inserted after measuring their required length with the measurement tool. Finally, the screws are tightened and the tulips (tabs) are removed.
Final x-rays should then be performed in both views lateral and AP (Fig. 8A, B).
Steps 7. Surgical site management
Constant saline irrigation is used during the endoscopic procedure not only for visualization but also for flushing the surgical site. Intraoperative bleeding can be controlled with the bipolar probe. Usually, a drain is not needed (Fig. 9). When necessary, it can be placed through the working channel of the endsocope, thus avoiding an additional incision. The fascia and skin are closed with 3–4 single sutures.
3. Postoperative Care (Step 8)
Steps 8. Postoperative care
Patients routinely receive a single dose of antibiotics (lasting for 5–7 days), unless there is an increased risk of infection. Glucocorticoids (8-mg dexamethasone) are administered orally on the day of surgery and in case of postoperative leg symptoms in decreasing doses for 9 days (8 mg on days 1–3, 4 mg on days 4–6, 2 mg on days 7–9). However, postoperative dysethesia is rarely seen. Patients are encouraged to do some light activities with a lumbar orthesis in the first 3 weeks postoperative and receive physiotherapy in weeks 3 to 6, starting with isometric and then biplanar exercises. Excessive and heavy activities should be prohibited for the first 3 months postoperative. A control x-ray after 3 months is recommended.
DISCUSSION
The endoscopic approach for TLIF is a well-known procedure in full-endoscopic spine surgery.
Complications related to interbody fusion surgery can include dural tear, nerve root injury, postoperative hematoma, motor weakness, temporary dysesthesia as well as infection. A summary of clinical results, operative time, and complications of selected studies are given in Table 1. Kamson describes his intraoperative complication rate as low as 0% with a total postoperative complication rate at 5.88% [21]. Shen [1] reports in their case series performing full-endoscopic interbody fusion 0% complications. Morgenstern and Morgenstern [22] report no major complications using percutaneous TLIF (pTLIF) with a posterolateral approach. Kolcun et al. [23] found a 4% conversion rate without major intraoperative or postoperative complications in patients who underwent minimally invasive transforaminal fusion without general anesthesia. Ahn [3] notes that the working tube might cause irritation of the exiting nerve root especially in cases of prolonged surgery time. Furthermore, motor weakness as well as temporary dysesthesia are reported as common endosocopic complications. Lumbar fusion is often associated with adjacent segment degeneration. Many complications are associated with the learning curve of minimally invasive TLIF and endsocopic procedures.
Reduction of neurologic injury can be achieved by conscious sedation, neuromonitoring and avoidance of excessive nerve root retraction. A preoperative MRI is useful to detect anatomical anomalies. Another important key factor is the knowledge of anatomical landmarks. The surgeon must ensure that the exact position of the instruments is known during the entire surgical procedure. This is essential to avoid nerve injury and can prevent insufficient decompression of the spinal nerve root.
The hospitalization time is widely varying between the different methods. Morgenstern and Morgenstern [24] report for pTLIF a median postoperative time to ambulation of 6 hours and a median postoperative time until hospital discharge of 26 hours. In contrast, MIS TLIF resulted in an ambulation of 3.2 days (1.9) and a mean postoperative time until hospital discharge of 9.3 days (2.6). Similar results are found by Shen [1] with a mean length of hospital stay of 1.2 days.
Appropriate selection of patients and appropriate surgical indication of the procedure might be key factors in obtaining good results [18]. Osteochondrosis L3/4 and spondylolisthesis ≤ grade 2 can be good indications. Important are a thorough end plate preparation and a high primary stability also to avoid implant migration. The author has not observed pseudoarthosis in his experience of about 50 cases. Clear advantages of the full-endoscopic decompression and cage insertion over open or other mimimally invasive surgery methods are demonstrated in clinical studies. Minimal iatrogenic trauma to muscles and stabilizing structures, less blood loss, shorter hospital stay, quick recovery, and fewer complications could be demonstrated. Direct visualization helps preventing nerve root injury and to verify a complete decompression and correct cage placement [1].
CONCLUSION
Endoscopic TLIF is a good surgical option to treat patients with degenerative disc disease, mechanical instability and/or spondylolisthesis. It minimizes iatrogenic approach-related injury of the segmental structures including the facet joint and shows favorable clinical outcomes in selected patients.
Notes
R. Wagner teaches endoscopic spine surgery for joimax and is joimax Faculty Member. M. Haefner is employee of joimax GmbH.
SUPPLEMENTARY MATERIALS
Supplementary video clip 1 can be found via https://doi.org/10.14245/ns.2040130.065.v.1.