Risk of Revision After Vertebral Augmentation for Osteoporotic Vertebral Fracture: A Narrative Review
Article information
Abstract
Osteoporotic vertebral fractures (OVFs) can hinder physical motor function, daily activities, and the quality of life in elderly patients when treated conservatively. Vertebral augmentation, which includes vertebroplasty and balloon kyphoplasty, is a commonly used procedure for OVFs. However, there have been reports of complications. Although serious complications are rare, there have been instances of adjacent vertebral fractures, cement dislocation, and insufficient pain relief due to cement failure, sometimes necessitating revision surgery. This narrative review discusses the common risks associated with vertebral augmentation for OVFs, such as cement leakage and adjacent vertebral fractures, and highlights the risk of revision surgery. The pooled incidence of revision surgery was 0.04 (0.02–0.06). The risks for revision are reported as follows: female sex, advanced age, diabetes mellitus, cerebrovascular disease, dementia, blindness or low vision, hypertension, hyperlipidemia, split type fracture, large angular motion, and large endplate deficit. Various treatment strategies exist for OVFs, but they remain a subject of controversy. Current literature underscores the lack of substantial evidence to guide treatment strategies based on the risks of vertebral augmentation. In cases with a high risk of failure, other surgeries and conservative treatments should also be considered as treatment options.
INTRODUCTION
Osteoporotic vertebral fractures (OVFs) can cause low back pain and inhibit physical motor function, activities of daily living, and quality of life in elderly patients treated conservatively [1-3]. Furthermore, OVFs increase the risk of mortality in elderly women [4]. Therefore, OVFs exert a significant health burden, especially in an aging society [5]. While a decreasing trend in the incidence of hip fractures is observed in many countries, occurrence of OVFs is increasing [6,7]. Recently, a declining trend in osteoporotic medication after OVFs is reported in the United States [8]. The treatment for osteoporosis should be improved to preserve population health by increasing the medication rate. There is no universal conservative treatment including bed rest, spinal orthosis, and physical therapy. Basically, immobilization owing to bed rest can be harmful because of the deterioration in bone loss, progressive muscle weakness, thromboembolic disease, joint contracture, and skin ulcer in elderly population [4]. Therefore, physical therapy under spinal orthosis is recommended after short immobilization period, although a recent prospective cohort study [9]. demonstrated that better compression ratio of vertebral body and lower surgical rate were obtained following 2 weeks of bed rest for patients with risk factors of poor prognosis. Physical therapy is effective to improve osteoporosis and prevent osteoporotic fracture (OF) [10,11]. In most cases, conservative treatment employing brace treatment is effective for the healing process of fractures. However, there is no apparent evidence regarding the type of spinal orthosis between soft and rigid brace [12]. Despite conservative treatment, residual back pain and neurological deficit are observed due to nonunion, severe deformity, and global spinal imbalance [13-15]. Particularly, nonunion is strongly associated with residual back pain compared to local alignment [16].
Vertebral augmentation comprising vertebroplasty and balloon kyphoplasty is a widely used procedure for OVFs, and its safety and usefulness have been reported in many papers [17-19]. Vertebral augmentation provides pain relief and vertebral heigh restoration [20]. As the population ages, cement augmentation is being considered as a treatment for patients who are increasingly older, specifically those who are more than 80 or 90 years old [21]. Additionally, a database study demonstrated that vertebral augmentation can improve the survival rate [22]. Cochrane systematic review [23]. shows weak evidence of vertebral augmentation for vertebral fracture in acute or subacute phase because randomized controlled studies could not find the difference between vertebroplasty and placebo procedure. Therefore, we considered the risk of vertebral augmentation to minimize the harm among elderly patients, which may improve the outcomes of this procedure. The timing of vertebral augmentation is important for surgical outcomes because there are changes in low back pain and compression ratio especially during the first 3 months. A prospective study demonstrated that the quality of life and low back pain improved until 3 months after injury and did not change thereafter until a mean follow-up of 5 years [24].
Previous papers suggest that late intervention worsen the severity of fracture [25,26] as compared to early intervention. However, early intervention may include unnecessary cases with OVFs, which recovery by conservative treatment. Therefore, several papers demonstrated the predicting factors for poor prognosis after conservative treatments to minimize the surgical invasiveness in elderly patients [3,13,18]. There have also been some reports of complications following surgery. Serious complications, which occur in less than 1% of cases [27], include cement leakage into the spinal canal, spinal cord injury, infection, and pulmonary embolization, which may require emergency treatment. Apart from serious complications, there have been cases of adjacent vertebral fractures or dislocation of cement and poor pain relief due to cement failure [28], which sometimes require revision surgery [29-32]. To the authors’ knowledge, there have been no reviews on the incidence and risk of revision surgeries after vertebral augmentation.
The purpose of this study was to investigate the incidence of revision after vertebral augmentation. In addition, we discuss the common risks of vertebral augmentation for OVFs, including cement leakage and adjacent vertebral fractures, and outline the risk of vertebral augmentation.
MATERIALS AND METHODS
We conducted a systematic review of the literature identifying revision surgery after vertebral augmentation in patients with vertebral fractures according to the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines and with guidance from the Cochrane Handbook for Systematic Reviews of Interventions.
1. Literature Search
For the identification of eligible articles, the individual steps of title and abstract screening, full-text review, and data extraction were performed independently using the MEDLINE database, Cochrane library, and Scopus database. The search strategy included combinations of the terms “vertebroplasty” or “balloon kyphoplasty” or “vertebral augmentation” and “revision” or “reoperation” and “vertebral fracture.” To optimize data mining, word variations and exploded medical subject headings were included whenever feasible. The last literature search was performed on January 9, 2023.
2. Study Selection and Quality Assessment
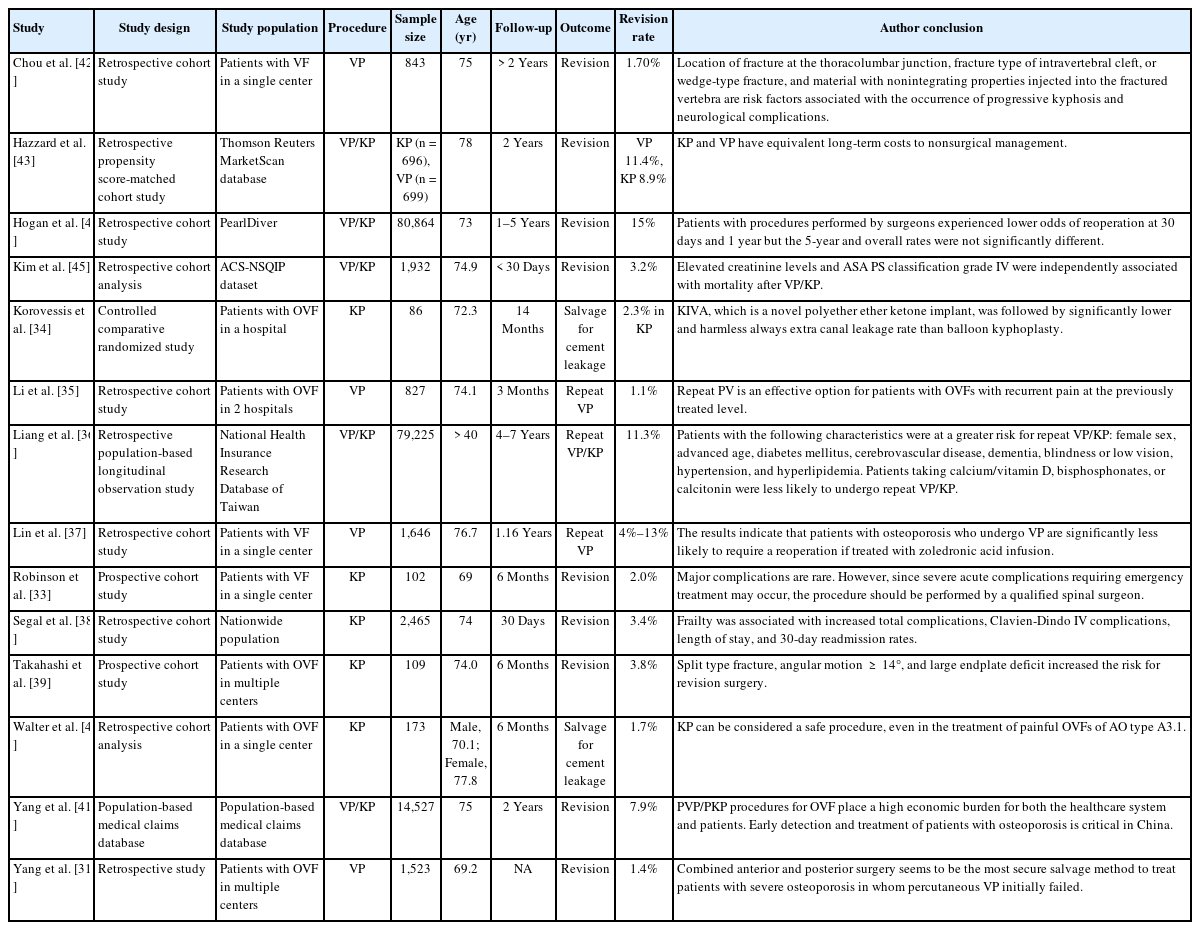
First, incomplete articles and non-English language articles were removed (Fig. 1). Next, review articles and case reports were excluded. Additionally, the articles that included only vertebral augmentation with instrumentations, only trauma cases, ankylosing spondylitis, pathological fracture, cervical spine, and sacral fracture were excluded. Next, the articles on only revision cases after vertebral augmentation were excluded because of lacking revision rate. Finally, we evaluated prospective or retrospective cohort studies to assess the incidence of revision surgery and the risks [31,33-45] (Table 1). The initial screening of titles and abstracts for relevance was conducted by a single reviewer (ST). Full texts of the remaining articles were obtained and assessed for eligibility by comparison with the inclusion criteria. Data were extracted from the included studies by 2 independent reviewers (ST and MI). Any discrepancies during the screening or extraction process were resolved by consensus agreement or adjudication with another author (KT). The risk of bias in each study was evaluated by 2 reviewers (ST and KT). The post intervention biases for selection and reporting were judged using the Risk Of Bias In Non-randomized Studies of Interventions tool as follows: low, moderate, serious, and critical [34] (Table 2). Confounding and information biases were excluded from the assessment because the reviewed articles were not compatible.
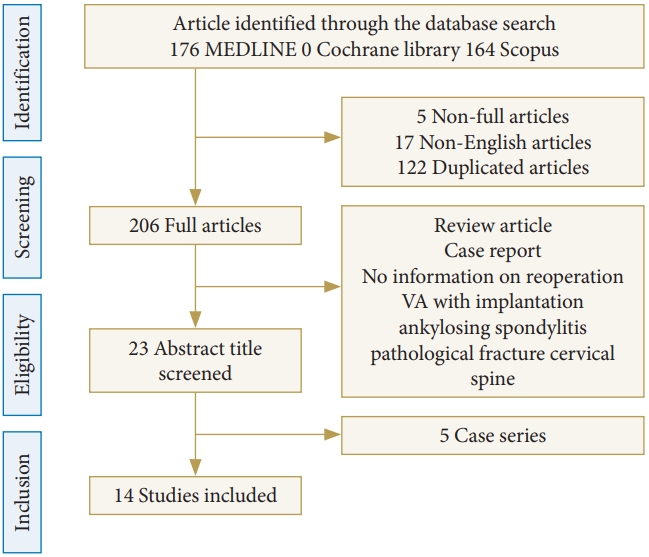
Flow chart of the literature search for systematic review of revision surgery after vertebral augmentation. VA, vertebral augmentation.
3. Data Extraction
Information on the study population, type of intervention, original sample size, average or median age, follow-up period, outcome of the study, revision rate, and risk of revision was collected. Revision surgery included failure of vertebral augmentation, repeated vertebral fracture for new fractures, and salvage for cement leakage. The authors’ main conclusions regarding revision or repeated surgery were summarized.
4. Data Analysis
Pooled incidence of revision after vertebral augmentation was obtained by random-effect meta-analysis using a normal-binomial generalized linear mixed model method in the R packages “meta” (version 4.2.3, R Foundation for Statistical Computing, Vienna, Austria) [46].
RESULTS
A flow diagram of the systematic literature search is provided in Fig. 1. The database search yielded 206 eligible articles after the removal of incomplete, duplicated and non-English articles. During the title and abstract screening, 206 articles were excluded for not meeting the inclusion criteria or for meeting the exclusion criteria. Of the remaining 19 articles, a further 5 records were excluded during full-text screening because they analyzed only revision cases. Finally, 14 articles were used for qualitative synthesis.
Six articles were nationwide or insurance database studies. Five studies were retrospective cohort and 2 were prospective cohort studies. One article was a randomized controlled trial that evaluated KIVA system, which is a novel polyether ether ketone implant, and balloon kyphoplasty [34]. Several studies confirmed osteoporosis as the cause of vertebral fractures, while the other studies included patients who underwent vertebral augmentation for vertebral fracture. The median age in most studies was > 70 years. Although the definition of revision surgery was different due to differences in the purpose of the studies, it was divided into revision surgery, repeat vertebral augmentation, and salvage surgery for cement leakage. In several articles, revision surgery included repeat vertebral augmentation and salvage surgery. The rate of revision ranged from 1.1% to 13%. Repeat vertebral augmentation was relative frequent [35-37]. Repeat vertebral augmentation was performed for new fractures. Salvage surgery was rarely performed for infection (0.36%). The frequency of salvage surgery for cement leakage was 1.7% to 2.3% [34,40]. The pooled incidence of revision surgery was 0.04 (0.02–0.06) (Fig. 2).
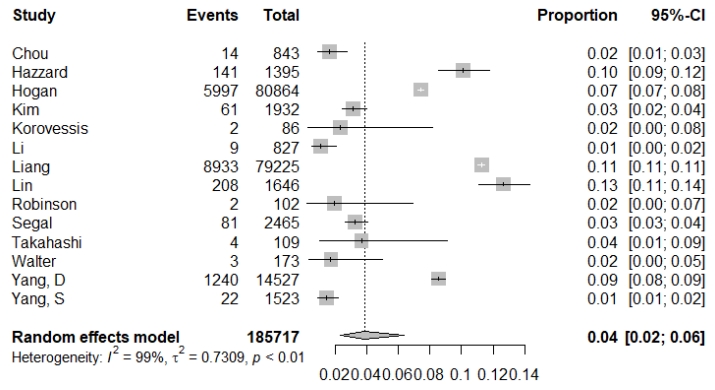
Random effects meta-analysis of the incidence of revision surgery after vertebral augmentation. CI, confidence interval.
The risk factors for revision were reported in several studies [36,39,42]. Chou et al. [42] demonstrated location of fracture at the thoracolumbar junction, fracture type of intravertebral cleft, or wedge-type fracture, and material with nonintegrating properties injected into the fractured vertebra were risk factors associated with the occurrence of progressive kyphosis and neurological complications. Database study demonstrated the following other risk factors for repeat vertebral augmentation: female sex, advanced age, diabetes mellitus, cerebrovascular disease, dementia, blindness or low vision, hypertension, and hyperlipidemia. Patients taking calcium/vitamin D, bisphosphonates, or calcitonin were less likely to undergo repeat vertebral augmentation. Another study [39] showed that split type fracture, greater angular motion, and large endplate deficit increased the risk for revision surgery. Additionally, frailty, elevated creatinine levels, and American Society of Anesthesiologists physical status classification grade IV were independently associated with mortality and complications [38,45]; treatment of osteoporosis, an experienced surgeon, and prophylactic vertebral augmentation may reduce the risk [36,37]. Vertebroplasty and kyphoplasty showed similar revision rates [43].
DISCUSSION
Revisions following vertebral augmentation have been reported previously. However, few reports have investigated the risk factors for revision. The risks for revision are as follows: female sex, advanced age, diabetes mellitus, cerebrovascular disease, dementia, blindness or low vision, hypertension, hyperlipidemia, split type fracture, angular motion ≥ 14°, and large endplate deficit [36,39,42]. Furthermore, intradiscal leakage may lead to secondary adjacent vertebral fractures [29,47] and a greater angular motion of the fractured vertebra may be associated with greater vertebral height reduction in vertebral augmentation [39]. A significant reduction in vertebral height is also known to be a risk factor for adjacent vertebral fracture [48]. Additionally, the presence of an intravertebral cleft with angular motion has been reported as a poor prognostic indicator after vertebroplasty [49]. A greater angular motion might indicate breakage or dysfunction of the anterior spinal elements, including the anterior longitudinal ligament and annulus, which may lead to failure in retaining the cement with the vertebral body. Anterior dislodgment of cement causes a loss of vertebral height and stability.
There is no standard classification of OVFs. Genant grading has been used for screening and evaluating the impact of OVFs on population health based on a semiquantitative assessment [50]. AO spine classification for thoracolumbar injury is also widely used to evaluate the fracture type and to aid therapeutic and surgical decision making [51]. However, the classification is adequate for trauma because OVFs are fragile fractures caused by minor injury. Recently, the new classification of OVFs, the OF classification, which is a morphologic classification of different types of OVF, was developed by the German Orthopedic and Trauma Society and was also adopted by AO Spine [51]. The classification comprises 5 subgroups as per the OVF severity including endplate fracture and posterior wall injury assessment: OF 1, no deformation (vertebral body edema in MRI-STIR); OF 2, deformation of one endplate without or with only minor posterior wall involvement; OF 3, deformation of one endplate with distinct posterior wall involvement; OF 4, deformation of both endplates with/without posterior wall involvement; and OF 5, injuries with anterior or posterior tension band failure. Additionally, a scoring system was developed for making decisions regarding surgical intervention using the information on severity of osteoporosis, deformity progression, pain, neurological symptoms, mobilization, and health status. The severity of OF was associated with the frequency of surgery and 2% patients required revision surgery. Further research is necessary to investigate the surgical outcome depending on this new classification.
Progressive kyphosis after vertebroplasty is a risk factor for revision surgery and the incidence is reported as 1.5% in a retrospective study [42]. There are risk factors associated with the occurrence of progressive kyphosis and neurological complications after vertebral augmentation, including location of fracture at the thoracolumbar junction, fracture type of intravertebral cleft, or wedge-type fracture, and material of nonintegration properties injected into the fractured vertebra [42,52]. Additionally, significant associations were found between cement distribution patterns and progressive kyphosis in cemented vertebrae, which affected the clinical outcome in patients after vertebral augmentation. The cement distribution included uninterlocked solid pattern, discontiguous trabecular pattern, and solid lump cement pattern [53,54]. The distribution of cement in vertebrae contributes to noncemented cancellous bones without load transfer causing recollapse. Another report stated that [55] insufficient cement distribution is responsible for unrelieved pain. Location of the fractured area is an independent risk factor for the occurrence of insufficient cement distribution; if the fracture is located in the superior portion of the index vertebra, there is a higher incidence of insufficient cement distribution. The recollapse after vertebral augmentation may lead to recurrent back pain and revision surgery; the latter is sometimes necessary to avoid serious consequences. Multiple balloon dilation, location of cement and cement volume may be important to avoid the recollapse.
A previous study [31] summarized the possible revision strategies for failed vertebral augmentation. The surgical strategy to treat cement leakage into the spinal canal causing neurological deficit is urgent laminectomy and fusion. Cement dislodgement or fragmentation also requires anterior or posterior surgery. For infection, extensive debridement is necessary, with combined anterior and posterior surgery being the safest method to treat this complication. Augmentation of pedicle screw fixation, using various bone cements, is less evident, although its use as an initial procedure to improve fatigue strength of instrumentation among patients with severe osteoporosis has been previously investigated [56]. Major complications are rare. However, since severe acute complications requiring emergency treatment may occur, the procedure should be performed by a qualified spinal surgeon [33]. Additionally, for patients with symptomatic failed vertebral augmentation, posterior transpedicular approach surgery with circumferential removal of leaked cement and anterior reconstruction show low complication rate and good clinical outcomes; this can be considered as an alternative method combining anterior and posterior approaches [57].
Vertebral augmentation carries the risk of bone cement leakage and pulmonary embolism [58,59]. Although symptomatic pulmonary embolism is a rare condition [60], pulmonary cement embolism leading to death can occur after uncontrolled leakage [61]. Pedicle violation caused by epidural cement leakage further carries the potential risk of neurological deficit [62]. Therefore, vertebral augmentation should only be performed by experienced surgeons following the identification of critical indications under fluoroscopic or computed tomography (CT) monitoring [61].
Minor cement leakage is frequently noted on CT but is asymptomatic in most cases [63]. Indeed, leakage has been reported to occur in 30%–65% of cases [64]. Decreased integrity of vertebral walls and the volume of injected cement significantly boost the potential risk of cement leakage [65]. Cement leakage adjacent to a disc is frequently encountered, while symptomatic neurological complications due to compression of a nerve root or the spinal cord are less frequent. Vertebral augmentation is also a viable option for the treatment of OVFs, even with posterior wall involvement [66]. High-viscosity cement results in lower bone cement leakage rate and better VAS score improvement compared with low-viscosity cement [58,67]. Application of a large void volume using the balloon and the smaller injected cement than that void might be useful to avoid leakage [66]. The unilateral approach may decrease the incidence of cement leakage due to lower cement dosage [68]. Symptomatic bone cement displacement, which causes poor outcomes after vertebral augmentation is reported to be approximately 2% along with the risks of intravertebral cleft, anterior leakage, and cement distribution [55].
Several reports have investigated the difference between vertebroplasty and kyphoplasty. A meta-analysis [69] that evaluated 121 reports demonstrated that the rate of asymptomatic cement leaks per treated patient was significantly higher for vertebroplasty than for kyphoplasty, although both procedures were effective in symptomatic vertebral compression fractures and there was no difference in mortality. Another meta-analysis further demonstrated that kyphoplasty has a reduced risk of cement leakage and it increases the postoperative vertebral height compared to vertebroplasty; however, it is more expensive and requires longer operative time durations [70]. However, several papers in the current review showed no differences in clinical outcomes and complications between vertebroplasty and kyphoplasty [43,44,71].
The incidence ratio of adjacent vertebral fracture after vertebral augmentation has been calculated as approximately 10%–40% in previous reports [28,72-80]. Most fractures occur within a few months. Vertebral augmentation is known to reduce vertebral kyphotic deformity at the fractured vertebrae, as reported in several previous studies [49,81]. However, correction after vertebral augmentation should be considered in the area including the upper and lower spinal segments. Even if correction can be achieved by kyphoplasty, correction loss may occur following adjacent vertebral fracture or correction loss of the vertebral body. Hard cement may further result in increased mechanical pressure, eventually causing a new fracture of the endplate in the adjacent vertebral body [79]. More severe wedge angle before surgery, correction degree, old OVF presence, and thoracolumbar levels were identified as predictive factors of adjacent vertebral fracture in previous studies [65,82]. Furthermore, a change in Cobb angle after operation and diabetes have been reported as a risk factor for postoperative adjacent vertebral fracture [65]. A correction degree of > 10° was further reported as an independent risk factor for adjacent vertebral fracture [82]. Several papers [73,75,83] have shown that decreased bone mineral density (BMD) increases the risk of adjacent vertebral fracture following vertebroplasty. Furthermore, cement leakage has been reported as the primary risk factor for new vertebral compression fractures [79,84]. However, if the cement is not in close contact with the endplates, it does not increase endplate deformation in the adjacent vertebrae, thereby minimizing the risk of adjacent vertebral fracture [85]. Several studies have developed scoring systems to predict adjacent vertebral fracture based on the identified risk factors [82,86,87]. The usefulness of these scoring systems may be revealed in the future.
Most adjacent vertebral fractures heal by conservative treatment [82]. The application of smaller volumes of cement has been shown to be effective in decreasing the risk of adjacent vertebral fractures, while maintaining sufficient stability [88]. Prophylactic augmentation into non-fractured vertebra may be effective to prevent further fractures and minimize the risk of revision surgery in osteoporotic patients [89]. However, several studies indicated that the cause of adjacent fracture was mainly related to the progression of osteoporosis rather than the vertebral augmentation [90] and the efficacy is inconsistent across the studies. Recent meta-analysis suggested that prophylactic augmentation could not reduce the risk of revision surgery [91]. The therapeutic effects of teriparatide were better than those of the combined vertebroplasty and an antiresorptive agent in fracture prevention, BMD increase, and sustained pain relief [74]. However, considering that most of the adjacent vertebral fractures occur within a few months, and the effect of increasing BMD appeared at least 3 months after the start of teriparatide use [92], teriparatide should be administered at least before surgery. A randomized controlled trail [93] showed that teriparatide might prevent adjacent vertebral fracture due to stimulation of bone formation and faster improvement of bone strength and quality than antiresorptive agents. Teriparatide may offset the pharmacy cost due to the reduction of the inpatient admission and repeat vertebral augmentation [94]. In addition, a reduced BMD might be a surrogate marker in patients with reduced activity of daily living because mechanical loading can inhibit bone resorption and increase bone formation [95].
As the limitation of this study, the criteria for revision surgery is not consistent according to the study population and surgeon’s decision. The lack of uniform definition in a heterogeneous topic can limit the generalizability of the study findings. However, it is important to recognize the possibility and risk for revision surgery after vertebral augmentation in OVF. Furthermore, the follow-up period is relatively short, and the long-term outcomes of the revision surgery are not evaluated. Additionally, publication bias may exist in this review. The investigators who did not identify the revision cases might not submit their data. Finally, this study did not assess the cost-effectiveness of revision surgery, which could have provided valuable information for healthcare providers and policy makers.
CONCLUSION
There are numerous treatment strategies for OVFs, which remain controversial. Current reviews have documented the low level of evidence currently available to inform the treatment strategy based on the risk of vertebral augmentation. Therefore, it is necessary to carefully evaluate the fracture to determine all relevant surgical indications. In cases with a high risk of failure, other surgeries and conservative treatments should also be considered as treatment options. However, implant complications such as cage subsidence, screw loosening, pull-out, and junctional failure are common in older patients with osteoporosis. Therefore, further investigation is necessary to create treatment strategies for OVFs.
Notes
Conflict of Interest
The authors have nothing to disclose.
Funding/Support
This study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author Contribution
Conceptualization: ST, HT, HN; Data curation: ST, HI, KT, MI; Formal analysis: ST, MI; Methodology: HI, KT, MI, HT, HN; Writing - original draft: ST; Writing - review & editing: ST, HT, HN.