A Propensity Score-Matched Cohort Study Comparing 3 Different Spine Pedicle Screw Fixation Methods: Freehand, Fluoroscopy-Guided, and Robot-Assisted Techniques
Article information
Abstract
Objective
This study aimed to compare the accuracy of robotic spine surgery and conventional pedicle screw fixation in lumbar degenerative disease. We evaluated clinical and radiological outcomes to demonstrate the noninferiority of robotic surgery.
Methods
This study employed propensity score matching and included 3 groups: robot-assisted mini-open posterior lumbar interbody fusion (PLIF) (robotic surgery, RS), c-arm guided minimally invasive surgery transforaminal lumbar interbody fusion (C-arm guidance, CG), and freehand open PLIF (free of guidance, FG) (54 patients each). The mean follow-up period was 2.2 years. The preoperative spine condition was considered. Accuracy was evaluated using the Gertzbein-Robbins scale (GRS score) and Babu classification (Babu score). Radiological outcomes included adjacent segmental disease (ASD) and mechanical failure. Clinical outcomes were assessed based on the visual analogue scale, Oswestry Disability Index, 36-item Short Form health survey, and clinical ASD rate.
Results
Accuracy was higher in the RS group (p < 0.01) than in other groups. The GRS score was lower in the CG group, whereas the Babu score was lower in the FG group compared with the RS group. No significant differences were observed in radiological and clinical outcomes among the 3 groups. Regression analysis identified preoperative facet degeneration, GRS and Babu scores as significant variables for radiological and clinical ASD. Mechanical failure was influenced by the GRS score and patients’ age.
Conclusion
This study showed the superior accuracy of robotic spine surgery compared with conventional techniques. When combined with minimally invasive surgery, robotic surgery is advantageous with reduced ligament and muscle damage associated with traditional open procedures.
INTRODUCTION
Spinal fusion is the primary treatment for degenerative diseases accompanied by spinal instability. The current standard for spinal fusion is posterior lumbar interbody fusion (PLIF) and pedicle screw fixation. Accurate screw insertion is crucial for a successful fusion procedure. Misplaced screws can lead to not only unstable fusion but also direct damage including neurovascular injuries and bony structure destruction, affecting the clinical outcomes of patients [1,2].
The conventional open surgical approach offers the advantage of direct visualization. However, extensive exposure is associated with significant tissue damage involving the muscles, ligaments, and facet joints. This can lead to severe postoperative pain and adjacent segment degeneration, requiring frequent reoperations [3,4]. To address these drawbacks, minimally invasive surgery (MIS) techniques such as transforaminal lumbar interbody fusion (TLIF) and percutaneous pedicle screw fixation have become popular. The entry and trajectory of screws for percutaneous placement can be determined by the tactile perception of the puncture needle on repeated and continuous fluoroscopic imaging. Although these MIS methods make use of image-guided systems, the placement of screws still requires human involvement, which reduces the accuracy of screw placement [5,6]. Additionally, the increase in radiation exposure during continuous fluoroscopic imaging for screw verification raises concerns about patient and surgeon health [7,8].
Robot-assisted surgery has attracted attention as an alternative treatment method due to the limitations of conventional and MIS procedures. Robotic systems allow accurate screw insertion by precisely following the preoperative planning and trajectory plan without the need for extensive tissue exposure or human intervention. This method significantly reduces radiation exposure and minimizes human subjectivity; thus, it is the most optimal approach for precise screw placement. Various studies have demonstrated the accuracy of robot-assisted surgery [8-10].
Our institution has also used a robotic surgical system known as CUVIS-spine (CUREXO Inc., Seoul, Korea) for the past 3 years. This is the first study incorporating clinical outcomes from the CUVIS-spine, a novel robotic spine surgery platform. We aimed to confirm its accuracy in robotic spine surgery and its noninferiority compared with conventional surgical methods by examining the radiological outcomes and the clinical improvement of spinal fusion patients. Comparing both C-arm guided and freehand screw insertion, widely used in the field of spine surgery, can help in selecting the surgical method.
MATERIALS AND METHODS
1. Study Design and Patient Eligibility Criteria
The design and protocol of this propensity score-matched retrospective cohort study were approved by the Institutional Review Board (IRB) of Yonsei University Severance Hospital (IRB No. 1-2020-0025). This trial was conducted in accordance with the principles outlined in the 2013 Declaration of Helsinki. Written informed consent was obtained from all patients included in this research. The STROBE (STrengthening the Reporting of OBservational studies in Epidemiology) statement was applied.
We included 162 patients who underwent surgical treatment for lumbar degenerative disease from September 2020 to July 2022. The participants were tracked for a mean of 2.2 ± 0.6 years (with a minimum of 1-year follow-up). We conducted a comprehensive review of electronic medical records (EMRs) and imaging data to evaluate baseline characteristics and follow-up outcomes.
The inclusion criteria were as follows: (1) a single surgical level, (2) persistent low back pain or radiating leg pain with neurogenic claudication lasting for more than 3 months, (3) lumbar degeneration evident on magnetic resonance imaging (MRI), (4) unresponsiveness to conservative treatment for a duration exceeding 3 months, (5) a general state of health suitable for withstanding anesthesia during surgery, and (6) a minimum follow-up period of 12 months. The exclusion criteria included: (1) unclear or suspicious symptoms, (2) previous instrumented spine surgery, (3) fractures or dislocations, (4) structural damage or deterioration of vertebral bodies, and (5) presence of infections or metastatic diseases.
We initially identified 54 patients who fulfilled the criteria for robot-assisted mini-open PLIF surgery (RS group, performed by SY). Subsequently, to facilitate propensity score matching (PSM), we enrolled 200 patients for each of the C-arm guided MIS-TLIF (CG group, performed by JYP) and freehand open PLIF (FG group, performed by DKC) surgeries, who satisfied the inclusion criteria. Each surgeon has been performing lumbar spinal fusion surgeries using the assigned approach for many years. We conducted PSM to ensure an equal number of patients in each group (54 patients) for analysis.
2. Surgical Techniques
All patients were operated on in the prone position under general anesthesia with confirmation of the spinal level by fluoroscopy. Three skilled surgeons conducted each surgical procedure. Each surgeon has more than 25 years of clinical experience in university hospitals.
1) Robot-assisted mini-open PLIF (robotic surgery, RS group) [11]
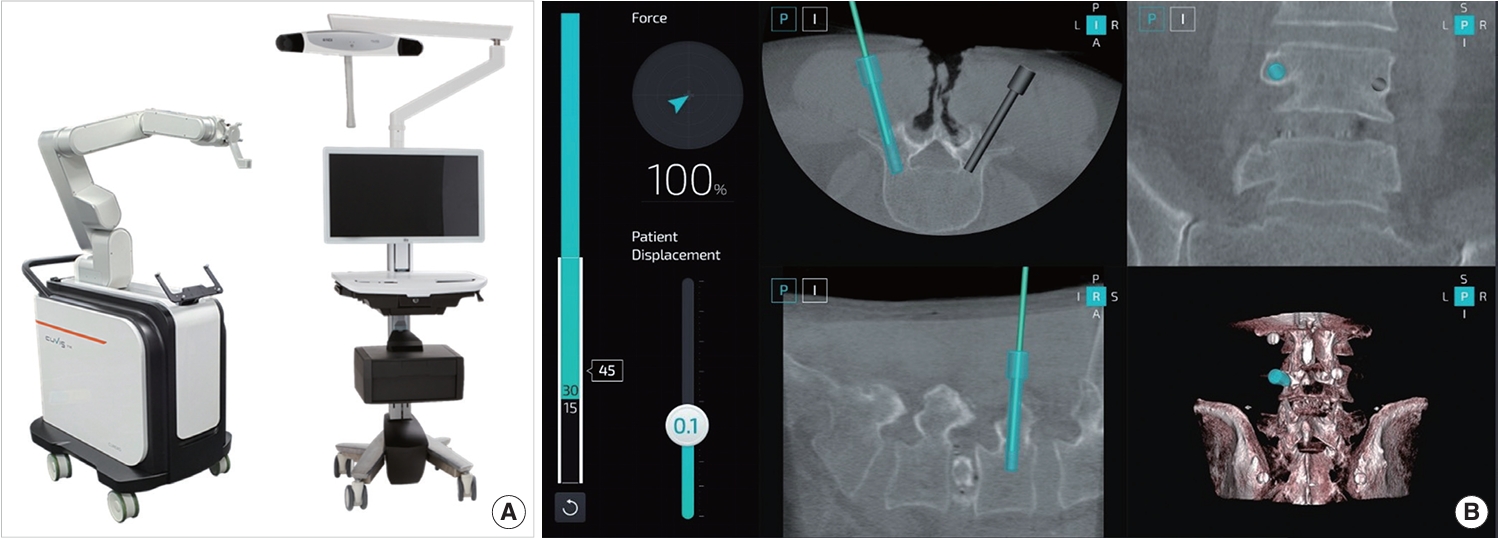
A skin incision ranging from 6 to 8 cm was made in the middle of the lesion site. A monopolar cautery was used to separate the fascia and paraspinal muscle from the spinous process and bilateral lamina subperiosteally. Subsequently, a high-speed drill and surgical tools were used for subtotal laminectomy. The surgical process included bilateral facetectomy, discectomy, and the placement of an interbody cage. A percutaneous reference tracker was positioned on the spinal process, followed by C-arm scanning and automatic registration. Then, surgeons plan the screw trajectories, with the robot subsequently guiding itself toward the chosen trajectory and indicating the entry point and direction. The guiding cannula was closely approached to the skin. Surgeons drilled the guidewires through the guiding tube into the pedicle. A fluoroscopic scan confirmed their position. Finally, pedicle screws were percutaneously inserted along the guidewires for fixation (Fig. 1).
2) C-arm guided MIS-TLIF (C-arm guidance, CG group) [12]
Paramedian incisions measuring 3 to 4 cm in length were created on the symptomatic side. Subsequently, a series of soft tissue dilators were introduced through the incision, reaching down to the facet complex. Facetectomy, discectomy, bone grafting, and the placement of an interbody cage were carried out using a tubular retractor. Further paramedian incisions were made on the opposite side, and pedicle screws were percutaneously inserted with the assistance of a C-arm fluoroscopic system.
3) Freehand open PLIF (free of guidance, FG group) [13]
Freehand open PLIF is the most conventional and fundamental posterior fusion method. The PLIF procedure in the RS group was identical to that in the FG group. The only difference was the need for a larger skin incision (around 8–10 cm) along the midline for screw insertion. In comparison with mini-open PLIF, a more extensive muscle dissection was performed in order to expose the facet joints, allowing for confirmation of the screw entry point and creating a sufficient medial angle. When inserting the screws freehand, the margin of the pedicle wall was directly visualized through laminectomy, facetectomy, and discectomy before insertion. Subsequently, the screw position was confirmed by intraoperative x-ray.
3. Evaluation
1) Preoperative spinal condition
Basic spinal alignment parameters, including pelvic tilt, pelvic incidence, lumbar lordosis, and sacral slope, were measured. Computed tomography (CT) and MRI were employed to assess disc and facet degeneration in the adjacent segments of the operational level. The disc condition was graded on the Pfirrmann scale [14] from 1 to 5, and the facet condition was measured on a scale of 0 to 3 following Weishaupt et al. [15]. To determine pedicle diameter, the width and height were measured. We performed the measurement procedure by selecting the most comparable preoperative and follow-up axial images at the same spinal level for comparison using anatomic landmarks.
2) Accuracy
Accuracy was evaluated by the Gertzbein-Robbins scale [16] (GRS) and superior facet joint violation assessment as described by Babu et al. [17] (Babu) postoperatively. The proposed parameters (GRS and Babu) were analyzed using the postoperative CT scan image. Accuracy can be categorized according to radiological parameters; however, as this was the primary outcome of our study, we have dedicated a separate section to this. PACS (ZeTTA PACS, TaeYoung Soft Co., Seoul, Korea) was used to obtain measurement data. The screw position was classified into 5 grades: grade A (screw entirely within the pedicle), grade B (pedicle cortical breach of less than 2 mm), grade C (pedicle cortical breach of 2 mm or more but less than 4 mm), grade D (pedicle cortical breach of 4 mm or more but less than 6 mm), and grade E (pedicle cortical breach of 6 mm or more) (Fig. 2). The grades A and B screw positions were considered clinically acceptable, whereas all other grades indicated improper placement. The Babu parameter describes the extent of proximal facet joint involvement. The criteria for grading the violation of the facet joints are as follows: grade 0 indicates that the screw is not within the facet; grade 1 suggests that the screw is in the lateral facet but not within the facet articulation; grade 2 implies the penetration of the facet articulation by the screw; grade 3 indicates that the screw travels within the facet articulation (Fig. 3). We conducted GRS assessment for 216 screws and Babu classification for 108 upper instrumented screws in each of the 3 groups consisting of 54 patients. To comprehensively assess the collective impact of these graded screws within a single patient, we used the sum of scores (GRS total and Babu total) assigned to each screw. Higher scores indicated higher accuracy levels. For GRS total, grade A received 5 points, and grade E received 1 point. For Babu total, grade 0 was assigned 4 points, and grade 3 received 1 point. Two neurosurgeons, blinded to the study, assessed the CT scans to determine the grading based on the radiological slice showing the greatest deviation from the pedicle. This evaluation considered the axial, coronal, and sagittal views. Additionally, we explored the orientation of failure in the placement of pedicle screws.
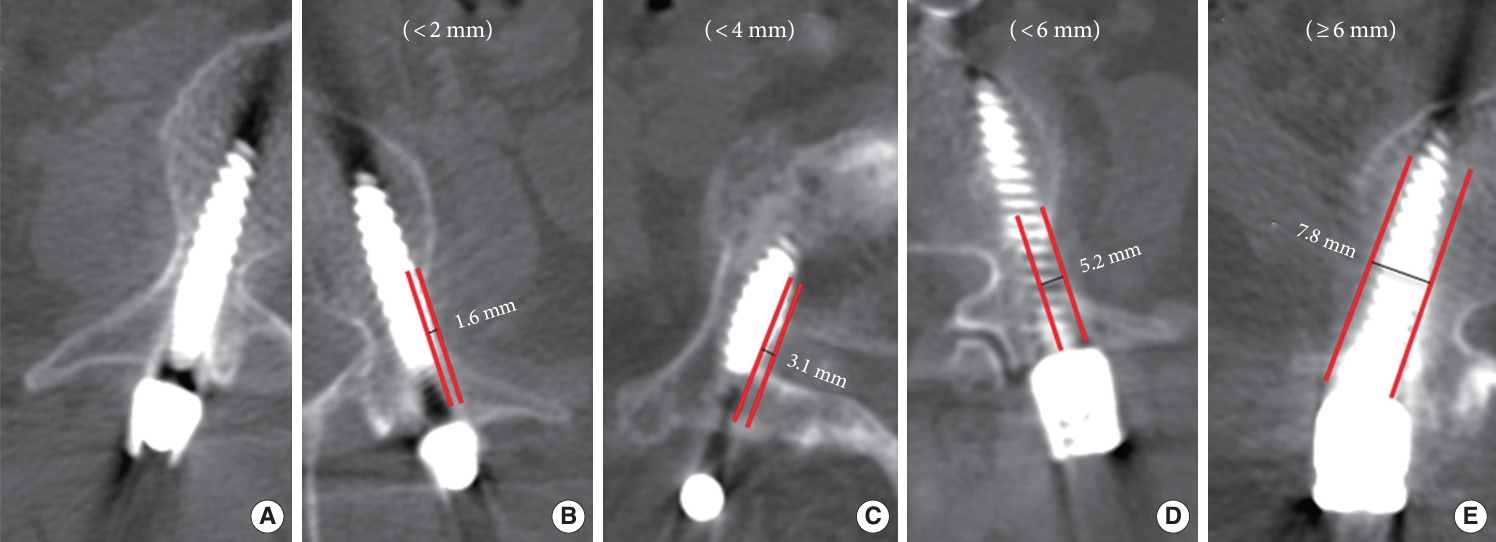
Computed tomography scans demonstrating the Gertzbein-Robbins classification. (A) Grade A: screw precisely within the pedicle; (B) grade B: pedicle cortical breach of <2 mm; (C) grade C: pedicle cortical breach of <4 mm; (D) grade D: pedicle cortical breach of <6 mm; and (E) grade E: pedicle cortical breach of ≥6 mm.
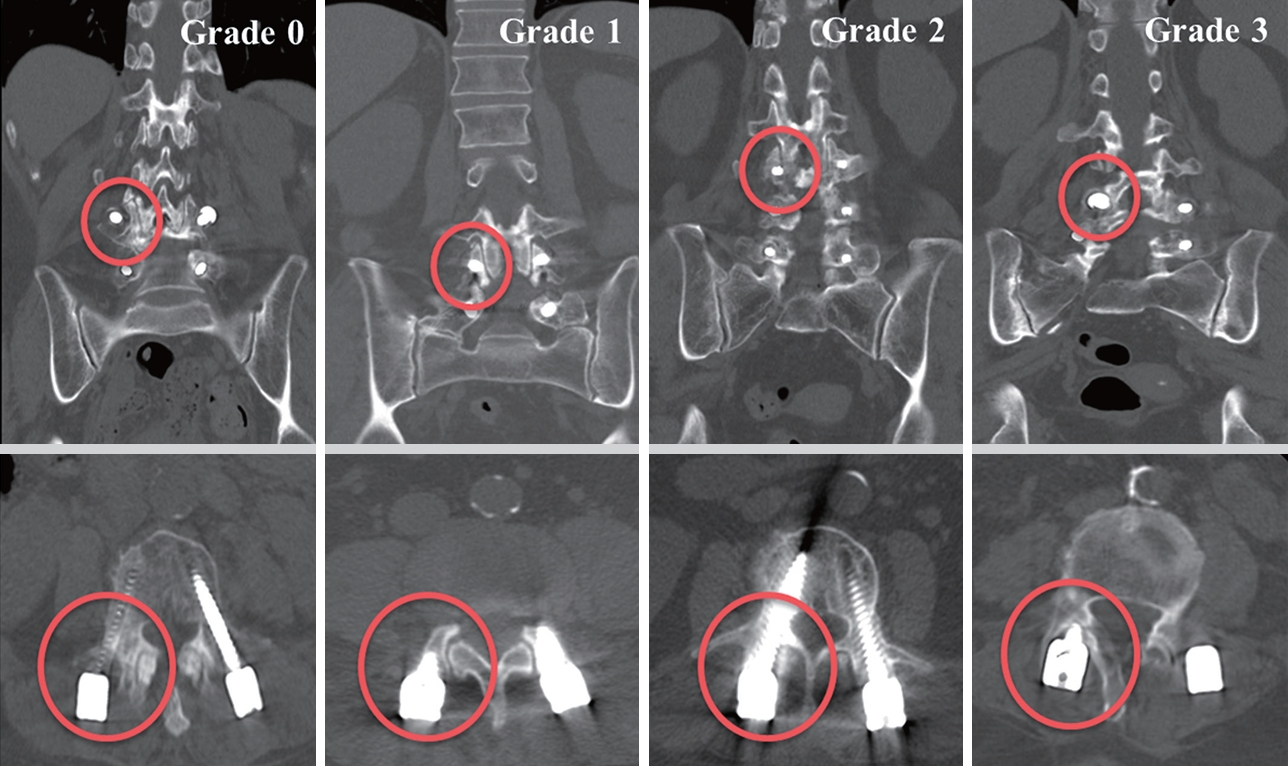
The Babu classification for grading the proximal violation of the facet joints. The violated screw has been marked with a red circle. Grade 0: a screw position outside the facet joint; grade 1: location of the screw in the lateral facet but not within the facet articulation; grade 2: penetration of the facet articulation by the screw; and grade 3: screw trajectory within the facet articulation.
3) Radiological parameters
Radiological results were analyzed, which involved using Weiner classification [18] for radiological adjacent segmental disease (ASD) and mechanical failure. Radiological instability at the adjacent level was assessed using specific criteria and graded as 0, 1, 2, or 3. Grade 0 indicates no degeneration, with a normal disc height, no spur formation, and no listhesis. Grade 1 implies mild degeneration, characterized by less than 25% narrowing of the disc height, small spur formation, and no listhesis. Grade 2 suggests moderate degeneration, with 25%–75% narrowing of the disc height, moderate spur formation, and listhesis ranging from 3 to 5 mm. Grade 3 indicates advanced or high-grade degeneration, featuring more than 75% narrowing of the disc height, large spur formation, and listhesis exceeding 5 mm. These criteria were used to evaluate radiological instability at the adjacent level. The occurrence of radiological ASD was defined as grades 2 and 3 according to Weiner’s classification. Mechanical failure was assessed based on the presence of a halo, screw pull-out, instrument fracture, and cage subsidence. Two neurosurgeons (mentioned in the accuracy section) reviewed the same PACS system.
4) Clinical outcomes
We reviewed outpatient medical records and questionnaires based on EMRs. Clinical ASD refers to symptomatic ASD, encompassing the concept of surgical ASD. This is defined as the emergence of clinical symptoms and signs attributed to the degeneration of adjacent segments [19]. We evaluated the occurrence of clinical ASD by assessing the presence of related symptoms in the outpatient follow-up and examining readmission and reoperation rates in the EMR. Patient-reported outcome measures (PROMs), which included the visual analogue scale, Oswestry Disability Index, and 36-item Short Form health survey, were obtained at both baseline and 12 months after surgery, allowing us to confirm any clinical improvement.
4. Statistical Analysis
We performed the Shapiro-Wilk test to determine if continuous variables followed a normal distribution, which showed a normal distribution. The average values and standard deviations are used to present these variables. On the other hand, categorical variables are presented them as counts and percentages (%). To compare groups, we performed PSM, based on the patient demographics (age, body mass index, and bone mineral density) and preoperative spinal condition (surgical level, pedicle diameter, and articular degeneration). Those factors are known to be associated with accuracy, ASD, and mechanical failure [20-23]. We used appropriate statistical methods such as analysis of variance and the chi-square test. Survival analysis and log-rank test were employed to account for the difference in follow-up periods between ASD rate and mechanical failure, ensuring an examination of the disparities in each group. We conducted Cox regression analyses not only for comparisons among groups but also for the entire patient cohort. We aimed to identify potential factors influencing radiological and clinical ASD, and mechanical failure by hazard ratios (HRs).
Statistical significance was defined as a 2-sided p-value less than 0.05. All statistical analyses were conducted using R ver. 4.1.0 (The R Foundation for Statistical Computing, Vienna, Austria).
RESULTS
Following PSM, 54 patients for each of the RS, CG, and FG groups were included in the final analysis. The demographics, baseline PROMs, surgery-related outcomes, complications, and preoperative spinal condition in the 3 groups are presented in Table 1, most of which showed no significant differences. The RS group required a longer operative time (243.3 ± 43.6 minutes, p < 0.001) and shorter hospital stay (8.5 ± 1.6 days, p < 0.001).
1. Accuracy
A detailed list of the pedicle screw placement accuracy grades is presented in Table 2. In the classification according to the GRS, the proportions of grades A and B screws indicating acceptable positioning in the RS, CG, and FG groups were 98.6%, 92.6%, and 95.8%, respectively (grade A: 92.1%, 72.2%, and 83.8%). In the classification according to the Babu scale, the proportions of grade 0 in the RS, CG, and FG groups were 84.3%, 77.8%, and 63.0%, respectively. Based on GRS and Babu total (the overall effect of the screws on a single patient), the RS group showed significantly higher accuracy (GRS total, p < 0.001, RS/FG vs. CG; Babu total, p < 0.001, RS/CG vs. FG). The direction of screw deviation was most frequently observed in the lateral direction. Among screws graded as C, D, E, there were no caudal deviations, and in terms of medial deviations, 2 were identified in the FG group (Table 3).
2. Radiological Parameters
Significant differences were not observed among the 3 groups in Weiner classification, radiological ASD, and mechanical failure (Table 4). In addition, survival analysis did not reveal significant differences among the groups (p > 0.05). The overall radiological ASD-free survival for RS, CG, and FG was estimated as a mean of 784.4, 801.6, and 808.0 days, respectively. For mechanical failure-free survival, the values were 800.7, 790.3, and 807.0 days (Fig. 4). In Cox regression conducted for radiological ASD and mechanical failure in all patients, significant explanatory variables were identified. In the case of radiological ASD, GRS, Babu total and preoperative facet degeneration demonstrated an effect. The HRs of GRS and Babu total were 0.78 (0.68–0.89, p < 0.001) and 0.43 (0.34–0.53, p < 0.001), respectively. Preoperative facet degeneration also exhibited a significant HR (1.71; 95% confidence interval [CI], 1.11–2.62; p = 0.014). Mechanical failure, GRS total, and age had a significant effect (HR, 0.84 and 1.11; p < 0.05). Table 5 summarize the results of Cox regression.

The Kaplan-Meier survival curves for each group were compared using the log-rank test. (A) Radiologic adjacent segment disease (ASD). (B) Mechanical failure. (C) Clinical ASD. RS, robotic surgery; CG, C-arm guidance; FG, free of guidance.
3. Clinical Outcomes
All clinical results were improved during the follow-up period in all groups. Postoperative PROMs and clinical ASD rates did not significantly differ among the 3 groups (Table 4). Similarly, no differences were observed in the survival analysis. The overall clinical ASD-free survival for RS, CG, and FG was estimated as a mean of 799.4, 790.3, and 807.0 days, respectively (Fig. 4). In most cases of clinical ASD, conservative treatment was provided, and extensive fusion surgery was performed only in 2 cases in the FG group. We conducted Cox regression in a similar manner for clinical ASD, which revealed that GRS, Babu total and preoperative facet degeneration were significant explanatory variables (HR, 0.75, 0.47, and 2.72; p < 0.05) (Table 5).
DISCUSSION
As society ages, spine surgeons increasingly encounter challenging surgeries required for degenerative patients, including revisions and severe scoliosis treatment. Particularly in elderly patients, complications related to screws, such as nerve and vascular injuries, could be life-threatening [24]. In these circumstances, the need for robot-assisted techniques is warranted. Robot-assisted spine surgery is rapidly gaining popularity, primarily due to its accuracy and other advantages including safety and decreased radiation exposure. Additionally, the use of robots could reduce the fatigue of surgeons, allowing them to maintain consistent performance [25,26]. At present, the application of robotics in spinal surgery is predominantly focused on pedicle screw insertion. However, research is being conducted in areas such as bony resection, including laminectomy and facetectomy, as well as deformity correction and tumor removal [27,28].
There might be differences in the performance of various platforms; nevertheless, this study primarily focused on a specific platform, CUVIS-spine (CUREXO Inc.). Notably, compared with other robotic systems, this platform allows real-time detection. It can navigate forces in real time, providing details on the lateral force and patient displacement during surgery. This ensures accurate screw positioning by identifying and addressing any deviations from the intended path. The real-time alarm system allows immediate corrections for excessive force or displacement, empowering surgeons to adjust the screw path or entry points as needed. The robotic system’s functions, the surgeon’s skill, and haptic feedback may collectively enhance precision in pedicle screw placement, reducing errors throughout the surgical process [29].
In this study, we conducted a comprehensive review of the accuracy, radiological results, and clinical outcomes of robot-assisted surgery through a comparison with conventional surgical methods (C-arm TLIF and freehand open PLIF). We initially performed PSM to ensure no significant differences among the 3 groups concerning not only demographics but also preoperative spinal conditions. An overlooked aspect in previous studies, this matching process allows our research to provide more reliable information, as it addresses factors that may influence the accuracy of screws, ASD rate and the occurrence of mechanical failure. Due to the registration process and navigation setup time, the surgical time was observed to be longer in the RS group. The RS group had a shorter hospitalization period, which mainly resulted from hospital policy. Therefore, making it an unreliable indicator of postoperative recovery or daily life transition, which primarily depends on patient functional improvement. In cases of root injury within the complications, it was not a violation caused by screws, but rather all damages occurred during foraminotomy.
Various studies have demonstrated the accuracy of robot-assisted surgery, and our research findings also confirmed a high level of accuracy in the RS group, considering the pedicle diameter. This comprehensive consideration enhances the objectivity of the data. When examining clinically acceptable screws, including grades A and B, based on the GRS, similar levels of accuracy were evident across all 3 groups. Interestingly, contrary to expectations, the FG group exhibited a higher level of accuracy compared with that of the CG group. Some studies have reported results similar to ours in this regard, which may be attributed to the ability to directly visualize the anatomy of the pedicle. In addition, it is possible to confirm the margin and perform insertion through tactile sensation. Verification of the screw threads after insertion is also part of the process, thus ensuring accuracy [9,30,31]. The lower accuracy observed in the CG group may be attributed to the lateral entry point adjustment to avoid facet violation and concerns about the dural structure, which did not seem to offer sufficient convergence. In Table 3, the CG group showed a higher proportion (83.3%) of lateral deviation, suggesting a pattern of screw malposition in the CG group. The Babu scale was used as a measure of accuracy, which could indicate proximal facet violation. The RS group demonstrated superiority, whereas the FG group was inferior compared with other groups. To create the entry point for screws outside the facet joints in open surgery, extensive skin incision and muscle dissection are required. Even with a longer incision to create an entry point outside the facet joints without violation, there is often tension on the muscles and fascia, making it challenging to achieve convergence. In most cases, screw insertion unavoidably includes a portion of the facet that is closer to vertical alignment rather than the ideal convergence. Certainly, since this study does not specifically focus on the entry point, offset, and trajectory, it may be challenging to assess the fixation patterns of screws for each surgical method. Further research addressing these tendencies seems necessary.
Differences in accuracy could affect radiological and clinical outcomes. Therefore, we conducted an analysis in relation to typical issues encountered in fusion surgery, including ASD and mechanical failure. ASD can be broadly categorized into radiological ASD, which exhibits significant imaging abnormalities, and clinical ASD, which shows symptomatic manifestations [32]. In studies with a mean follow-up duration of less than 5 years, ASD has been observed in 5%–25% of patients [33]. Kim et al. [34] reported rates of 9.8% for radiological ASD and 1.6% for ASD requiring surgery during a follow-up period of 20.5 months. A comparison of the 3 groups showed no significant differences in terms of radiological ASD, clinical ASD, and mechanical failure. We conducted a relatively short-term study with a mean duration of 2.2 years. To maximize the application of all available follow-up data for comparison, we investigated the occurrence and time of events, and incorporated survival analysis into the study. In the log-rank test as well, there was no significant difference observed among the 3 groups. Considering the low occurrence rate, it is unlikely that a significant difference would be observed in our study period. Therefore, Cox regression was conducted for the entire cohort of 162 patients. This allowed us to identify factors that may have a significant effect on patients experiencing ASD or mechanical failure. The results showed that GRS, Babu total and preoperative facet degeneration primarily influenced ASD, whereas GRS total and age played a role in mechanical failure. For preoperative facet degeneration, numerous studies have investigated its association with ASD [34,35]. In a biomechanical study, Kim et al. [36] reported that a facet violation model yielded an increase in the facet contact force and intradiscal pressure. Proietti et al. [37] also found that screw invasion of the facet joints increased the rate of degeneration of the facet joints at 6–8 months after percutaneous screw placement. The issue of screw loosening in mechanical failure is known to be caused by pull-out strength. Increasing the major diameter with a longer insertion depth can enhance strength. Biomechanical studies have been conducted to continuously reduce pull-out forces [38]. In the case of robot-assisted surgery, through planning, an optimal size can be confirmed in advance, and insertion can be performed without breaches. The mechanisms underlying the influence of age remain unclear. There is substantial research indicating that the muscle condition may significantly influence the occurrence of ASD and mechanical failure [34], which is presumed to be related to factors such as frailty, accompanied by sarcopenia and consequent back muscle atrophy. However, various mechanisms associated with aging require further research. A comparison of postsurgical PROMs, which could indicate clinical improvement, did not reveal significant differences among the 3 groups. However, the follow-up period was limited. As established in a previous study [39], if there were differences in terms of ASD or mechanical failure, they would likely be reflected in the extent of clinical improvement.
Nevertheless, we compared each surgery in a blinded manner. However, it should be noted that our study was retrospective, and there may be potential bias associated with robot-assisted surgeries due to their inherent sensitivity toward accuracy, which may have influenced the results. Furthermore, there was a lack of data on radiation exposure, and the lack of standardization in materials such as screws, rods, and cages used in fusion surgery was also a limitation. It was challenging to precisely determine the repositioning within the surgical field based on the surgical records. Additional research is warranted considering that the evaluation of ASD and mechanical failure did not take into account factors such as the extent of muscle involvement or adjacent disc degeneration. The results should be further validated in a prospective study with a larger patient population and an extended follow-up period.
CONCLUSION
Owing to the ability to precisely insert screws within the pedicle and minimize facet joint invasion, robot-assisted surgery was proven to be the most superior in terms of accuracy. Although the follow-up period was relatively short, no significant differences were observed among the groups. However, our study found that the overall accuracy of screw placement could influence ASD and mechanical failure. Ensuring precise screw insertion is crucial for maintaining clinical stability in patients over an extended period. In the open with freehand group, direct visualization of the pedicle was possible with fewer breaches; however, there was proximal facet joint damage. On the other hand, in the C-ram guided MIS-TLIF group, joint surface damage was reduced; however, there were challenges with positioning in the pedicle. Therefore, screw fixation using a robot, which combines the advantages of both platforms, may be the most optimal insertion technique.
Notes
Conflict of Interest
The CUVIS-spine robot mentioned in this paper is produced by CUREXO Inc. Seong Yi serves as a paid consultant for CUREXO Inc. Yoon Ha Hwang, Byeong-Jin Ha, Hyung Cheol Kim, Byung Ho Lee, Jeong-Yoon Park, and Dong-Kyu Chin state that they have no commercial or financial associations that might be perceived as a potential conflict of interest.
Funding/Support
This research received funding from CUREXO Inc., Republic of Korea (Grant/award number: 2017-31-1035, 2019-31-0831); Korea Institute for Robot Industry Advancement; and Domestic Medical Device Training Support Center, Ministry of Health and Welfare, and Korea Health Industry Development Institute.
Author Contribution
Conceptualization: SY; Formal Analysis: YHH; Investigation: YHH, BJH, HCK; Methodology: YHH; Project Administration: SY, JYP, DKC; Writing - Original Draft: YHH; Writing - Review & Editing: BHL, JYP, SY.