INTRODUCTION
Symptomatic cervical deformity (CD) can be a debilitating condition, with a health impact comparable to blindness, emphysema, renal failure, and stroke [1]. Although it is not as prevalent as thoracolumbar deformity (TLD), its incidence is increasing as the population ages, and the widespread use of cervical fusion surgeries is also producing more iatrogenic deformities. Junctional kyphosis from thoracolumbar spine surgery can also lead to CD. Successfully treating CD is often not straightforward, and many of the most important components of the process come before the surgery itself. The aim of this review is to provide a systematic approach for the evaluation, classification, and preoperative planning for CD that can aid in achieving optimal surgical results.
EVALUATION
1. Clinical Workup
As with any spine patient, a thorough history and physical exam should be obtained, with special attention paid to signs and symptoms of myelopathy (balance difficulties, poor hand dexterity, dropping objects, hyperreflexia, Hoffman’s sign, Romberg sign, clonus, motor weakness). The patient should be queried about activities of daily living, to determine if there is any trouble with pain, fatigue, eating or swallowing, or respiratory compromise. If previous spine surgeries have taken place, a thorough understanding of them is critical and prior operative reports should be obtained if possible. The patient should also be asked to stand upright with the head as erect as possible, so as to understand the global alignment and any reserve of extension that may exist.
A general medical evaluation should also be conducted. Heart disease, respiratory problems, diabetes, smoking, and previous infections can all increase the risk of surgical complications and even potentially preclude safe surgery. In scenarios where prior anterior cervical surgery has taken place, ENT (ear, nose, and throat) evaluation is also advisable to ensure protection of critical structures.
2. Radiographic Workup
Any patient with a suspected CD should undergo a battery of imaging studies that includes: static and dynamic plain films with lateral and anteroposterior (AP) views, full-length standing films (EOS [Canon, Tokyo, Japan] if available), a computed tomography (CT) scan with the appropriate reconstructions, and magnetic resonance imaging (MRI)[2]. Static plain films reveal the morphology and severity of the deformity, and also show any pre-existing instrumentation. Dynamic films, such as flexion-extension and lateral-bending views, provide information about the flexibility or rigidity of the deformity. This is critical when formulating operative plans. Full-length standing radiographs are often important, because a patient’s CD can be concomitant or linked to a TLD. Ames et al. [3] have reported that pelvic incidence (PI) is correlated with lumbar lordosis (LL), LL is correlated with thoracic kyphosis, and thoracic kyphosis is correlated with cervical lordosis (CL). Treating CD without accounting for TLD can exacerbate global spinal alignment and necessitate future reoperations. As a case in point, Jalai et al. [4] found that patients who underwent surgery to correct TLD developed worse cervical alignment parameters than their counterparts who did not undergo surgery.
A computed tomography scan with sagittal and coronal reconstructions can be invaluable because it reveals where the spine may be fused and provides anatomical details for instrumentation planning. And finally, an MRI is helpful to understand where the locations and severity of any cord compression or nerve root compression [2].
3. Radiographic Parameters
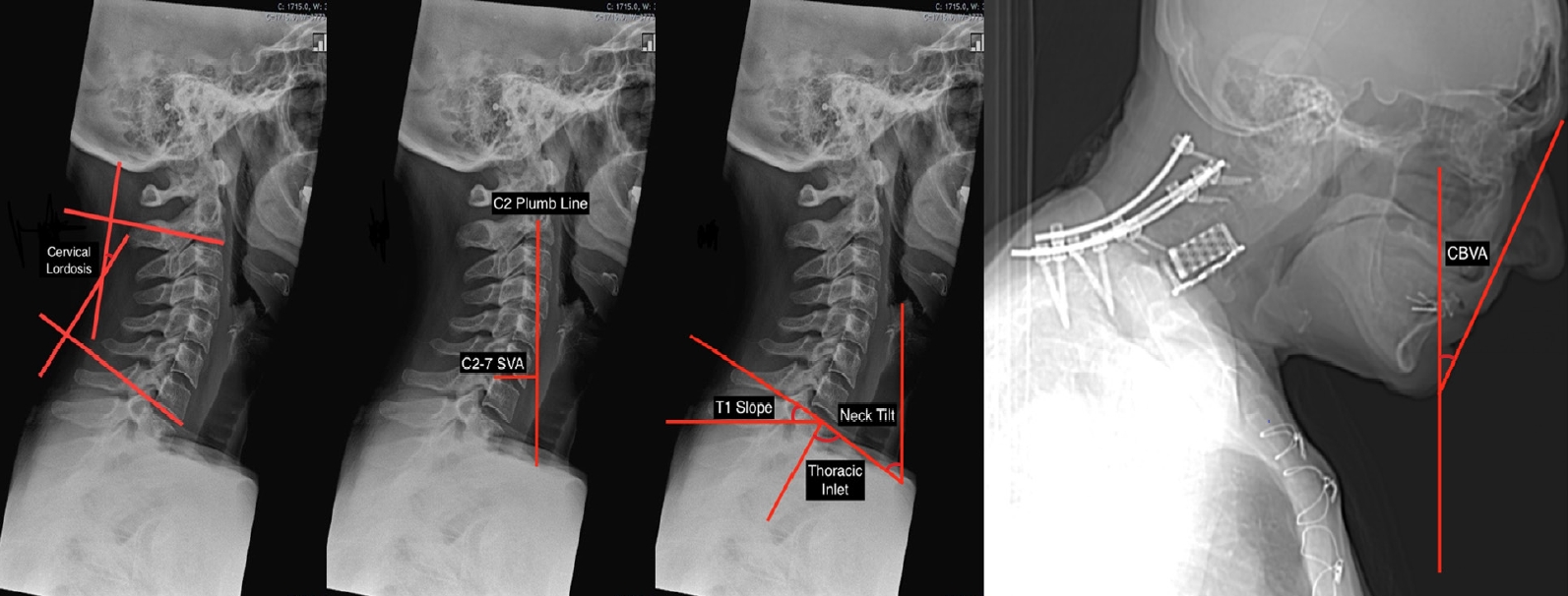
When assessing a CD, the following parameters should be measured using lateral radiographs in the neutral and extension position: CL, C2–7 sagittal vertical axis (C2–7 SVA), chinbrow vertical angle (CBVA), T1 slope (T1S), thoracic inlet angle (TIA), and neck tilt (Fig. 1).
There are at least 4 established methods for measuring CL: the modified Cobb method (mCM), the Ishihara index, Harrison’s posterior tangent method, and Jackson physiological stress lines [3,5]. The mCM is the most common approach, wherein 2 lines are drawn along the C2 and C7 inferior endplates, after which 2 additional lines perpendicular to the original ones are drawn. The angle subtended by the perpendicular lines is the CL. In actuality, modern software will compute the angle directly based on the lines parallel to the endplates. It is conventional to use negative angles for lordosis and positive angles for kyphosis. Of note, one modification is to use the line connecting the anterior and posterior tubercles of C1 as the upper reference line. This can be helpful, because the average total CL in asymptomatic adults is thought to be approximately -40°, with nearly 80% of the lordosis coming from C1–2 [6]. Only 15% of the total CL, or roughly 6°, can be found at C4–7. Men and women do not differ in their total CL, but older adults are known to have increased CL as a compensatory mechanism for increasing thoracic kyphosis and decreasing LL [6,7].
The C2–7 SVA is a widely used measure of regional sagittal alignment, and has been associated with health-related quality of life (HRQoL) metrics [8]. It is computed by measuring the distance between a plumb line dropped from the centroid of C2 (the odontoid) and a vertical line drawn from the posterior edge of the superior endplate of C7. One of the few studies to measure the C2–7 SVA in asymptomatic adults, using upright EOS films, reported an average value of 21.3 mm [9]. Theoretically, values significantly greater than that should be a source of disability because energy must be expended on paraspinal and neck muscles to maintain horizontal gaze. However, high-level evidence of a relationship between disability and increased C2–7 SVA on neutral lateral radiographs has yet to emerge.
An indirect measure of horizontal gaze is the CBVA, which is obtained by computing the angle subtended by a vertical line and a line drawn from the chin to the eyebrow. CBVA can be measured using either upright EOS films that include the skull or using clinical photographs. The patient must be standing with the hips and knees in extension and the cervical spine in a neutral or fixed position. Of note, CBVA can be estimated using either the slope of the line of sight (anterior/inferior margin of the orbit to the external auditory meatus) or the slope of McGregor’s line (posterior edge of the hard palate to the most caudal point of the occipital curve) [10]. By convention, the CBVA is considered positive when the head is facing down and negative when the head is facing up. There are varying reports as to normative values of the CBVA - one study suggests a value of -1.7° based on a cohort of asymptomatic adults [9], while another study reported that values between -4.7° and 17.7° are associated with minimal disability [10]. Many experts caution against overcorrection of cervical kyphosis, because a slightly positive CBVA (flexion) is necessary for many activities of daily living such as walking and using the bathroom. Suk et al. [11] have shown that achieving a postoperative CBVA < -10° can prevent the appropriate gaze when walking down stairs. The CBVA has gained traction in recent years as a guiding metric for the surgical correction of CD, and a general postoperative goal of 10° has been reported [5].
The final 3 parameters—TIA, T1S, and neck tilt—are interrelated. TIA is the angle subtended by the line emanating from the center of the T1 endplate and perpendicular to the T1 endplate and a line connecting the center of the T1 endplate and the upper end of the sternum. It can be measured using lateral radiographs, computed tomography, or MRI. T1S is the angle subtended by a horizontal line and a line parallel to the T1 upper endplate, and neck tilt is the angle subtended by a vertical line and a line connecting the sternum to the center of the T1 upper endplate. These 3 parameters are analogous to pelvic parameters - neck tilt is akin to pelvic tilt (PT), T1S is akin to sacral slope (SS), and TIA is akin to PI. In fact, TIA=T1S+neck tilt, just as PI=SS+PT [3].
T1S is emerging as a uniquely important parameter for CD because it can help predict the ideal CL for a patient, just as PI can be used to determine the appropriate LL. Specifically, Staub et al. [12] have reported that CL=T1S–16.5°± 2°.
CLASSIFICATION
1. Simmons
Before reviewing the formal classification schemes that have emerged in recent years, it is important to touch on the efforts of Simmons et al. [13] who were the first to describe a large series of extension osteotomies for CD. They reported results for 131 cases over 36 years, and their indications for surgery offer early insight regarding the stratification of patients. The surgeons operated on the subset of patients that had a severe flexion deformity causing either an impaired visual field, difficulty with personal hygiene and function, or poor swallowing. They also classified patients as either having a flexion deformity secondary to long-standing ankylosing spondylitis, or as having a flexion deformity due to trauma. Furthermore, the group distinguished between CD patients who had fractures of the cervical spine and those that did not, and also used flexion-extension films to identify subset of patients with C1–2 subluxation. Although these patterns do not have the makings of a comprehensive classification scheme, they were one of the first attempts to characterize operative CD patients.
2. Ames-International Spine Study Group (ISSG)
One attempt at defining a common language for CD was premised on a modified Delphi method, wherein a panel of experienced cervical spine surgeons relied on available literature and their expert opinion [14]. This classification system begins with 5 “descriptors” that provide a broad stratification of deformity types. The first 3 descriptors correspond to primarily sagittal deformities, with the apex of the deformity being the distinguishing feature: type “C” if the apex is in the cervical spine, type “CT” if it is at the cervicothoracic junction, and type T if it is in the thoracic spine. The fourth descriptor is for primarily coronal deformities which have a C2–7 coronal Cobb angle greater than 15° (type “S”), and the fifth one is for primarily craniovertebral junction abnormalities (type “CVJ”).
Once a CD is assigned one of these broad descriptors, 5 modifiers are then applied with the goal of incorporating sagittal, regional, and global spino-pelvic alignment features as well as myelopathy. The aim of the first modifier is to describe translation of the cervical spine in the sagittal plane, and the C2–7 SVA was chosen for this because it is associated with patient-reported outcome measures (PROMs) such as the Short Form 36 physical component score, the Neck Disability Index (NDI), and the modified Japanese Orthopaedic Association (mJOA) score for myelopathy [8,15]. Tang’s analysis suggested that a C2–7 SVA greater than 4 cm is associated with worse NDI scores, and so the modifier is scored accordingly. A C2–7 SVA< 4 cm is given a “0,” a measurement of 4–8 cm is given a “1,” and a measurement > 8 cm is given a “2.”
The second modifier is the CBVA, given the importance of horizontal gaze. If the CBVA is 1°–10° the score is “0,” if it is -10° to 0° or 11°–25° the score is “1,” and if the CBVA is < -10° or > 25° the score is “2.” The third modifier is the T1S–CL, which is a measure of regional alignment and is analogous to the mismatch between PI and LL that is central to TLD. In the absence of robust PROM-related data, expert opinion was used to define the following: a score of “0” for T1S–CL< 15°, a score of “1” for a T1S–CL of 15°–20°, and a score of “3” for T1S–CL> 20°.
The fourth modifier is based on myelopathy, because severe cervical kyphosis can cause axonal stretch, spinal cord injury, and ischemia. The mJOA is utilized here because it is a widely accepted scale for quantifying cervical spondylotic myelopathy (CSM), and because it has been correlated with cervical sagittal balance [3,15]. Borrowing from studies of decompressive surgeries for CSM [16], the modifier was defined as follows: a score of “0” if mJOA is 18 (no myelopathy); a score of “1” if mJOA is 15–17 (mild myelopathy); a score of “2” if mJOA is 12–14 (moderate myelopathy); and a score of “3” for mJOA < 12. The fifth and final modifier is the SRS (Scoliosis Research Society)-Schwab classification of for TLD, which has been validated and linked to HRQoL measures [17]. This modifier is based on the understanding that cervical and thoracolumbar deformities can be interdependent and concomitant, and that a global perspective on spinal alignment is always prudent. Fig. 2 summarizes the classification system.
The intra- and interobserver reliabilities of this classification suggested moderate agreement, and so it is a useful starting point for categorizing CD. However, one weakness is that certain components were introduced a priori without definitively proving that they are responsible for the health impact of symptomatic CD. This has been corroborated by Bakouny et al. [18] who found that 2 of the modifiers—CBVA and T1S–CL—are not specific to patients with CD and can also be seen in asymptomatic adults.
3. Kim-ISSG
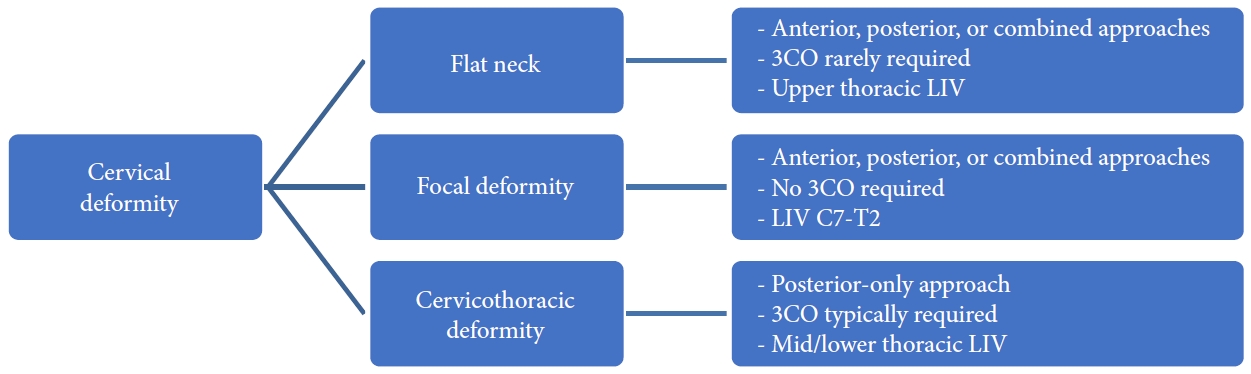
A second classification system for CD has been developed more recently based on lessons learned from the previous attempt [19]. A major premise behind this system is that distinct morphologies of sagittal alignment can be uncovered by incorporating dynamic radiographs, which provide information on a patient’s ability to correct malalignment. The development process began by collecting radiographic parameters in the neutral and extended position for patients with severe sagittal CD and then determining which of them correlated with PROMs. C2–7 flexibility, T1S–CL flexibility, and maximum kyphotic flexibility correlated with neck pain, and maximum focal kyphosis correlated with mJOA. These parameters then formed the basis of a cluster analysis which revealed 3 distinct subgroups of sagittal CD: “flat neck” (FN), “focal deformity” (FD), and “cervicothoracic deformity” (C-T), with a fourth type being a coronal plane deformity (cervical scoliosis). Fig. 3 summarizes this classification system.
Classification of patients into one of these groups hinges mostly on 3 parameters: lateral neutral cervical SVA, the change in T1S–CL between the lateral neutral and lateral extension film, and the neutral T1S. Patients in the FN group (type I) tend to have a large T1S–CL mismatch due to insufficient CL, but it partly corrects with extension. Those in the FD group (type II) have a significant localized kyphosis across 2 adjacent vertebrae, however, the overall cervical SVA is within normal limits and the T1S is generally small. Finally, patients in the CT group (type III) have a T1S–CL mismatch not because of insufficient CL, but because the T1S is very large. They attempt to compensate with hyperlordosis of the cervical spine but it is not adequate.
A feature of this classification system is that it can help identify the main driver of a patient’s CD. Most patients in the FN group have a cervicothoracic driver and most in the FD group have a cervical driver, whereas for the CT group there is usually a thoracic or thoracolumbar driver of the deformity. It must be re-emphasized, however, that a prerequisite for making these determinations is the availability of flexion/extension radiographs, making this classification system unique when compared to previously published classifications.
Another insight from the approach of Kim et al. [19] is that cervical sagittal alignment parameters, as measured on neutral radiographs, do not appear to correlate with HRQoL measures. This is consistent with the aforementioned work by Bakouny et al. [18] who showed that the C2–7 SVA and CBVA modifiers from the Ames-ISSG classification are seen in asymptomatic adults. One study has shown an association between neutral sagittal alignment and HRQoL, but it was only for postoperative patients [8]. It is clear that extension films are critical for the classification and appraisal of CD, because they help reveal a patient’s ability to restore sagittal alignment and allow for radiographic parameters that are linked to PROMs.
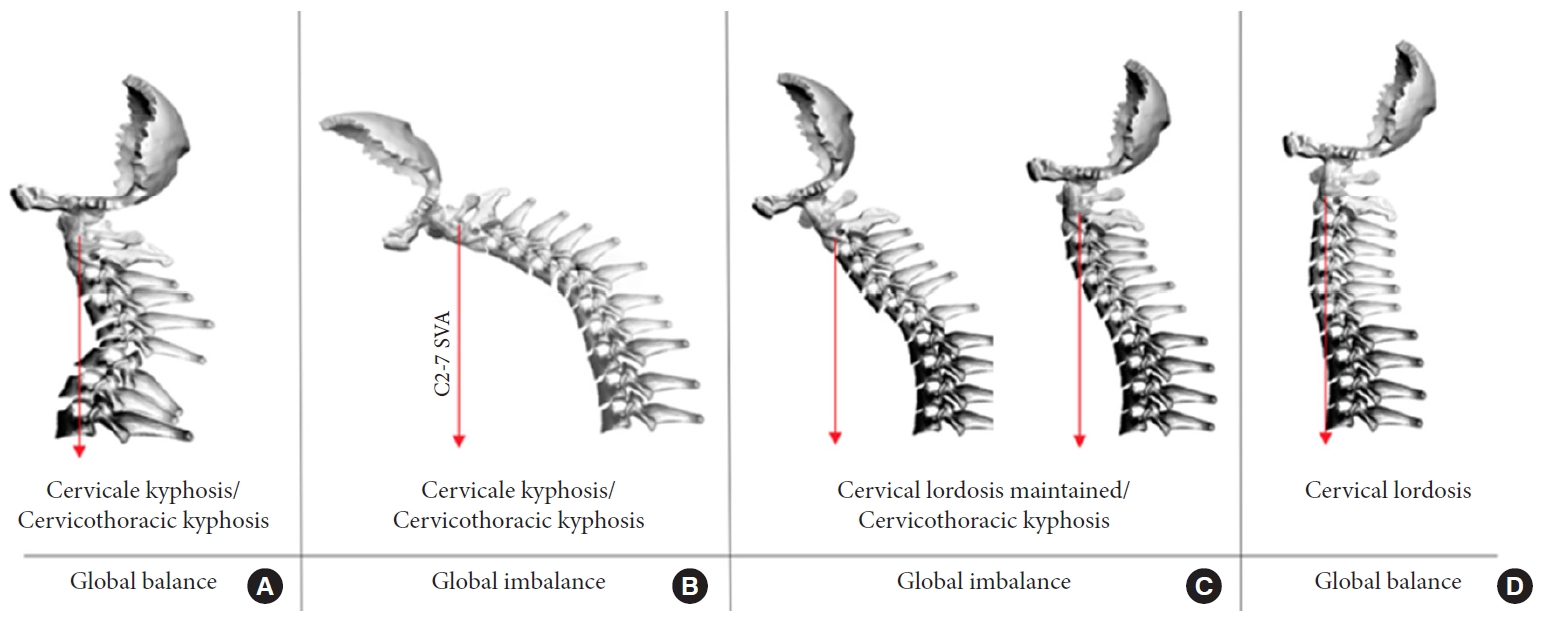
4. CSRS-Europe
A third classification system for CD was developed in 2019 by the Cervical Spine Research Society (CSRS)-Europe [20]. It does not rely on a combination of radiographic parameters that are linked to HRQoL measures, but it does provide a useful and efficient short-hand for surgeons. Type A refers to patients that have cervical or cervicothoracic kyphosis but have reasonable regional and global balance as measured by the C2–7 SVA and the C2–S1 SVA. Type B is for patients that have cervical or cervicothoracic kyphosis leading to a large C2–7 SVA and a large C2–S1 SVA, signifying regional and global imbalance. Type C is for patients with cervicothoracic kyphosis who can partly compensate with lordosis, but who nevertheless have a global imbalance. And finally, type D is for patients who have enough CL to achieve global balance. Fig. 4 summarizes this classification system.
The classification of CD patients based on this scheme is not simply a descriptive exercise, because there are practical implications for preoperative cervical alignment, myelopathy, osteoporosis, and surgical techniques [20]. Koller et al. [20] found that baseline C2–7 SVA increases significantly as one transitions from type A, to type B, to type C. The prevalence of CSM also varied significantly based on the subtype: 66% for type A, 36% for type B, and 12% for type C. Moreover, the incidence of osteoporotic vertebrae varied significantly: 22% for type A, 44% for type B, and 65% for type C. A posterior surgical approach was significantly more favored with type A (94%), whereas it was utilized 56% of the time for type B and only 28% of the time for type A. The proportion of patients receiving shorter fusion constructs (instrumentation not extending below T2) was 22% for type A, 44% for type B, and 65% for type C. And not surprisingly, the mean osteotomy grade as defined by the Ames classification [21] was 3.6 for type A, 4.1 for type B, and 5.5 for type C. Based on these operative differences, the change in C2–7 SVA from baseline to postoperative follow-up also increases as one moves from type A to type B to type C.
SURGICAL PLANNING
The successful correction of CD starts with a clear understanding of the surgical goals. In general, these goals include: deformity correction with restoration of horizontal gaze, decompression of neural elements where necessary, and spinal stabilization with arthrodesis [22]. To determine the amount and location of deformity correction, the aforementioned radiographic parameters (CL, C2–7 SVA, CBVA, T1S) should be carefully studied so that the amount of postoperative targets can be determined. As a rule of thumb, surgeons can aim to have the postoperative C2–7 SVA < 4 cm, the CBVA between 0° and 10°, and the CL equal to the TS - 16.5°. The notable point here is that for patients with a T1S less than 16.5°, the ideal alignment goal may be a non-lordotic or even a kyphotic cervical spine.
Tan et al. [22] described a comprehensive list of the crucial factors for planning CD corrections. They are: (1) neural compression and any associated symptoms; (2) flexibility of the deformity; (3) presence of anterior or posterior ankylosis; (4) location of the deformity; (5) prior surgery; (6) degenerative changes at the proximal/distal levels; and (7) general medical status. Neural compression is best seen on MRI. As a general rule, if there is ventral cord compression then an anterior approach will need to be incorporated into the surgical plan in order to achieve adequate decompression [23]. An exception to this might be an aggressive 3-column posterior osteotomy that accomplishes indirect ventral decompression through correction of a kyphotic deformity.
As mentioned previously, dynamic films provide information about the flexibility of a CD. If a patient can self-correct cervical kyphosis with extension, a posterior approach can be sufficient. If there is insufficient reserve of extension, however, an AP approach is often necessary to restore adequate lordosis. The computed tomography scan provides additional information in this regard, as it can reveal anterior or posterior ankylosis, prior areas of solid arthrodesis versus pseudoarthrosis, prior laminectomy defects, positioning of instrumentation, as well as a sense of the flexibility of segments because the studies are done with patients in the supine position. It is important to note, however that deformed patients may have their heads supported by rolls of pillows which would make a flexibility assessment unreliable. In these situations, the scout images can provide an idea of how the patient was positioned for the study. Regardless of the particular position, if anterior ankylosis is seen on the computed tomography but the facet joints are not fused, then anterior correction alone can be sufficient. If the facets are also fused, then the patient may require a posterior osteotomy, followed by anterior correction (with or without an anterior osteotomy), and then posterior instrumentation [22].
The Kim-ISSG classification also provides practical insights for treatment planning by taking advantage of information from dynamic films. Patients in the flat neck group are more likely to have a deformity that is passively correctable, obviating the need for a 3-column osteotomy in the surgical plan. However, the spinal mobility does often warrant longer constructs than one might otherwise plan for, especially for those with an elevated T1S [19]. Most patients in this flat neck group will require a combined AP surgical approach. Patients in the FD group, on the other hand, rarely have significant global imbalance and so localized corrections can be effective and fusion levels can stay within the cervical spine without crossing the cervicothoracic junction. FD patients also often require a combined AP surgical approach. Finally, patients in the cervicothoracic deformity group generally have a longer-segment deformity relating to a high T1S. The high T1S can be due to upper thoracic kyphosis or proximal junctional kyphosis in the lower thoracic spine, such that correcting the deformity in those locations can reduce the T1S and normalize cervical alignment. In these scenarios, the correction actually decreases the T1S, such that less CL is required to achieve horizontal gaze. The correction of cervicothoracic deformities will often require a 3-column osteotomy, a posterior-only approach, and fusion constructs that extend into the lower thoracic spine (Fig. 5).
These nuances emphasize the fact that the location of the deformity apex is critical, because this is where the corrective maneuvers will be focused. An anterior corpectomy can often be used to correct focal kyphosis in the cervical spine, whereas a C7 or T1 pedicle subtraction osteotomy (PSO) is usually necessary to correct severe focal deformities at the cervicothoracic junction. We do not recommend the use of a posterior-only 3-column osteotomy in cephalad segments where the vertebral artery is located within the foramen transversarium. Some surgeons favor performing 3-column osteotomies for correction of CD at more distal levels (T2 or T3) when feasible in order to reduce the risk of compromising the C8 and T1 nerve roots.
It is also worth mentioning that, in the setting of prior anterior surgery, the contralateral side can be used to avoid scar tissue, although ENT evaluation should first be obtained to ensure full vocal cord function. Many patients with CD may also have pre-existing dysphagia [24]. In these cases, an anterior approach may worsen the dysphagia and so an ENT evaluation accompanied by a barium swallow or esophagram may be necessary prior to surgery.
CASE EXAMPLES
1. Case 1
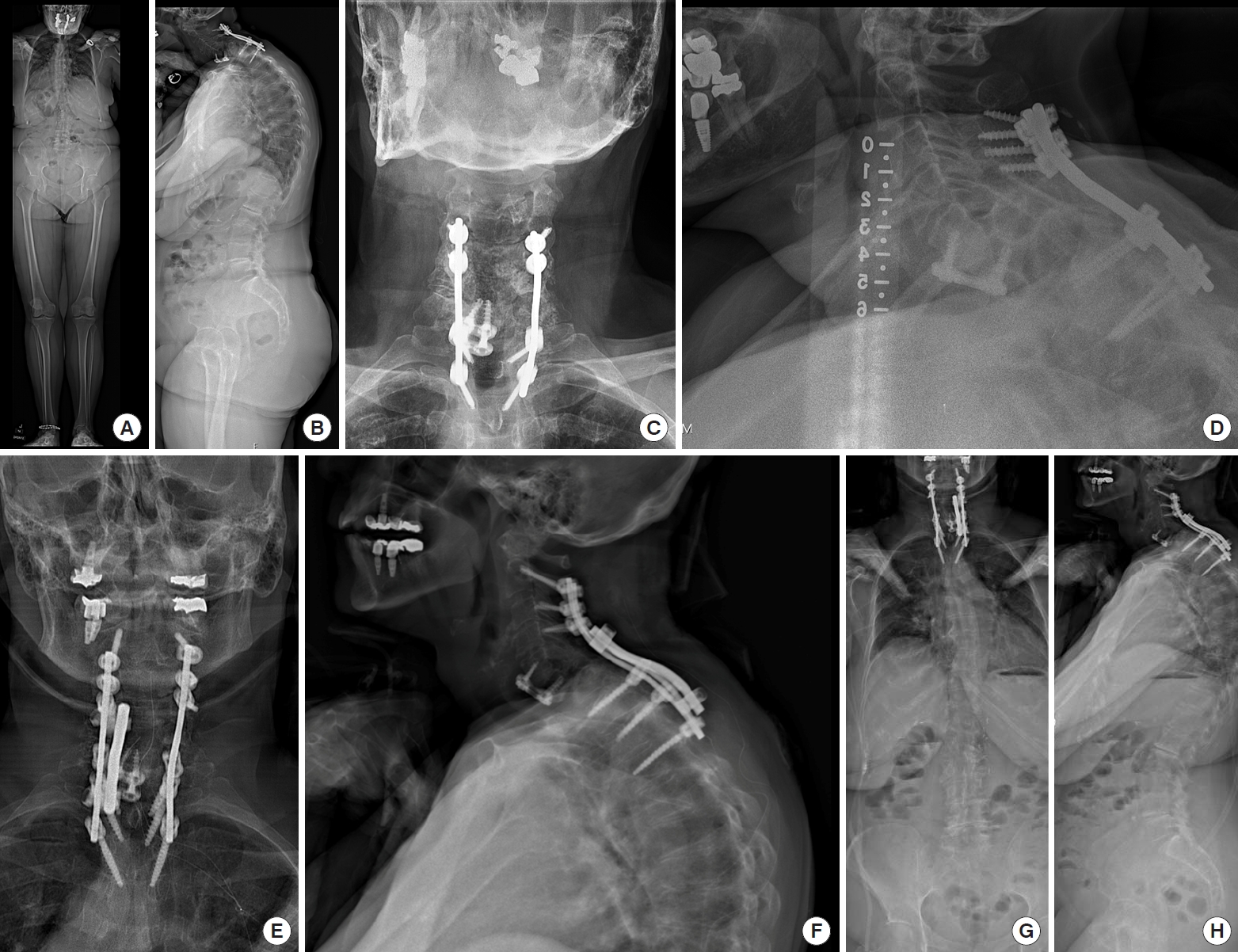
To synthesize the information presented here, we present 2 cases of symptomatic CD. The first patient is 71 years old and relatively frail, with chronic kidney disease (on dialysis). She previously underwent a C2–T1 posterior fusion, but is now having difficulty holding her head straight. She has also developed difficulties with swallowing and pain in the upper back and neck. Radiographic assessment reveals a CL of 16° and a T1S of 62°, representing a mismatch far beyond the normative gap of 16.5°. Per the Ames-ISSG classification, she would have the descriptor “C-T” and the following modifiers: 2, 2, 2, 1, N+++. Per the CSRS-Europe classification, she would be a type C, given that her cervicothoracic kyphosis can likely be partly corrected with hyperextension, but still leads to global imbalance. Per the KimISSG classification, she would be in the “C-T” group (type III) given her cervicothoracic deformity, suggesting the need for a 3-column osteotomy and instrumentation that extends into the mid/lower thoracic spine. The patient did opt for surgery to correct her CD and underwent a revision C2–T9 posterior fusion with a PSO at T2. See Fig. 6 for pre- and postoperative films.
2. Case 2
This patient is a 70-year-old female with an acquired solitary kidney (living donor nephrectomy), esophagitis, and dysphagia who previously underwent a C5–7 anterior cervical discectomy and fusion at an outside hospital for facet fractures after a motor vehicle accident. One month later there was progressive spondylolisthesis of C6, necessitating a posterior cervical fusion from C4–T2 and a C5–7 laminectomy. Since that time she has developed progressive and persistent neck pain, bilateral arm pain, problems with balance, and difficulty holding her head up. Radiographs demonstrate lateral mass screw pull-out and a grade 4 spondylolisthesis at C6–7. Her exam is notable for 4/5 strength in the left triceps, hyperreflexia, poor tandem gait, and a positive Hoffman’s sign. Radiographic assessment reveals a CL of 17° and a T1S of 38°, representing a mismatch of 21°. The C2–7 SVA is 6.8 cm. Per the Ames-ISSG classification, she is a type C with the following modifiers: 1, 2, 2, 2, N/0/0/0. Per the CSRSEurope classification she is a type B, given that she has cervical kyphosis that creates a large C2–7 SVA and regional imbalance. Per the Kim-ISSG classification, she is a type I, given the inability of the CL to fully compensate for the given T1S (on standing and extension x-rays) due to the C6–7 spondylolisthesis. The patient underwent a revision C2-T3 posterior fusion and revision decompression with duralysis at C5–T1, and the deformity was corrected with bivector traction and posterior column osteotomies only. The postoperative CL improved to 26° and the C2–7 SVA improved to 3.7cm. See Fig. 7 for pre- and postoperative films.
CONCLUSION
The evaluation and classification of CD is a complex and evolving endeavor. Careful study of radiographic parameters is critical, but recent work has highlighted the importance of understanding them with dynamic films and not simply static ones. The reserve of extension that a patient possesses is a key variable when formulating a treatment plan. The classification systems presented here not only aid in the care of individual patients, but also generate questions regarding the optimal plan for correcting specific subtypes of CD. More study is needed to arrive at best practices for the treatment of CD that are patient-specific and reduce the risk of postoperative complications and reoperations.