 |
 |
- Search
|
|
||
Abstract
C1 stenosis is often an easily missed cause for cervical myelopathy. The vast majority of cervical myelopathy occurs in the subaxial cervical spine. The cervical canal is generally largest at C1/2, explaining the relatively rare incidence of neurological deficits in patients with odontoid fractures. However, some subjects have anatomical anomalies of the atlas, which may cause stenosis and result in clinical symptoms similar to subaxial cord compression. Isolated pure atlas hypoplasia leading to stenosis is quite rare and may be associated with other anomalies, such as atlas clefts or transverse ligament calcification. It may also be more commonly associated with syndromic conditions such as Down or Turner syndrome. Although the diagnosis can be easily made with a cervical magnetic resonance imaging, the C3/2 spinolaminar test using a lateral cervical plain radiograph is a useful and sensitive tool for screening. Surgical treatment with a C1 laminectomy is generally necessary and any atlantoaxial or occipito-atlanto instability must be treated with spinal stabilization and fusion.
Degenerative cervical spondylosis is the most common cause of myelopathy in elderly patients, and most frequently as a result from stenosis and spinal cord compression below C2 [1]. Although cervical stenosis has been extensively studied in the subaxial spine, fewer studies evaluated the role and prevalence of stenosis at the level of the atlas, also known as C1 stenosis. The spinal canal is generally large at the atlantoaxial region, which partially explains the low rate of neurological deficits at presentation in patients with odontoid fractures [2,3]. Congenital and acquired (due to transverse ossification or facet osteophytes, for instance) C1 stenosis is an unusual entity that may cause cervical myelopathy with a clinical presentation similar to the subaxial cervical compression [3]. The literature concerning C1 stenosis is generally based on anecdotal cases or small series [4,5]. For this reason, we performed a literature review of the general considerations, diagnosis and management of C1 stenosis.
The atlas is named after the titan in Greek mythology that supports the globe on his back, with the analogy that the atlas supports the weight of the cranium [6]. It is a unique vertebra: it has no clear spinous process, and is composed of 2 lateral masses that are linked together by the anterior and posterior arches forming the C1 ring [6]. The lateral masses support the weight transmitted from the head into the spine and then transmit it on to the superior C2 facet joints.
The anterior tubercle is the attachment of the longus colli muscles and anterior longitudinal ligament and it is located in the midline of the anterior arch [6]. In the posterior portion of the anterior arch, there is a concave area that articulates with the odontoid process of C2 that it is contained by the transverse ligament, preventing anterior translation of the atlas above the axis when flexing the neck. The posterior arch comprises the posterior portion of the spinal canal where one can find the attachment of the posterior atlantooccipital membrane [6]. The vertebral arteries ascend through the transverse foramina located in the lateral portion of the lateral masses and then directs medially just above the posterior arch of C1 to enter the dura mater [6,7].
Biomechanically, the atlantoaxial joint is responsible for about 50% of the cervical rotation due to its unique anatomy and 2 synovial joints; the atlanto-occipital joints are responsible for most of the flexion/extension of the neck (nodding the head) [8,9].
During embryological development, the atlas has 3 ossification centers – one anteriorly that forms the anterior arch and tubercle, along with 2 posteriorly (1 for each lateral mass) that eventually fuse together to form the posterior arch [6]. The median age for complete atlas ossification is about 8.5 years (ranging from 5.5 to 1.3 years) [10]. It should be noted that some patients may not have ossification of the anterior and/or posterior arch. Gehweiler et al. [11] reported some cases of congenital atlas malformations that are part of the differential diagnosis of an atlas fracture – total or partial aplasia of the posterior arch, hemi aplasia, clefts or remnant midline rachischisis, among others. A careful evaluation is necessary to differentiate these small congenital variations from spinal trauma.
We performed a literature search in the PubMed Database for “Congenital C1 Stenosis” and also cross-referenced articles. We included age of presentation, radiological findings and treatment performed in Table 1 [12-19]. Of note, the largest case series we found was recently published by Nehete et al. [12], in 2018, with 20 patients. There were 14 men and 6 women in their series, with age ranging from 4 to 60 years (mean, 22.85) and 12 patients with less than 18-year-olds. Two patients required surgery on an emergent basis due to acute neurological worsening and respiratory distress. Four patients had associated syndromes: 2 pediatric patients and 1 adult with type 4 Morquio disease and 1 additional adult with ankylosing spondylitis. Surgical treatment consisted in isolated C1 arch resection in 10 cases, concomitant C1 and C2 or foramen magnum decompression in 5 cases and concomitant fusion+C1 laminectomy in 1 patient (C2–3). Four patients did not have the treatment performed described.
The dimensions of the subaxial cervical spinal canal have been studied extensively due to the higher prevalence of cervical myelopathy at these levels. For instance, Lee et al. [2] evaluated the distance from the posterior most aspect of the vertebral body to the anterior most aspect of the spinolaminar structure from C3 to C7 in 469 adult skeletal specimens of the cervical spine, adopting the threshold of a canal diameter of <12 mm as stenotic. They reported that the average anteriorposterior canal diameter (and standard deviation) was 14.1±1.6 mm, ranging from 9 to 20.9 mm. Men had larger spinal canals than women and subjects who were older than sixty had narrower spinal canals compared with younger subjects. After adjusting for increased body size, they estimated that the prevalence of bony cervical spinal stenosis was 4.9% in adults, 6.8% in those older than 50 years and 9% in those older than 70.
Specifically evaluating the upper cervical spine canal, Steel [20] had proposed the “Rule of Thirds” in 1968, where the spinal cord, the dens, and the space with cerebrospinal fluid occupy one-third each of the canal diameter at the level of the atlas. More recently, Kelly et al. [3] performed a cadaveric study with 543 cervical spine specimens. They measured with digital calipers the sagittal and coronal diameters of the atlas, axis, and C3. The C1 sagittal diameter ranged from 23.5 to 38.1 mm, with a mean of 30.8±2.4 mm. Adopting C1 hypoplasia as a sagittal diameter value representing the lower 2.5% subjects’ measurements, the criteria for defining hypoplasia initially was ≤26.1 mm. After correction for size and magnification of radiographs, hypoplasia of the atlas was defined when the inner sagittal diameter of the atlas was less than 28.9 mm. Interestingly, the authors reexamined the Steel Rule of Thirds and reported that 10% of cases had the dens occupying more than 40% of the spinal canal at C1, in disagreement with the Steel’s Rule of Thirds. This study is important because it defines a specific and objective criterion for atlas stenosis in adults.
Isolated pure atlas hypoplasia is rare, while fissures and segmentation defects are relatively common. A posterior midline cleft is the most frequently encountered abnormality (observed in about 3%–4% in 1,440 cervical radiographs), with much higher prevalence than anterior arch defects (estimating of less than 1%) [4,21]. Of note, arch anomalies of the atlas rarely cause compression since they usually increase the canal space (acting as a natural laminectomy in some cases). Another associated anomaly that may be associated with hypoplasia and canal stenosis is an ossified transverse ligament leading to chronic and progressive myelopathy [18]. Some syndromic diseases that may be associated with hypoplasia of the atlas include: Down Syndrome, Turner Syndrome, Morquio disease, ankylosing spondylitis, achondroplasia, gonadal dysgenesis, and spondyloepiphyseal dysplasia congenital [5,12]. Achondroplasia, on the other hand, is more commonly associated with foramen magnum stenosis. Of note, the reason that a congenital malformation presents with clinical symptoms relatively later in life (as opposed to at birth or in children) is that symptoms generally require superimposed age-related degenerative changes, such as ligamentum flavum buckling, pannus behind the dens or synovial cyst formation. Because of this reason, depending upon the degree of congenital stenosis, the age of presentation of C1 stenosis may vary from small children to older adults.
The clinical presentation of C1 stenosis is similar to subaxial cervical myelopathy: long-tract signs (sensorial and motor dysfunction), gait difficult, impaired sphincter function, and balance problems. Although the diagnosis can be made easily with a magnetic resonance imaging (MRI) visualizing cervical spinal cord compression at the atlas level, Oshima et al. [22] proposed a screening test for the hypoplasia of C1 on lateral radiographs based on test the sensitivity of the C3–C2 spinolaminar line (also called as the spinolaminar line test, as shown in Fig. 1). In their study to evaluate the test, the space available for the cord (SAC) of the atlas was measured on neutral lateral radiographs using a correction for magnification of 10%. The C1 SAC was measured from the posterior border of the dens to the ventral lamina of C1. The spinolaminar line from C3 to C2 was drawn, extending cranially. If the ventral lamina of the atlas lay anterior to the line, the test was considered positive. An MRI was then performed to check for spinal cord compression and computed tomography (CT) scan for evaluating the sagittal diameter of the spinal canal. Among the 487 patients included (they included patients with cervical spine complaints but excluded those with rheumatoid arthritis, congenital deformities, trauma, infections, and prior surgeries), 21 (4.3%) had a C1 ring ventral to the C3–C2 spinolaminar line (positive test). All of them had a C1 SAC of less than 19.4 mm. Of note, the kappa coefficient for different observers applying the test was excellent (a high rate of agreement). Eight patients had the C1 SAC smaller than C2 SAC, all with a positive test. The sensitivity of the spinolaminar test to detect a small C1 SAC was 100%, with a specificity of 80% and positive and negative predictive values of 385 and 100%, respectively. Of the 8 patients with smaller C1, 4 had effacement of the subarachnoid space and 2 had spinal cord compression at C1. No patients with a C1 larger than C2 SAC had evidence of spinal cord compression. Authors concluded that the spinolaminar test is a simple and accurate screening test for C1 stenosis using lateral plain radiographs.
An illustrative case of cervical myelopathy secondary to atlas stenosis is presented in Fig. 2.
The treatment of C1 stenosis consists of a posterior C1 laminectomy. However, while isolated C1 hypoplasia is rare, it is necessary to perform a thorough evaluation of the cervical spine and the craniovertebral junction. Evaluation for atlantoaxial instability is mandatory, as well as for occipito-atlantal instability, which is rare. Dynamic sagittal CT and MRI scans are useful to provide good visualization of the upper cervical spine joints, occult subluxation, and dynamic soft tissue compression. When atlantoaxial or craniocervical junction instability are found in association, concomitant fusion is necessary.
C1 stenosis is an uncommon cause of cervical myelopathy. It is important for clinicians to understand the unique embryology, anatomy, as well as potential malformations that is unique to the atlas. The spinolaminar line test is a useful screening tool based on simple plain radiographs, followed by a cervical MRI to confirm spinal cord compression. Surgical treatment via C1 laminectomy is recommended, and if there is segmental instability, spinal stabilization and fusion is necessary to achieve optimal outcome.
Fig. 1.
The C3–C2 spinolaminar line is drawn beginning at C3 and extending cranially up to C2 (yellow line). Then, this line is extended up to C1. In a normal cervical spine, the ventral aspect of the C1 lamina is posterior to this line (A). If the ventral lamina (yellow arrow) of C1 is anterior to this line, the test is defined as positive (B), meaning that there is a possibility of C1 stenosis.

Fig. 2.
Patient with cervical myelopathy secondary to atlas stenosis. (A) Three-dimensional computed tomography (CT) scan reconstruction with a bifid posterior arch of the atlas, (B) view from inside the cranial cavity down into the foramen magnum showing the posterior arch of the atlas inside the spinal canal. Axial CT scan (C) and axial T1 sequence magnetic resonance imaging (MRI) (D) of the atlas showing severe canal compression. (E) Sagittal T2 sequence MRI with spinal cord compression at the level of the atlas.
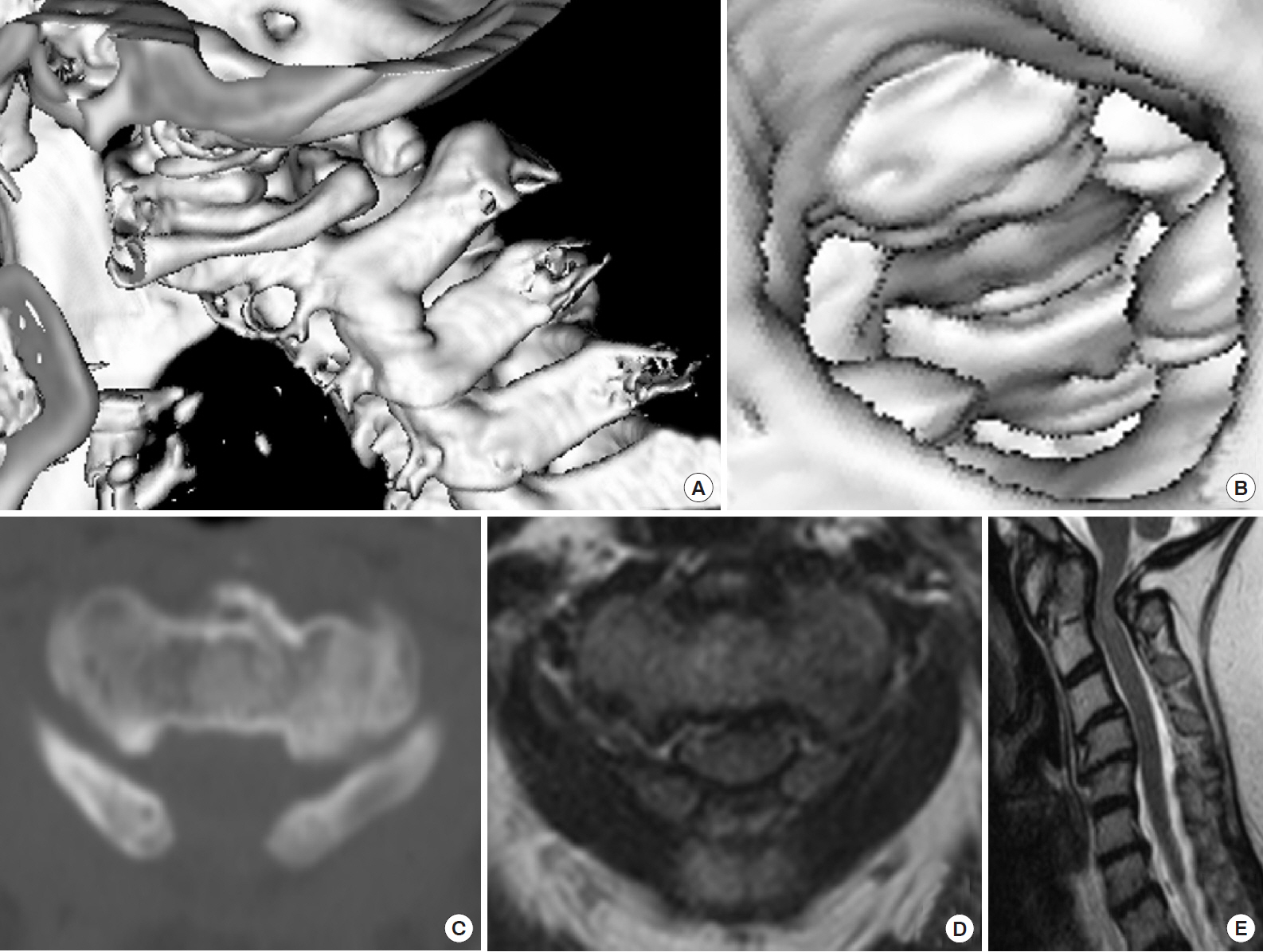
Table 1.
Summary of literature review on congenital C1 stenosis
| Study | Description | Treatment and outcome |
|---|---|---|
| Phan et al., [13] 1998 | Two male patients with progressive cervical myelopathy during the last months to years. No trauma history | Posterior decompression with removal of both posterior arch of C1 |
| Imaging – 2 hypoplastic but complete posterior C1 arch with severe stenosis (80- and 75-year-old man) | Both patients had improvement of their clinical symptoms during the follow-up | |
| Benitah et al., [14] 1994 | Two cases of upper cervical spinal cord compression – one stenosis due to acquired extensive unilateral osteophytes on the left C1–2 joint in a violinist and the other with congenital hypertrophy of the laminae of C1 and C2 (78-year-old man and 41-year-old woman) | Symptoms improved after laminectomy and ligamentum flavum resection of the atlas and axis for both patients |
| Nishikawa et al., [15] 2001 | Three cases of cervical myelopathy due to congenital stenosis from hypoplasia of the atlas (82-year-old man, 72-year-old man, and 42-year-old woman) | Laminectomy of C1 was performed in all 3 patients with clinical improvement |
| Atasoy et al., [16] 2002 | A 30-year-old man with a posterior arch hypoplasia in a bipartite atlas with an os odontoideum with cervical myelopathy. Marked atlantoaxial instability in dynamic cervical plain radiographs | Patient declined surgical treatment |
| Connor et al., [17] 2001 | An 8-year-old child with cervical myelopathy due to compression of the spinal cord by the medial posterior hemiarches of a bifid C1 leading to severe C1 stenosis | Laminectomy of the posterior arch of C1 was performed with clinical improvement |
| Bokhari and Baeesa, [18] 2012 | A 68-year-old woman with cervical myelopathy due to a hypoplastic intact posterior arch of atlas and concomitant ossified transverse ligament | Laminectomy of C1 with clinical improvement |
| Pascual-Gallego et al., [19] 2014 | A 5-year-old boy with Down syndrome and cervical myelopathy due to an anomaly of the atlas leading to stenosis | Laminectomy of C1 was performed with clinical improvement |
| Nehete et al.,[12] 2018 | The largest series of 20 patients with C1 arch stenosis. There were 12 pediatric patients (< 18 years old) and 8 adults – mean age was 22.85 years. Four had syndromic association | Laminectomy of C1 was the treatment of choice with improvement in symptoms |
REFERENCES
1. Joaquim AF, Ghizoni E, Tedeschi H, et al. Management of degenerative cervical myelopathy - an update. Rev Assoc Med Bras (1992) 2016 62:886-94.



2. Lee MJ, Cassinelli EH, Riew KD. Prevalence of cervical spine stenosis. Anatomic study in cadavers. J Bone Joint Surg Am 2007 89:376-80.


3. Kelly MP, Oshima Y, Yeom JS, et al. Defining hyoplasia of the atlas: a cadaveric study. Spine (Phila Pa 1976) 2014 39:E1243-7.



4. Currarino G, Rollins N, Diehl JT. Congenital defects of the posterior arch of the atlas: a report of seven cases including an affected mother and son. AJNR Am J Neuroradiol 1994 15:249-54.


5. Martich V, Ben-Ami T, Yousefzadeh DK, et al. Hypoplastic posterior arch of C-1 in children with Down syndrome: a double jeopardy. Radiology 1992 183:125-8.


6. Standring S. Gray’s anatomy: the anatomical basis of clinical practice. 41st ed. Edinburgh: Churchill Livingstone Elsevier; 2015.
7. Joaquim AF, Mudo ML, Tan LA, et al. Posterior subaxial cervical spine screw fixation: a review of techniques. Global Spine J 2018 8:751-60.



8. Dickman CA, Crawford NR, Brantley AG, et al. Biomechanical effects of transoral odontoidectomy. Neurosurgery 1995 36:1146-52.



9. Dickman CA, Locantro J, Fessler RG. The influence of transoral odontoid resection on stability of the craniovertebral junction. J Neurosurg 1992 77:525-30.


10. Piatt JH Jr, Grissom LE. Developmental anatomy of the atlas and axis in childhood by computed tomography. J Neurosurg Pediatr 2011 8:235-43.


11. Gehweiler JA Jr, Daffner RH, Roberts L Jr. Malformations of the atlas vertebra simulating the Jefferson fracture. AJR Am J Roentgenol 1983 140:1083-6.


12. Nehete LS, Bhat DI, Gopalakrishnan MS, et al. Unusual cause of high cervical myelopathy-C1 arch stenosis. J Craniovertebr Junction Spine 2018 9:37-43.



13. Phan N, Marras C, Midha R, et al. Cervical myelopathy caused by hypoplasia of the atlas: two case reports and review of the literature. Neurosurgery 1998 43:629-33.


14. Benitah S, Raftopoulos C, Balériaux D, et al. Upper cervical spinal cord compression due to bony stenosis of the spinal canal. Neuroradiology 1994 36:231-3.



15. Nishikawa K, Ludwig SC, Colón RJ, et al. Cervical myelopathy and congenital stenosis from hypoplasia of the atlas: report of three cases and literature review. Spine (Phila Pa 1976) 2001 26:E80-6.


16. Atasoy C, Fitoz S, Karan B, et al. A rare cause of cervical spinal stenosis: posterior arch hypoplasia in a bipartite atlas. Neuroradiology 2002 44:253-5.



17. Connor SE, Chandler C, Robinson S, et al. Congenital midline cleft of the posterior arch of atlas: a rare cause of symptomatic cervical canal stenosis. Eur Radiol 2001 11:1766-9.



18. Bokhari R, Baeesa S. Atlas hypoplasia and ossification of the transverse atlantal ligament: a rare cause of cervical myelopathy. Case Rep Neurol Med 2012 2012:893284.




19. Pascual-Gallego M, Budke M, Villarejo F. Spinal stenosis at the level of atlas in a boy with Down syndrome. A case report and literature review. Neurocirugia (Astur) 2014 25:29-32.


20. Steel H. Anatomical and mechanical considerations of the atlantoaxial articulations. J Bone Joint Surg Am 1968 50:1481-2.




























